Abstract
Introduction
Psoriatic arthritis (PsA) is underdiagnosed and has a substantial impact on quality of life, disability, and work productivity. The population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey examined the impact of PsA on patients’ activities of daily living and unmet treatment needs.
Methods
This large-scale, random digit dialing, telephone survey of patients self-reporting a diagnosis of psoriasis and/or PsA was conducted in North America and Europe.
Results
In all, 3426 patients participated in the survey, including 712 (21%) who identified themselves as having PsA. Over half of the patients reported severe PsA involving more than four joints. Eighty-three percent of patients with PsA visited a health-care provider within the past 12 months. Approximately one-quarter saw their primary care provider or dermatologist most often for their disease; 37% responded that their rheumatologist was the health-care provider seen most often for PsA. Patients with PsA reported a substantial impact of disease on physical function. One-third of patients with PsA reported missing work because of their disease and PsA impacted their ability to work full time. Over half of the patients with PsA (58%) reported receiving no treatment or topical therapy only, leaving their joint disease untreated. Factors associated with lack of adherence were perceived lack of efficacy and concerns about long-term safety.
Conclusions
The MAPP survey confirms that PsA has a significant impact on physical function and activities of daily living. Undertreatment of PsA suggests a need for improved screening and diagnosis as well as education about treatment options and adherence.
Funding
Celgene Corporation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Psoriatic arthritis (PsA) is a chronic, systemic inflammatory disease, with symptoms that typically include swelling and pain in the peripheral joints, enthesitis, and dactylitis. The clinical presentation of PsA varies. It occurs in approximately 30% of patients with psoriasis [4, 12, 16, 25, 28] and typically follows the onset of psoriasis by approximately 8–10 years [12, 22]. Nail involvement typically occurs more often and is more severe in patients with PsA than in those with psoriasis alone and may predict the development of PsA in patients with psoriasis [10, 12, 16, 20, 24, 30, 35].
The burden of ongoing skin and joint inflammation characteristic of PsA is often overwhelming. In particular, the disease negatively affects quality of life (QoL) and work productivity, and leads to high rates of health-care utilization [4, 24, 32, 33]. Patients with PsA report reduced QoL and have more work-related problems, health-care visits, and hospitalizations than patients with psoriasis alone [4]. The progression of joint disease is associated with greater disability, and QoL sharply declines as limitations in daily activities increase [24, 32]. Likewise, PsA is associated with increased medical costs and loss of work productivity as the level of severity increases [4]. In addition, evidence suggests that persistent systemic inflammation fosters and worsens the severity of other co-morbidities, such as obesity and heart disease [4, 6, 11, 15].
The Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP), a population-based survey of 3426 patients and nearly 781 physicians in North America and Europe, is the largest probability survey of psoriatic disease conducted to date [22, 23, 34]. Here, we explore the results compiled from the subset of patients with PsA participating in the MAPP survey. We hypothesized that PsA would have a substantial impact on disease burden in the subset of patients with PsA in the MAPP survey.
Methods
Abt SRBI, Inc. (New York, NY, USA) conducted the MAPP survey between June and August 2012 [22]. Via telephone, Abt SRBI staff surveyed households in the USA, Canada, France, Germany, Italy, Spain, and the UK. The MAPP survey followed the American Association for Public Opinion Research guidelines for developing and conducting surveys [1]. Institutional review board approval was granted for data collection in the USA (Abt SRBI 5454), and the other countries abided by standards and ethical practices according to their codes or guidelines.
Random digit dialing was performed to provide a probability sample, and participants were invited to complete the survey if they were ≥18 years of age and were ever diagnosed with psoriasis and/or PsA by a health-care provider (HCP). The survey questions used have been published previously [22] and included questions regarding types and severity of psoriasis and/or PsA symptoms, factors contributing to the severity of disease, if and how psoriasis and/or PsA affects activities of daily living and work, interactions with HCPs for psoriasis and/or PsA, current and prior treatments, and factors affecting the choice of treatments.
Survey questions related to joint symptoms and physical function are provided in Table 1.
