Abstract
The incidence of HIV in Uganda as reported by UNAIDS (2012) was increased from 6.7% in 2004 to 7.6% to 2012. The main threat to HIV-infected patients following surgery is the development of sepsis. Inadequacy of surgical supplies and human resources further hastens and complicates the postoperative sepsis in HIV patients. The objective of the study was to determine incidence and risk factors associated with postoperative sepsis, among HIV seropositive with acute abdomen. A prospective study ran for a period of 11 months from October 2015 to April 2016 in Mulago Hospital in Kampala. Eligible patients were recruited and included. Study variables included postoperative wound sepsis, type of surgery, and CD4 counts. Thirty-eight data were collected using a questionnaire then entered in the Epidata software 3.1 and analyzed by Stata software version. Sixty-two patients were recruited; of these, 42 were male, 37 were HIV-negative and 25 were HIV-positive. The proportion of patients with postoperative sepsis in the HIV-positive group was 7 (28%) and in the HIV-negative group was 8 (21.6%). The number of patients discharged in HIV-positive group was 24 (96%) and in HIV-negative group was 35 (94.6%). Among the HIV-positive group was 1 out of 25 (4) % and HIV-negative was 2 out of 37 (5.4%). The overall postoperative sepsis incidence rate was 3 per 100 person days for under observation (95% CI 0.02–0.1), and the incidence rate ratio of HIV-positive patients and HIV-negative was 1.04 (95% CI 0.32–3.3; P = 0.47. The limited health resource was associated with developing postoperative sepsis. There was a higher risk of positive operative sepsis among HIV-positive compared to HIV-negative patients undergoing surgery for acute abdominal conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Globally, an estimated 35.3 million people were living with HIV in 2012. HIV infection was first recognized in Uganda in 1982 [1]. It has been a global pandemic and major cause of morbidity and mortality for over the last three decades. Sub-Saharan Africa bears the greatest burden of this disease with 68% of the global burden [2]. A study showed that HIV-infected patients with preoperative CD4 count ≤ 200 cells/μL had overall higher postoperative sepsis morbidity. Preoperative CD4 count was used as a useful indicator for postoperative sepsis in HIV-infected patients undergoing abdominal operations. Postoperative sepsis was only major postoperative event associated with long-term mortality. Postoperative sepsis may reflect a deep impairment of immune response, which is potentially associated with mortality [3]. An increase in ART usage from previous years is observed as more people are receiving the life-saving antiretroviral therapy. There were 2.3 (1.9–2.7) million new HIV infections globally, showing a 33% decline in the number of new infections from 3.4 (3.1–3.7) million in 2001. At the same time, the number of AIDS deaths is also declining with 1.6 (1.4–1.9) million AIDS deaths in 2012, dropping from 2.3 (2.1–2.6) million in 2005 [4]. The development of sepsis creates a substantial health care burden and limited epidemiologic information exists with regard to postoperative sepsis. Patients who undergo major surgery are at a high risk of postoperative sepsis. HIV-infected patients are more likely to develop postoperative infectious complications [5]. Severe sepsis in two Ugandan hospitals was found to have a mortality rate of up to 43%. In-hospital mortality was increased and the average length of stay was 6 days for those who survived to be discharged. Lack of surgical materials and human resources contributes to the inability to achieve improved management of sepsis in resource-constrained setting [6]. To a greater or lesser extent, the HIV disease now influences the performance and outcome of surgery in every country of the world. Surgical studies show that this impairment has not resulted in an increased incidence of postoperative infections or failure of wound healing in the asymptomatic HIV-positive patient. Current surgical management seems adequate to prevent increased risk of early postoperative infection in the symptomatic HIV-positive patient undergoing surgical procedures [7]. HIV-seropositive patients had more complications, including more perioperative infections, impaired wound healing, and greater mortality and these results were used as justification to withhold surgery in certain circumstances [8]. In this study, we estimated the incidence of postoperative sepsis and the associated risk factors among HIV-seropositive and HIV-negative patients presenting with acute abdomen.
