Abstract
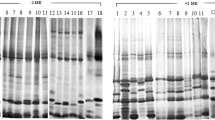
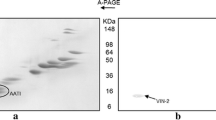
Zeins from Zea mays L cv. Maya and coixins from Coix lacryma-jobi L. cv. Adlay were fractionated to obtain α-, β-, and γ-zein and α-, β-, and γ-coixin. The α-coixins were composed of 4 polypeptide classes of 27 kDa (C1), 25 kDa (C2), 17 kDa (C4) and 15 kDa (C5) with solubility properties very similar to those of the 22 kDa and 19 kDa α-zeins. Like the α-zeins, the C1 and C2 α-coixins corresponded to 80% of total Coix prolamins. The fraction corresponding to γ-coixin contained only one protein band of 22 kDa (C3). This coixin fraction has solubility properties similar to those of γ-zein and represents 15% of the total coixin. The β-zein fraction was composed of a major 17 kDa protein band, while the β-coixin fraction consisted of a mixture of α- and γ-coixins.
Polyclonal antibodies raised against C1 recognized C1 and C2 and cross-reacted strongly with the 22 kDa α-zein, as did C4 and C5 antisera. The antiserum against γ-coixin showed strong cross-reaction with γ-zein. The homology between coixins and zeins was further investigated by using Southern hybridization analyses. The genomic DNA of maize and Coix were digested with several restriction enzymes and probed with cDNA clones representing 19 and 22 kDa α-zeins as well as the 28 and 16 kDa γ-zeins. The Coix genome showed complex cross-hybridization sequences with the 22 kDa α-zein cDNA, while no cross-hybridization was observed with the 19 kDa cDNA clone. The cDNA clone representing the 28 kDa γ-zein cross-hybridized with only one band of Coix genomic DNA, in contrast to the three bands observed in maize. This same Coix sequence also cross-hybridized with the cDNA clone representing the 16 kDa γ-zein. The relevance of these findings are discussed in the context of the origin of zein and coixin genes.
Similar content being viewed by others
References
Arora RK: Job's tears (Coix lacryma-jobi)—a minor food and fooder crop from north eastern India. Econ Bot 31: 358–366 (1977).
Burr B, Burr FA, St John TP, Thomas M, Davis RW: Zein storage protein gene family of maize. An assessment of heterogeneity with cloned messenger RNA sequences. J Mol Biol 154: 33–49 (1982).
Dretzen G, Bellard M, Sassone-Corsi P, Chambon P: A reliable method for the recovery of DNA fragments from agarose and acrylamide gels. Anal Biochem 112: 295–298 (1981).
Esen A: Separation of alcohol-soluble proteins (zeins) from maize into three fractions by differential solubility. Plant Physiol 80: 623–627 (1986).
Esen A, Bietz JA, Paulis JW, Wall JS: A 23.8-kDa alpha-zein with N-terminal sequence and immunological properties similar to 26.7-kD alpha-zeins. Plant Mol Biol 9: 421–430 (1987).
Feinberg AP, Vogelstein B: A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132: 6–13 (1983).
Hager DA, Burgess RR: Elution of proteins from sodium dodecyl sulphate, and renaturation of enzymic activity: results with sigma subunit of Escherichia coli RNA polymerase, wheat germ topoisomerase and other enzymes. Anal Biochem 109: 76–86 (1980).
Heidecker G, Messing J: Structural analysis of plant genes. Ann Rev Plant Physiol 37: 439–466 (1986).
Johnson DA, Gautsch JW, Sportsman JR, Elder JH: Improved technique utilizing non-fat dry milk for analysis of proteins and nucleic acids transferred to nitrocellulose. Gene Anal Techn 1: 3–8 (1984).
Laemmli UK: Cleavage of structural proteins during assembly of phage T4 head. Nature 227: 680–685 (1970).
Larkins BA, Pedersen K, Marks MD, Wilson DR: The zein proteins of maize endosperm. TIBS July: 306–308 (1984).
Ludevid MD, Martinez-Izquierdo JA, Armengol M, Torrent M, Puigdomenech P, Palau J: Immunological relations between glutelin-2 and low molecular weight zein-2 proteins from maize (Zea mays L.) endosperm. Plant Sci 41: 41–48 (1985).