Results
Patients and Disease History
A total of 139,948 households were screened, and 3426 patients agreed to participate in the MAPP survey. Of the overall MAPP patient population, 712 respondents (21%) reported a diagnosis of PsA. The mean age of the patients with PsA was 56 years, the majority were female, and nearly three-quarters reported being overweight (33.6%) or obese (36.4%; Table 2). Co-morbidities were common in patients with PsA, with arthritis (unspecified), hypertension, depression, diabetes, and heart disease reported most often. The average age of patients at the time of PsA diagnosis was 42 years, with an average delay between the onset of joint symptoms and diagnosis of PsA of 5 years. Most patients with PsA (72%) developed skin signs and symptoms first, but 21% reported that they had joint signs and symptoms first, and 7% reported that skin and joint signs and symptoms appeared concurrently. PsA was diagnosed on average 8 years after psoriasis; 27% of the surveyed patients reported that they were diagnosed with psoriasis and PsA at the same time.
Current Symptoms
At the time of the survey, 88% of patients reported current joint pain or soreness; of these, 60% reported that more than four joints were affected (Fig. 1a). Of the patients reporting joint pain, 41% had knee pain, 26% had finger pain, 19% had hip pain, 19% had ankle pain, 18% had back pain, and 16% had wrist pain. In all, 31% of patients reported symptoms resembling enthesitis (i.e., pain/swelling of heel), and 45% reported symptoms resembling dactylitis (i.e., having “sausage” digits). In addition, patients reported itching (62%), scales (58%), flaking (58%), redness (58%), and pain (51%) associated with psoriasis lesions. Nail symptoms were present in nearly twice as many patients with PsA (21%) compared with patients with psoriasis alone (11%). Scalp involvement was reported by 44% of patients with PsA and 48% with psoriasis alone.
Self-perceived Severity
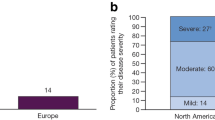
Patients were asked to rate their disease severity at its worst on a scale of 1 (very mild) to 10 (very severe). More than half of the patients with PsA (53%) rated their disease as severe (score: 8–10), 35% rated their disease as moderate (score: 4–7), and 12% rated their disease as mild (score: 1–3; Fig. 1b). In comparison, 27% of psoriasis patients rated their disease as severe [22]. As expected, the most important factor affecting self-perceived severity in patients with PsA was pain or swelling of the joints (45%); other commonly reported factors included itching (18%), location and size of skin lesions (10%), and lack of sleep (7%).
Impact on Physical Function: the HAQ-8
The proportions of PsA patients answering “with much difficulty” or “unable to do” on the eight-item Health Assessment Questionnaire (HAQ-8 or modified HAQ [27]) progressively increased for those with more than four affected joints and those with symptoms resembling enthesitis or dactylitis; patients with enthesitis or dactylitis reported the most disability on all items of the HAQ-8 (Fig. 2a). Likewise, the proportions of patients with PsA answering “with much difficulty” or “unable to do” increased for most questions in patients with higher palm counts (Fig. 2b), suggesting that skin disease also contributes to disability.
Impact of PsA on the 8-item Health Assessment Questionnaire and the proportion of patients responding “with much difficulty” or “unable to do” (a), and impact of psoriasis palm count on the 8-item Health Assessment Questionnaire in patients with PsA and the proportion of patients responding “with much difficulty” or “unable to do” (b). PsA psoriatic arthritis
Employment Status and Work Impact
PsA severely impacted patients’ work productivity (Fig. 3). Nearly one-third of patients with PsA (31.5%) reported that they had missed work in the past 12 months because of their disease. Similarly, 31.6% reported that PsA had impacted their ability to work full time. Approximately one-quarter of patients with PsA reported “a lot” or “some” impact of their disease on getting a job, keeping a job, and choice of career. These results are consistent with survey results of patients with psoriasis, which found that patients with psoriasis were significantly more likely to miss work time and to experience impairment while working compared with controls [36].
Medical Care and Treatment
In all, 83% of patients visited an HCP in the past 12 months for PsA, and 71% were evaluated by a rheumatologist at some point since their diagnosis. Moreover, they were followed specifically for PsA by different types of physicians: rheumatologist (37%), primary care provider (28%), and dermatologist (24%). When analyzed by country, the proportions of patients seen by rheumatologists for PsA ranged from 26% in Germany to 53% in Italy and Spain, and the proportions of patients seen by dermatologists for PsA ranged from 13% in the UK to 46% in Germany. Those patients who had not visited an HCP in the past 12 months reported that they did not believe an HCP could help (30%), their symptoms were not bad enough (30%), symptoms were absent (20%), current treatment was working (7%), and cost- or insurance-related issues were a factor (5%).