Methods
Study Design
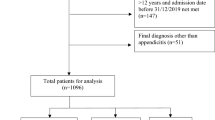
A prospective cohort study was carried out between 1st of October 2015 and 30th of April 2016 for a period of 7 months. The research was carried out in MNRH general surgical wards. MNRH is a teaching Hospital for Makerere University College of Health Sciences and also a National Referral Hospital located in Kampala, the capital city of Uganda. It is a 1500-bed capacity hospital and runs a specialized HIV unit. Mulago Hospital provides specialized inpatient care to patients referred from district and regional hospitals around the country and from various outpatient HIV care centers. Presentation of acute abdomen is one to three patients per day who undergo surgery in Mulago Hospital. HIV prevalence in the general adult population in Uganda is 7.3%, and approximately 64% of patients hospitalized in Mulago Hospital are infected with HIV. Participants were recruited from the surgical emergency ward as well as the general surgical wards of the hospital. Patients on A&E and the general surgical wards were suspected of having acute abdominal surgical manifestation; HIV-seropositive and HIV-negative patients at MNRH were interviewed and enrolled in the study after the patient signed a written consent form (Fig. 1). So all HIV-positive and HIV-negative patients with acute abdominal surgical manifestation taking ART or not were recruited in the study at MNRH and they were part of the target population. All patients (women, men, and > 15 years of age) who were present at MNRH were directly or referred with surgical manifestation. All acute abdomen patients above 15 years of age received at MNRH between 1st of October 2015 and 30th of April 2016 gave informed consent (for adults 18 years and above) and assent plus guardian consent (for patients younger than 18 years) to participate in the study. Unconscious surgical patients, whose attendants refused to participate in the study. Mentally ill, and unstable patients.
Sampling Procedure
Patients admitted to the surgical unit of the Accidents and Emergency department and general wards with acute abdomen were recruited to this study according to the eligibility criteria. An HIV test was performed using the Determine method which is rapid test. Any positive test was confirmed using Statpak and Unigold. CD + 4 counts and CBC were obtained before surgery for all HIV-positive patients. The participants were grouped as HIV-positive or HIV-negative and henceforth analyzed in those groups. The initial diagnosis at first assessment was recorded. The final diagnosis after investigations and/or surgery were also recorded. In this study, we used consecutive selection.
Sample Size
The sample size was determined using the Kish and Leslie formula (1965), n = 63 patients. Our dependent variables were sociodemographic data: age, sex, address, level of education, occupation, HIV status, acute abdomen diagnosis, CD4 level, ART drugs, duration of surgery, duration of antibiotic use, type of surgery, and health system challenges. Our dependent variable was postoperative sepsis.