Marks MD, Larkins BA: Analysis of sequence microheterogeneity among zein messenger RNAs. J Biol Chem 257: 9976–9983 (1982).
Melcher V: Heterogeneity of Zea mays protein body messenger RNA. Plant Sci Lett 18: 133–141 (1980).
Marks MD, Lindell JS, Larkins BA: Nucleotide sequence analysis of zein mRNAs from maize endosperm. J Biol Chem 260: 16451–16459 (1985).
Okita TW, Krishnan HB, Kim WT: Immunological relationship among the major seed proteins of cereals. Plant Sci 57: 103–111 (1988).
Ottoboni LMM, Leite A, Targon MLN, Crozier A, Arruda P: Characterization of the storage protein in seed of Coix lacryma-jobi var Adlay. J Agric Food Chem (In press).
Paulis JW, Wall JS: Fractionation and properties of alkylated-reduced corn glutelin proteins. Biochem Biophys Acta 251: 57–69 (1971).
Pedersen K, Devereux J, Wilson DR, Sheldon E, Larkins BA: Cloning and sequencing analysis reveal structural variation among related zein genes in maize. Cell 29: 1015–1026 (1982).
Plikaytis BD, Carlone GM, Edwards P, Mayer W: Robust estimation of standard curves for protein molecular weight and linear-duplex DNA based-pair number after gel electrophoresis. Anal Biochem 152: 346–364 (1986).
Prat S, Cortadas J, Puigdomenech P, Palau J: Nucleic acid (cDNA) and amino acid sequences of the maize endosperm protein glutelin-2. Nucl Acids Res 13: 1493–1504 (1985).
Prat S, Perez-grau L, Puigdomenech P: Multiple variability in the sequence of a family of maize endosperm proteins. Gene 52: 41–49 (1987).
Rivin CJ, Zimmer EA, Walbot V: Isolation of DNA and DNA recombinants from maize. In: Sheridan WF (ed) Maize for Biological Research, pp 161–164. University Press, University of North Dakota. Grand Forks, USA (1982).
Rubenstein I: The zein multigene family. In: Sheridan WF (ed) Maize for Biological Research, pp 189–195. University Press, University of North Dakota, Grand Forks, USA (1982).
Sodek L, Wilson CM: Amino acid composition of proteins isolated from normal, opaque-2, floury-2, corn endosperms by a modified Osborne procedure. J Agric Food Chem 19: 1144–1150 (1971).
Southern EM: Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98: 503–517 (1975).
Viotti A, Sala E, Alberi P, Soave C: Heterogeneity of zein synthesized in vitro. Plant Sci Lett 13: 365–375 (1978).
Vitale A, Smaniotto E, Longhi R, Galante E: Reduced soluble proteins associated with maize endosperm protein bodies. J Expt Bot 33: 439–448 (1982).
Wahl GM, Stern M, Stark GR: Efficient transfer of large DNA fragments from agarose gels to diazobenzloxymethal-paper and rapid hybridization by using dextran sulfate. Proc Natl Acad Sci USA 76: 3683–3687 (1979).
Wang SZ, Esen A: Primary structure of a proline-rich zein and its cDNA. Plant Physiol 81: 70–74 (1986).
Weinand U, Feix G: Zein specific restriction enzyme fragments of maize DNA. FEBS Lett 116: 14–16 (1980).
Wilson CM: Seed protein fractions of maize, sorghun and related cereals. In: Gottschalk W, Muller HP (eds) Seed Proteins: Biochemical, Genetics, Nutritive Value, pp 271–307. Martinus Nijhoff/Junk, The Hague, Netherlands (1983).
Wilson CM: Serial analysis of zein by isoelectric focusing and sodium dodecyl sulfate gel electrophoresis. Plant Physiol 82: 196–202 (1986).
Wilson CM, Shewry PR, Miflin BJ: Maize endosperm proteins compared by sodium dodecyl sulphate gel electrophoresis and isoelectric focusing. Cereal Chem 58: 278–281 (1981).
Wilson DR, Larkins BA: Zein gene organization in maize and related grasses. J Mol Evol 20: 330–340 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leite, A., Ottoboni, L.M.M., Targon, M.L.P.N. et al. Phylogenetic relationship of zeins and coixins as determined by immunological cross-reactivity and Southern blot analysis. Plant Mol Biol 14, 743–751 (1990). https://doi.org/10.1007/BF00016507
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00016507