Current Treatment
More than half of the patients with PsA (59%) reported that they were receiving no treatment or topical therapy only and were not receiving any systemic medication for their joint disease. Overall, the proportions of patients with PsA who reported current treatment were 31% for topical therapy only, 19% for conventional oral therapy, 14% for biologic therapy, and 8% for the combination of oral and biologic therapy. Although these treatment rates were low for patients with PsA, they were higher than the treatment rates reported by patients with psoriasis, of whom only 5% were receiving conventional oral therapy [disease-modifying antirheumatic drugs (DMARDs)] and 3% were receiving biologic therapy at the time of the survey [22].
Conventional oral therapies that patients with PsA (n = 196) reported receiving included methotrexate (69%), cyclosporine (6%), soriatane (3%), fumaric acid (2%), sulfasalazine/sulfazine (2%), celecoxib (1%), folic acid (1%), prednisone (1%), and other (14%). Injectable medications that patients with PsA (n = 160) reported receiving included adalimumab (35%), etanercept (34%), infliximab (11%), ustekinumab (6%), methotrexate (5%), golimumab (4%), certolizumab (3%), alefacept (1%), and other (2%).
Treatment Burden
Conventional Oral Therapy (DMARDs)
More than 50% of patients who had received oral DMARDs found it to be burdensome, with one-third of patients reporting that the therapies were very or moderately burdensome. The most common reason patients found oral DMARDs burdensome was side effects; other reasons included the need for laboratory monitoring (Fig. 4a). Methotrexate was the oral DMARD most commonly reported as current therapy (n = 100), and 34% of patients reported it as “very burdensome.” The most common reasons patients found methotrexate burdensome were side effects (69%) and need for laboratory monitoring (26%). The most common reasons patients discontinued conventional oral therapy were lack or loss of effectiveness and lack of tolerability (Fig. 4b).
Biologic Therapy
Among patients who received injectable biologic therapy, 54% found it to be burdensome, with nearly one-third reporting that these medications were very or moderately burdensome. The most common reasons patients with PsA found biologic therapy burdensome were side effects (25.8%), fear of injections (22.7%), and inconvenience (9.1%; Fig. 4c). Adalimumab and etanercept were the injectable biologics most commonly reported as current therapy. The most common reasons patients with PsA discontinued biologic therapy included lack or loss of effectiveness and cost or insurance issues (Fig. 4d).
Overall, 64% of patients with PsA expressed concern about the health risks associated with long-term therapy, and 90% of patients with PsA felt there was a need for better therapies for psoriasis and PsA.
Discussion
The MAPP survey is the largest multinational probability survey of patients with psoriasis and PsA to date [22, 23, 34]. PsA was reported in 21% of the overall MAPP patient population, which is generally consistent with published prevalence estimates [4, 12, 16, 25, 28].
PsA was likely present, but undiagnosed in a proportion of psoriasis patients. In the global MAPP findings for psoriasis patients without a diagnosis of PsA (n = 2714), 44% reported pain or soreness in any joint, 16% reported symptoms resembling enthesitis, and 26% reported symptoms resembling dactylitis [22]. Although a number of conditions may be associated with joint pain, previous studies have shown that a high proportion of patients have undiagnosed PsA [2, 14, 28]. This could partially be related to the differential diagnosis of PsA; in the physician portion of the MAPP survey, dermatologists cited the greatest challenge in managing patients with PsA was differentiating PsA from other arthritic diseases, while rheumatologists felt the greatest challenge in managing patients with PsA was the delay in referral to their practice.
Patients with PsA experience a substantial burden of disease [3, 4, 24, 29, 32, 33]. More than half of the patients with PsA participating in the MAPP survey reported their disease as severe, compared with 27% of patients with psoriasis. A large proportion of patients with PsA reported pain or soreness in more than four joints. Although patients may have overestimated or underestimated the number of involved joints, the HAQ-8 results did confirm an increased impairment in physical function in these patients compared with the overall population of patients with PsA. The physical impact of PsA was greater in patients with symptoms of enthesitis and/or dactylitis. In addition, physical function in patients with PsA was affected by the extent of psoriasis, as measured by palm count, with more severe disability reported in patients with greater palm counts. PsA also impacted productivity and functionality, with 20–30% of patients reporting that PsA affected their ability to work full time, choose a career, and get or keep a job. These findings are consistent with the published literature [4, 24, 29, 32, 33] and highlight the substantially greater impact of joint disease on patients’ physical function and the added burden of PsA on psoriasis patients.