Data Management
We pre-coded and pre-tested data collection tool. The raw data then was entered using Epidata Entry Version 3.1. We collected data by using an administered questionnaire. We tested HIV patients with acute abdomen using the Determine test, and confirmed using Statpak. Any discrepancies were resolved with a third tie-breaker test—Unigold. We have done CD4 count, CBC, consent was obtained, and their contacts taken. At this stage, the questionnaire was partially filled and fully completed after surgery and follow-up. The informed consent process as well as questionnaire interviews were conducted in the local language (Luganda) for the majority of patients and in English for a few patients. We collected data over a specified period of time and interviewed at the arrival time, except patients who were unstable such as those in coma or shock. We summarized data and presented it in tables for easy interpretation. Epidata version 3.1 was used for the entry, validation, and consistency of data; double data entry to check for errors was done. We analyzed data by STATA version 12 computer program and was presented in tabulation form and frequency graphs. It was summarized as tables, pie charts, and bar graphs. In the analysis described, patients are characterized and presented as percentage (%) and mean (SD). The incidence of HIV patients calculated as the proportion of all patients with acute abdomen who are HIVpositive were presented as number (percentage) or mean (SD). The incidence of postoperative sepsis used 95% CI. We divided the participants into two groups, HIV-positive and HIV-negative. The proportions of the various identified etiologies of acute abdomen were calculated for each group. The chi-square statistic was used to test for significance of the difference in the proportions. Similarly, the postoperative risk factors associated with acute HIV accompanying acute abdomen were compared between the two groups. The incidence of postoperative sepsis was estimated as the proportion of patients who had HIV presenting with acute abdomen among the study participants. Logistic regression was used to identify factors associated with HIV presenting with acute abdomen by deriving the odds ratio (CI) and p value of the respective relationships. In all analyses, a p value < 0.05 was considered statistically significant. The postoperative sepsis and associated risk factors used Poisson regression methods to estimate the incidence rate of occurrence of postoperative sepsis among HIV patients as compared to HIV-negative and used incidence relative risk (IRR) (95%CI). Analytics were used to assess the confounders using the same regression method. We pretested the questionnaire and cross-checked for completeness and internal consistency. Research assistant was trained. Inconsistent data evaluated and necessary adjustments were made. Approval was obtained from Makerere University College Board (IRB); the nature of study, its potential risks, and benefits to the patients on the community were explained to the patients. The consent forms were in both Luganda and English. Written informed consent was obtained by signature or thumbprint using a consent form.
Results
A total of 62 patients were recruited and followed-up to discharge or death from 1st of October 2015 to April 2016. The median age and inter quartile of HIV-negative patients was 35 (IQR, 25) while the HIV-positive group was 37 (IQR, 8). The urban residents were 22 out of 37 (59.46%) in HIV-negative and 15 out of 25 (59.68%) in HIV-positive while rural was 15 out of 37 (40.54%) in HIV-negative and 10 out of 25 (40) in HIV-positive. Most of the patients were married, 26 out of 37 (70.26%) in HIV-negative group and 20 out of 25 (74.19%) for HIV-positive followed by the single status which presented 11 out of 37 (29.73%) and 4 out of 25 (16%) respectively. The educational level for most of study participants were Secondary School level both HIV-negative and HIV-positive 56.76% and 52% respectively. Occupation was professional with 16 (43%) in HIV-negative and 8 (32%) in HIV-positive; businessmen, HIV-negative 5 out of 37 (13.51%), HIV-positive 8 out 25 (32%); and peasant, HIV-negative 7 out of 37 (18.92%) and HIV-positive 8 out of 25 (32%) respectively. The most common presentation was abdominal pain. The HIV-negative group with 33 out 37 (89.19%), and HIV-positive with 24 out of 25 (96%). Within the HIV-positive group, the most common condition was intestinal obstruction at 12 out of 25 (48%) followed by acute appendicitis which presented in 5 out of 25 (20%). In the HIV-negative participants, obstructed hernia was most common condition in 14 out of 37 (37%) followed by intestinal obstruction in 10 out of 37 (27%). Anemia was the most common co-morbid association condition for HIV-positive and HIV-negative patients with 7 out 37 (18%) and 5 out 25 (20%) respectively. Exploratory laparotomy was the most frequently performed procedure in HIV-negative representing 22 out of 37 (59.56%) and HIV-positive with 15 out of 25 (56%) respectively. Baseline demographic characteristics of the study participants are shown in Table 1.
Finding at Laparotomy
The most common finding at surgery was gut perforation observed 19 out of 62 (30.64%) operations. Most of the perforations were observed in the small intestine in 14 cases and the stomach in 4 cases. Incarcerated or strangulated hernia, is the second most common finding observed in 16 out of 62 (25.8%) patients. Small intestine obstruction was the third most common finding observed in 11 out of 62 (17.7%) and was caused by bands, adhesions, or pyoperitoneum. Inflamed appendix in 6 out of 62 (9.7%) and sigmoid volvulus in 5 out of 62 (8.1%) ranked 4th and 5th respectively.
Postoperative Outcomes
Fifteen out of 62 patients (24.2%) in the study developed postoperative sepsis, whom 7 out of 25 (28%) were HIV-positive and 8 out of 37 (21.62%) were HIV-negative. Twenty-four out of 25 (96%) of the HIV-positive patients were discharged compared to the 35 out of 37 (94.59) of the HIV-negative patients. Patients died following surgery were 1 out of 7 (4%) of whom was HIV-positive and 2 out of 37 (5.4%) were HIV-negative.
Incidence of Postoperative Sepsis
The total person time of the study participants was 489 and patients who developed postoperative sepsis were 15 cases. The overall incidence rate was 3 per 100 person days under observation (95% CI 0.02–0.051). The incidence rate of postoperative HIV-positive patients was 3.1 per 100 person days under observation while that in HIV-negative was 3.0 per 100 person days under observation; the person time in HIV-negative group was 265 while in the HIV-positive was 224. The incidence rate ratio for HIV-positive and HIV-negative was 1.035 (95% CI 0.319–3.29; P = 0.47).
Study Variables
Most emergency cases were operated by surgical residents, i.e., 54 out 62 (87%) cases with 3 years mean experience followed by those done by the Registrar (junior consultants) with 6 out of 62 (8.1%). Antiseptic solution was used in all cases except in one case where normal saline was used as an antiseptic solution. Most HIV-positive patients in this study were in stage І, 23 out of 25 (92%). Most of the HIV patients were on HARRT, 18 out of 25 (68%) followed none, and 8 out of 25 (32%) used Septrin. All patients who developed postoperative sepsis during this study presented in 1 out of 15 (6.7%). Most surgeons who operated patients in this study were experienced but faced a problem of limited health resources like unavailability or shortage of surgical instruments, gowns, antiseptic solution, and anesthesiologist. Most of the patients operated in this study had been given preoperative antibiotics for an average of 1.7 ± 2.13 days, the minimum was 1 day and the maximum was 22 days. The mean duration of surgery was 1.7 ± 0.6 h, with a minimum of 1 h and maximum of 3 h. Surgical experience ranged from 1 to 30 years with a mean of 4.7 ± 5.3 years. SHO were the least experienced surgeons. The mean duration of symptoms was 5.4 ± 3.73 days with a minimum of 2 days and maximum of 14 days. Mean length of hospital stay was 7.88 ± 8.4 days with a minimum of 1 day and maximum of 37 days. Sixteen out of 25 (64%) HIV-positive patients who had CD4 count, ranged from 200 to 500 uL. These were protected from getting postoperative sepsis (IRR 0.678; 95% CI 0.152—0.03; P = 0.611). Patients who had CD4 > 200 uL were 7 out of 25 (28%) and more protected CD4 > 500 uL were 2 out of 25 (8%).
Risk Factors for Postoperative Sepsis in HIV+ Participants
We assessed for factors associated with postoperative sepsis using Poisson regression model. Bivariate analysis factors which had P value of 0.2 were considered statistically significant and included in the multivariate Poisson regression model. At multivariate analysis, factors were considered to be statistically significant if they had a P value of 0.05. The health system challenges were found to have an association with postoperative sepsis WBC, platelets, duration of surgery were included. The risk of postoperative sepsis increased by 3.17 times for every additional hour spent in surgery (95% CI 10.66–15.15; P = 0.14). Patients who had leukocytosis were 1.11 times (95% CI 0.951–1.31; P = 0.17) more likely to develop sepsis.
Bivariate Analysis of Risk Factors for Postoperative Sepsis HIV− Participants
Married patients were less likely to develop postoperative sepsis (IRR 0.39; 95% CI 0.138–1.14; P = 0.08). Duration of surgery increased (IRR 3.34; 95% CI 1.04–11.5; P = 0.043) folds of getting postoperative sepsis. Patients who had anemia had increased incident-related risk (IRR 23.4; 1.25; P = 0.18) of developing sepsis. The status of being married, duration of surgery, and limited health resources were associated with postoperative sepsis and were presented with a P value less than 0.2 which were taken for multivariate analysis.