Symptoms resembling enthesitis (i.e., pain/swelling of heel) were reported by 31% of patients, and symptoms resembling dactylitis (i.e., “sausage” digits) were reported by 45% of patients. Enthesitis and dactylitis, important distinguishing features of PsA in many patients, are associated with more severe disease; both can be difficult to treat and can lead to disability [5, 13, 31]. Approximately twice as many patients with PsA (21%) had nail involvement compared with patients with psoriasis alone (11%), and 44% of patients with PsA reported scalp involvement; however, overall these rates are lower than those previously reported [8, 17, 18, 21, 26]. The presence of skin disease in these areas has been found to have a disproportionate effect on QoL and is often difficult to treat, with limited effective treatment options [7, 9, 19, 21]. Lastly, co-morbidities, including unspecified arthritis, hypertension, depression, diabetes, and heart disease, were commonly reported by patients with PsA, as was obesity. This finding is consistent with prior reports of a higher prevalence of cardiovascular disease and associated risk factors, such as diabetes and other chronic diseases, in patients with PsA compared with psoriasis patients or matched controls in the community [4, 6, 11, 15]. The multiple co-morbidities, often present in a single patient, complicate the selection of therapeutic agents due to safety concerns and create challenges in the assessment of the functional impact of PsA.
Patients diagnosed with PsA were more likely to be treated compared with patients with psoriasis only [22]; however, many patients with PsA remain untreated or undertreated. Among patients with PsA, 42% reported currently receiving systemic treatment [conventional oral (19%) or biologic (22%)]; this rate of systemic treatment is far lower than the 97% of patients with PsA reported to have been prescribed a systemic treatment [conventional oral (63%) or biologic (33%)] by rheumatologists who participated in the MAPP survey [34]. The majority of dermatologists (65%) also reported prescribing systemic treatment. Interestingly, the most common reason patients with PsA did not seek treatment by an HCP in the previous 12 months was that they did not think it would help, which suggests that education regarding the availability of effective treatments is needed. Approximately 30% of patients participating in the MAPP survey had not seen a rheumatologist for their PsA; just over one-third indicated that their rheumatologist was the HCP they saw most often for their PsA, while about one-quarter saw their primary care provider or their dermatologist. Some of these differences may represent variations in health-care delivery and access to specialists across the countries surveyed. However, these results underscore the need for timely diagnosis in patients with PsA and ensure their appropriate treatment and/or referral to a specialist. Treatment differences between patients and physicians may also reflect disparities between written prescriptions and prescriptions that are filled and taken by patients. Thus, it is important for physicians to discuss patient concerns about medications, improve patient adherence, and, in turn, improve clinical responses.
As with any survey, the MAPP survey is limited by the ability of patients to recall information, the nature of the questions, and the interpretation of the questions. The sample included a higher proportion of females than in the general population, suggesting potential recruitment bias or over-reporting of this population. Because the survey relies on patient self-reporting, it is possible that patients had a misperception of PsA and the symptoms they experienced, such as nail involvement or the more complicated enthesitis and dactylitis. In addition, because this survey was blinded, there was no way to re-contact patients for clarification of answers. The survey did not include a control group, and information on treatments was limited to drug therapy classes. Few questions were asked about specific treatment options; thus, no meaningful comparison among treatments can be made. The survey also did not account for differences in country, ethnicity, age, and direct and indirect costs of health care. Further limitations of this study include the uncertainty about the specific diagnoses and the treatments received. Patients may have misunderstood the diagnosis given by their HCP, or their HCP may have made an incorrect diagnosis. The effect of this uncertainty on the results is unknown, but it does mean that the results should be interpreted with caution. On the whole, the clinical data obtained are consistent with what is known about PsA; however, the high proportion of females in the PsA group suggests that this sample may not be representative of the population as a whole.
Conclusions
The MAPP survey results underscore the substantial disease burden associated with PsA and the significant impact on physical function. Survey results suggest undertreatment of PsA as well as a high rate of treatment burden.