Multivariate Analysis of Risk Factors for Postoperative Sepsis among HIV-Positive and HIV-Negative Participants
After controlling for CD4 < 200,WBC, and platelets, the risk of having postoperative sepsis among HIV-positive patients was 2.1 times higher among patients whose health care worker experienced limited health resource (IRR 3.6; 95% CI − 0.2–4.4; P = 0.072). The risk factors for postoperative sepsis among HIV-negative. The controlling factors were absence of core staff and unavailability; these were not statistically and significantly associated with postoperative sepsis (IRR 3.5; 95% CI 0.61–207; P = 0.500) as shown in Table 2.
Discussion
We set out to find the incidence and risk factors of postoperative sepsis among HIV patients presenting with acute abdomen at Mulago Hospital. We found that the overall incidence rate of postoperative sepsis was 0.03 per person days under observation (IR 0.03; 95% CI 0.02–0.051). This is equivalent to three cases of postoperative sepsis per 100 person days under observation. The incidence rate ratio for postoperative sepsis in HIV-positive versus HIV-negative participants was 1.035 (95% CI 0.319–3.29; P = 0.47). King et al. in a similar study done in a veterans’ hospital in the USA found an incidence rate ratio of 2.11 (95% CI 1.41–3.17; P < 001) [9]. This rate was higher than that in our study possibly because they followed-up patients for a longer period. It is possible that some patients developed postoperative sepsis in the days after being discharged from hospital. Such patients would be missed by our study which followed-up participants up to discharge or death. Some studies presented data as incidence and percentages of patients with postoperative sepsis. In the discussion below, we discuss our findings using statistics presented by other studies in order to aid the comparison. Nandyala et al. [10], in a study done in India, found the postoperative sepsis was 30%. From our study, the incidence of postoperative sepsis was 28% (7 out of 25). Zhang et al. [11], in a study done in China, found an incidence of postoperative sepsis of 2.6% in HIV-infected patients versus HIV-negative patients. Mafana et al. [12] collected data from six different hospitals in South Africa and found an incidence of postoperative sepsis of 3.6% among the HIV-negative patients and 3.7% among HIV-postoperative sepsis. These were lower than what we found in our study probably because of differences in study setting and study patients.
Risk Factors for Postoperative Sepsis
In all study participants irrespective of HIV status, longer duration of surgery increased the risk of postoperative sepsis. The risk of postoperative sepsis increased threefold for every extra hour spent under operation (IRR 3.4; 95% CI 0.66–15.5; P = 0.14) but this finding was not statistically significant. In a similar study done by Berthus et al. in the Netherlands, researchers also found that longer duration of surgery increased risk of developing postoperative sepsis [13]. This finding was statistically significant with P value as 0.01. It is possible that surgery deplete patients’ energy levels and affects the hemodynamics required to fend off infection. It is also possible that longer duration of surgery exposes patients to the risk of contamination and infection especially if the conditions under which surgery is performed have reduced sterility. In this study, we found that HIV patients were 2.1 times more likely to develop postoperative sepsis, where health workers reported limited health resources such as lack of sundries, drapes, antiseptic solution, gowns, and other materials necessary to care for patients in the postoperative period (IRR 13.9; 95% CI 1.5–130) and this finding was statistically significant (p = 0.021) after controlling all other possible confounders. Indeed, other studies have found results similar to ours. Kizza et al. found that lack of material and human resources contributes to the inability to achieve improved management of sepsis in Mulago Hospital. The high mortality associated with sepsis syndromes resulted from the lack of materials and human resources necessary for optimal care [14]. Having anemia in which we used WHO guideline < 9 mg/dl have increased the risk of postoperative sepsis 4.5 times (IRR, 95% CI 1.5–13.1; P = 0.006) at bivariate analysis although this finding was not statistically significant after controlling other factors (IRR 1.66; 95% CI 0.99–27.8, p = 0.72). It is possible that there are other factors confounding the relationship between anemia and postoperative sepsis that have to be explored further to tease out the actual effect of anemia on the development of postoperative sepsis. We found that having a higher CD4 cell count reduced the risk of developing postoperative sepsis among patients who were HIV-positive (IRR − 0.31; 95% CI − 1.96–1.3; P = 0.7), for HIV-positive patients with CD4 count between 200 and 500 versus patients with CD4 count < 200/μl. This finding is in close agreement with a similar study done in China by Xia et al. who found incidence of postoperative sepsis in patients with preoperative CD4 count < 200cells/μl marked higher than those with CD4 counts > 200/μl (83.3% versus 17.65%) [15].
Clinical Findings and Findings at Surgery Among Study Patients
Almost all patients had abdominal pain (96%) and abdominal distention (81%). Similar to a Tanzanian study by Chalya et al., it was found that abdominal pain was the main clinical presentation; hospital stay in Chalya et al. study was 26 days, which was greater than the median duration of hospital stay in our study of 7.8 (IQR, 8.4 days) [16]. In our study, the most common finding at surgery was gut perforation 30.64% and this consistently agreed with findings from studies done by Clark et al. in South Africa [17], Weledji et al. from Nigeria [18], and Nandyala et al. from India (10) where the most common finding was gut perforation presenting in 30% of patients. Galukande et al. found appendectomy followed by hernia, and there was similarity to this study whereby the second most operation was herniorrhaphy [19]. In our study, strangulated hernia was found in 17.7% of patients; a finding similar to that was found by Doumgba et al. in Central Africa in which he found 16% of cases had strangulated hernia [20]. The mean hemoglobin of our study was 12.3 mg/dl and it was similar to the study done by Ischie et al. in Nigeria [21] and Okuma et al. [19] in Uganda which they found 12.3 mg/dl and 12.1 mg/dl respectively. CD4 < 200/μl in our study was associated with postoperative sepsis which Misauno et al. found the same in Nigeria [21]. In our study, the proportion of postoperative sepsis in HIV-positive patients was 28% similar to what Montoya et al. found in Peru in 2014 which was 28.1% [22].
Comparison of Incidence of Postoperative Sepsis Among HIV-Positive and HIV-Negative Patients
The incidence rate of postoperative sepsis in this study for HIV-positive patients was 3.1/100 person days under observation while that of the HIV-negative patients was 3.0/100 person days under observation. This gave an incidence rate ratio of 1.1 for HIV-positive versus HIV-negative patients (IRR 1.035; 95% CI 0.32–3.30; p = 0.47) but this finding was not statistically significant. Other studies presented data as incidence instead of incidence rates. Our study showed higher incidence of 7 out of 25 (28%) in the HIV-positive group compared to similar studies done elsewhere. A study done by Zhang et al. showed an incidence of postoperative sepsis of 2.6% in HIV patients [11]. Hadnott et al. [23] and Yiiet et al. [24] found incidence of sepsis of 13% (17 out of 51 patients).
The proportion of patients with postoperative sepsis in HIV-positive patients was 28% (7 out of 25) and 21.62% (8 out of 37) in HIV-negative patients. In comparison, a study done by out of 2 (20%) and in the HIV-negative group was 7 out of 76 (9.2%) [19]. Kakande et al. found postoperative sepsis in 7 cases out 76 (9.9%) [25].
Study Limitations
Most patients were from Kampala metropolitan so generalization of results to the country is limited. The study was subjected to the potential of recall bias, e.g., age and duration of symptoms. Some of the patients were not able to remember, we minimized this by asking patients’ ages, and duration of symptoms, and cross-checking with the admission file. Duration of operation, antibiotic usage, and antiseptic solution were obtained from admission files. In case they were not written or reported, information was obtained from the surgeon. This did not have any net effect on the internal validity of the results.