References
American Association for Public Opinion Research website. Best practices. Available at: http://www.aapor.org/Standards-Ethics/Best-Practices.aspx. Accessed 8 Feb 2016.
Armstrong AW, Robertson AD, Wu J, Schupp C, Lebwohl MG. Undertreatment, treatment trends, and treatment dissatisfaction among patients with psoriasis and psoriatic arthritis in the United States: findings from the National Psoriasis Foundation surveys, 2003–2011. JAMA Dermatol. 2013;149:1180–5.
Armstrong AW, Schupp C, Wu J, Bebo B. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003–2011. PLoS ONE. 2012;7:e52935.
Boehncke WH, Menter A. Burden of disease: psoriasis and psoriatic arthritis. Am J Clin Dermatol. 2013;14:377–88.
Carneiro S, Bortoluzzo A, Goncalves C, et al. Effect of enthesitis on 1505 Brazilian patients with spondyloarthritis. J Rheumatol. 2013;40:1725.
Costa L, Caso F, D’Elia L, et al. Psoriatic arthritis is associated with increased arterial stiffness in the absence of known cardiovascular risk factors: a case control study. Clin Rheumatol. 2012;31:711–5.
Crowley J. Scalp psoriasis: an overview of the disease and available therapies. J Drugs Dermatol. 2010;9:912–8.
Crowley JJ, Weinberg JM, Wu JJ, Robertson AD, Van Voorhees AS. Treatment of nail psoriasis: best practice recommendations from the medical board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151:87–94.
de Vries AC, Bogaards NA, Hooft L, et al. Interventions for nail psoriasis. Cochrane Database Syst Rev. 2013;1:CD007633.
Ficco HM, Citera G, Cocco JA. Prevalence of psoriatic arthritis in psoriasis patients according to newer classification criteria. Clin Rheumatol. 2014;33:1489–93.
Gladman DD, Ang M, Su L, Tom BD, Schentag CT, Farewell VT. Cardiovascular morbidity in psoriatic arthritis. Ann Rheum Dis. 2009;68:1131–5.
Gladman DD, Antoni C, Mease P, Clegg DO, Nash P. Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis. 2005;64(Suppl 2):ii14–ii17.
Gladman DD, Ziouzina O, Thavaneswaran A, Chandran V. Dactylitis in psoriatic arthritis: prevalence and response to therapy in the biologic era. J Rheumatol. 2013;40:1357–9.
Helliwell P, Coates L, Chandran V, et al. Qualifying unmet needs and improving standards of care in psoriatic arthritis. Arthritis Care Res. 2014;66:1759–66.
Husted JA, Thavaneswaran A, Chandran V, et al. Cardiovascular and other comorbidities in patients with psoriatic arthritis: a comparison with patients with psoriasis. Arthritis Care Res (Hoboken). 2011;63:1729–35.
Ibrahim G, Waxman R, Helliwell PS. The prevalence of psoriatic arthritis in people with psoriasis. Arthritis Rheum. 2009;61:1373–8.
Jiaravuthisan MM, Sasseville D, Vender RB, Murphy F, Muhn CY. Psoriasis of the nail: anatomy, pathology, clinical presentation, and a review of the literature on therapy. J Am Acad Dermatol. 2007;57:1–27.
Kragballe K. Management of difficult to treat locations of psoriasis. Scalp, face, flexures, palm/soles and nails. Curr Probl Dermatol. 2009;38:160–71.
Kragballe K, Menter A, Lebwohl M, Tebbey PW, van de Kerkhof PC. Long-term management of scalp psoriasis: perspectives from the International Psoriasis Council. J Dermatolog Treat. 2013;24:188–92.
Langenbruch A, Radtke MA, Krensel M, Jacobi A, Reich K, Augustin M. Nail involvement as a predictor of concomitant psoriatic arthritis in patients with psoriasis. Br J Dermatol. 2014;171:1123–8.
Langley RG, Dauden E. Treatment and management of psoriasis with nail involvement: a focus on biologic therapy. Dermatology. 2010;221(Suppl 1):29–42.
Lebwohl MG, Bachelez H, Barker J, et al. Patient perspectives in the management of psoriasis: results from the population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis Survey. J Am Acad Dermatol. 2014;70:871–81.