Conclusion
The incidence rate ratio for HIV-positive and HIV-negative patients was 1.035 (95% CI 0.319–3.29; P = 0.47). Incidence rate of HIV-positive patients was 3.1 per 100 person time under observation compared to HIV-negative patients 3.0 per 100 person time under observation. Risks of postoperative sepsis among both HIV-positive and HIV-negative patients increased by longer duration of surgery, anemia, and limited health resources.
Availability of Data and Materials
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Kabenge C, Ng S, Muyinda Z, Ameda F. Diagnostic ultrasound patterns of parotid glands in human immunodeficiency virus-positive patients in Mulago, Uganda. DMFR. 2014;39(7):389–99.
Namutebi A, Kamya M, Byakika-Kibwika P. Causes and outcome of hospitalization among HIV-infected adults receiving antiretroviral therapy in Mulago hospital, Uganda. Afr Health Sci. 2014;13(4):977–85.
Su J, Tsun A, Zhang L, Xia X, Li B, Guo R, et al. Preoperative risk factors influencing the incidence of postoperative sepsis in human immunodeficiency virus-infected patients: a retrospective cohort study. World J Surg. 2013;37(4):774–9.
Priscilla Idele P, Gillespie A. Global report: UNAIDS report on the global AIDS epidemic 2013. Geneva, Switzerland: UNAIDS; 2013. Joint United Nations Programme on HIV/AIDS Tropical Medicine journal, Lippincott Williams & Wilkins. 2014;66:144–53.
Liu B-c, Zhang L, J-s S, Tsun A, Li B. Treatment of postoperative infectious complications in patients with human immunodeficiency virus infection. World J Emerg Med. 2014;5(2):103–6.
Jacob ST, Moore CC, Banura P, Pinkerton R, Meya D, Opendi P, et al. Severe sepsis in two Ugandan hospitals: a prospective observational study of management and outcomes in a predominantly HIV-1 infected population. PLoS One. 2009;4(11):7782–4.
Luck J Jr. Orthopaedic surgery on the HIV-positive patient: complications and outcome. Eur PMC pus. 1993;43(9):543–9.
Smith L. Gynaecological surgery in the HIV-positive patient. S Afr Med Assoc J. 2010;28(8):372–5.
King JT, Perkal MF, Rosenthal RA, Gordon AJ, Crystal S, Rodriguez-Barradas MC, et al. Thirty-day postoperative mortality among individuals with HIV infection receiving antiretroviral therapy and procedure-matched, Uninfected Comparators. JAMA Surg. 2015;150(4):343–51.
Nandyala VNR, Pallam P, Babu S, Kundarapu G, Pallagani L. Surgical emergencies in a HIV positive patient. Evid Based Med Healthc. 2015;3(25):1131–4.
Zhang L, Liu B-C, Zhang X-Y, Li L, Xia X-J, Guo R-Z. Prevention and treatment of surgical site infection in HIV-infected patients. BMC Infect Dis. 2012;12(1):1471–2334.
Čačala S, Mafana E, Thomson S, Smith A. Prevalence of HIV status and CD4 counts in a surgical cohort: their relationship to clinical outcome. Ann R Coll Surg Engl. 2006;88(1):46–51.
Bennett-Guerrero E, Welsby I, Dunn T, Young L, Wahl T, Diers T, et al. The use of a postoperative morbidity survey to evaluate patients with prolonged hospitalization after routine, moderate-risk, elective surgery. Anesth Analg. 1999;89(2):514–9.
Jacob ST, Banura P, Baeten JM, Moore CC, Meya D, Nakiyingi L, et al. The impact of early monitored management on survival in hospitalized adult Ugandan patients with severe sepsis: a prospective intervention study. Crit Care Med. 2012;40(7):2050–8.
Xia X-j, Liu B-c, Su J-s, Pei H, Chen H, Li L, et al. Preoperative CD4 count or CD4/CD8 ratio as a useful indicator for postoperative sepsis in HIV-infected patients undergoing abdominal operations. J Surg Res. 2012;174(1):25–30.
Chalya PL, Mabula JB, Chandika AB, Giiti G. Dynamic bowel obstruction: aetiology, clinical presentation, management and outcome at Bugando Medical Centre, Mwanza, Tanzania. Tanzan J Health Res. 2014;16(1):1–9.
Islam J, Clarke DL, Thomson SR. Lessons from emergency laparotomy for abdominal tuberculosis in the HIV/AIDS era. S Afr J Surg. 2014;52(1):10–2.
Weledji EP, Nsagha D, Chichom A, Enoworock G. Gastrointestinal surgery and the acquired immune deficiency syndrome. Ann Med Surg. 2015;4(1):36–40.
Okuma G, Makobore P, Kaggwa S, Kambugu A, Galukande M. Effect of emergency major abdominal surgery on CD4 cell count among HIV positive patients in subsaharan african tertiary hospital. BMC Surg. 2013;13(1):1471–2482.
Doumgba A, Kamoune J, Ngarhio L, Zoguereh D, Dan-Houron J, Nali N. Surgery and HIV in Bangui (Central African Republic). Sante (Montrouge, France). 2005;16(3):173 -.
Isichei M, Misauno M, Shitta A, Isichei C. Outcome of emergency surgery in HIV infected patients in Jos. IOSR J Dent Med Sci. 2014;13(4):21–3.
Montoya L, Rodríguez E, Zúñiga G, Yamamoto G, González E. Acute abdomen in patients with HIV/AIDS seen in a national hospital of Lima, Peru. Rev Peru Med Exp Salud Pública. 2014;31(3):515–9.
Hadnott TN, Dawes A, Strength K, Moran G, Gibbons MM, Holschneider CH. Risk of sepsis in the postoperative setting [48]. Obstet Gynecol. 2015;125(48):24–8.
Yii MK, Saunder A, Scott DF. Abdominal surgery in HIV/AIDS patients: indications, operative management, pathology and outcome. Aust N Z J Surg. 1995;65(5):320–6.
Kitara D, Kakande I, Mugisa B, Obol J. The postoperative complications prediction in Mulago Hospital using POSSUM scoring system. East Central Afr J Surg. 2010;15(2):35–65.
Loef BG, Epema AH, Smilde TD, Henning RH, Ebels T, Navis G, et al. Immediate postoperative renal function deterioration in cardiac surgical patients predicts in-hospital mortality and long-term survival. J Am Soc Nephrol. 2005;16(1):195–200.
Funding
This work was supported by Maryam Ali and Abdullahi Awale. The funders has no role in the design and conduct of the study, collection, management, analysis and interpretation of the data.
Author information
Authors and Affiliations
Contributions
AMA collected data and took responsibility for integrity of the data and the accuracy of data analysis, acquisition analysis, and drafting of manuscript. MG is DELTA/THRIVE fellow under DEL15-011/07742/Z/15/Z, TM, and GR participated in study design data analysis and manuscript writing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declared that they have no conflict of interest.
Ethics Approval and Consent to Participate
This study was approved by institute research board (2016-03-03REC REF 2016–033). The written informed consent was obtained from all individual participants included in the study.
Consent for Publication
Written consent for publication was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Surgery
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Awale, M.A., Makumbi, T., Rukundo, G. et al. Postoperative Sepsis Among HIV-Positive Patients with Acute Abdomen at Tertiary Hospital in Sub-Saharan Africa: a Prospective Study. SN Compr. Clin. Med. 1, 465–472 (2019). https://doi.org/10.1007/s42399-019-00061-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42399-019-00061-w