Lebwohl MG, Kavanaugh A, Armstrong AW, Van Voorhees AS, Kalb RE. Patient perspectives on psoriasis management: US results of the population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis survey. Psoriasis. Forum. 2014;20:124–31.
McHugh NJ, Balachrishnan C, Jones SM. Progression of peripheral joint disease in psoriatic arthritis: a 5-yr prospective study. Rheumatology (Oxford). 2003;42:778–83.
Mease PJ, Gladman DD, Papp KA, et al. Prevalence of rheumatologist-diagnosed psoriatic arthritis in patients with psoriasis in European/North American dermatology clinics. J Am Acad Dermatol. 2013;69:729–35.
Ortonne J, Chimenti S, Luger T, Puig L, Reid F, Trueb RM. Scalp psoriasis: European consensus on grading and treatment algorithm. J Eur Acad Dermatol Venereol. 2009;23:1435–44.
Pincus T, Summey JA, Soraci SA Jr, Wallston KA, Hummon NP. Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis Rheum. 1983;26:1346–53.
Reich K, Kruger K, Mossner R, Augustin M. Epidemiology and clinical pattern of psoriatic arthritis in Germany: a prospective interdisciplinary epidemiological study of 1511 patients with plaque-type psoriasis. Br J Dermatol. 2009;160:1040–7.
Rosen CF, Mussani F, Chandran V, Eder L, Thavaneswaran A, Gladman DD. Patients with psoriatic arthritis have worse quality of life than those with psoriasis alone. Rheumatology (Oxford). 2012;51:571–6.
Rouzaud M, Sevrain M, Villani AP, et al. Is there a psoriasis skin phenotype associated with psoriatic arthritis? Systematic literature review. J Eur Acad Dermatol Venereol. 2014;28(Suppl 5):17–26.
Sakkas LI, Alexiou I, Simopoulou T, Vlychou M. Enthesitis in psoriatic arthritis. Semin Arthritis Rheum. 2013;43:325–34.
Singh JA, Strand V. Spondyloarthritis is associated with poor function and physical health-related quality of life. J Rheumatol. 2009;36:1012–20.
Sokoll KB, Helliwell PS. Comparison of disability and quality of life in rheumatoid and psoriatic arthritis. J Rheumatol. 2001;28:1842–6.
van de Kerkhof PCM, Reich K, Kavanaugh A, et al. Physician perspectives in the managment of psoriasis and psoriatic arthritis: results from the population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis survey. J Eur Acad Dermatol Venereol. 2015;29:2002–10.
Wilson FC, Icen M, Crowson CS, McEvoy MT, Gabriel SE, Kremers HM. Incidence and clinical predictors of psoriatic arthritis in patients with psoriasis: a population-based study. Arthritis Rheum. 2009;61:233–9.
Wu Y, Mills D, Bala M. Impact of psoriasis on patients’ work and productivity: a retrospective, matched case-control analysis. Am J Clin Dermatol. 2009;10:407–10.
Acknowledgments
The MAPP survey was sponsored by Celgene Corporation, Summit, NJ, USA. The article processing charges for this publication were funded by Celgene Corporation. The authors received editorial support in the preparation of the manuscript from Jennifer Schwinn, RPh, Amy Zannikos, PharmD, and Peloton Advantage, LLC, funded by Celgene Corporation. The authors, however, directed and are fully responsible for all content and editorial decisions for this manuscript. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Disclosures
Arthur Kavanaugh has served as an investigator for AbbVie, Amgen, Bristol-Myers Squibb, Celgene Corporation, Janssen, and UCB. Philip Helliwell has received honoraria from Celgene Corporation. Christopher T. Ritchlin has been an investigator and/or consultant for AbbVie, Amgen, Eli Lilly, Janssen, Pfizer, and UCB.
Compliance with Ethics Guidelines
The MAPP survey followed the American Association for Public Opinion Research guidelines for developing and conducting surveys. Institutional review board approval was granted for data collection in the USA (Abt SRBI 5454), and the other countries abided by standards and ethical practices according to their codes or guidelines.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kavanaugh, A., Helliwell, P. & Ritchlin, C.T. Psoriatic Arthritis and Burden of Disease: Patient Perspectives from the Population-Based Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) Survey. Rheumatol Ther 3, 91–102 (2016). https://doi.org/10.1007/s40744-016-0029-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40744-016-0029-z