Abstract
Introduction
To compare perceptions of disease control and treatment satisfaction between patients with psoriatic arthritis (PsA) in North America and Europe, and between participating countries within each region.
Methods
Data were collected from patients with self-reported PsA diagnoses using an online survey. Results from questions on perceptions of overall health, disease severity, PsA symptoms, PsA impacts, and treatment satisfaction/preferences were reported using descriptive statistics and Chi-square tests.
Results
A total of 456 patients from North America (Canada, n = 155; US, n = 301) and 417 patients from Europe (France, n = 123; Spain, n = 135; UK, n = 159) were included in this analysis. Patients in North America were more likely to rate their overall health as excellent/good compared with those in Europe (49 vs. 14%), but also rate their disease as severe (27 vs. 15%). Despite treatment, patients in North America and Europe still experienced musculoskeletal (92 vs. 91%) and skin/nail (62 vs. 58%) symptoms. Similar proportions of patients in North America vs. Europe experienced a social impact (81 vs. 85%); more patients in Europe vs. North America experienced PsA-related work impacts (83 vs. 74%). Satisfaction with PsA medication was more common in North America (89%) vs. Europe (79%), and more common in Spain (91%) vs. the UK (82%) or France (66%). Across all regions and countries, ≥ 75% of patients agreed that symptoms were controlled. However, ≥ 66% wished they had more medication choices, and ≥ 84% wanted to change something about their medication.
Conclusions
Although perception of overall health and disease severity varied, many patients from both regions still experienced symptoms despite receiving medications for PsA, wished they had greater choice of medications, and/or would like to change an aspect of their medications. While these survey findings are subject to selection bias, they do indicate there is scope to improve the treatment of PsA.
Plain Language Summary
Psoriatic arthritis (PsA) is a disease that can cause joint pain and stiffness, and is often associated with a skin rash called psoriasis. These symptoms can affect quality of life, and patients and doctors should work together when choosing treatment. There has not been a lot of information on what patients think about their disease and their medicines. We found that patients from different regions and countries had different opinions, and that treatment of PsA can be improved. For example, patients in North America were more likely to say that their overall health was excellent or good, compared with patients in Europe. However, more patients in North America than in Europe described their PsA disease as severe. Similar numbers of patients in both regions experienced impacts on their social life due to their PsA, but patients in Europe were more likely to report that PsA affected their work life compared with patients in North America. More patients in North America than in Europe were satisfied with their medicines, but patients across all regions and countries still had symptoms even when they took medicines. Many patients also wished they had more options and wanted to change something about their medicines. These findings were based on an online survey. Patients from North America (Canada and the US) and Europe (France, Spain, and the UK) answered questions about their PsA disease and medicines. We only compared answers between patients from North America and Europe, and between countries within each region.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
There is limited information about patient perceptions of how psoriatic arthritis (PsA) impacts their health and life, or about patient treatment preferences. |
We used survey data to compare the perception of disease management and treatment preferences between patients with PsA in North America and Europe, and between participating countries within each region. |
What was learned from the study? |
Our survey highlighted that perceptions of overall health, disease severity, symptoms related to PsA, impact of PsA, as well as treatment satisfaction and preference, varied between patients in North America and Europe, and between different countries within each region. |
Overall, despite receiving medications for PsA, many patients still experienced symptoms, wished they had a greater choice of medication, and/or would change something about their medication; this suggests there is scope to improve the treatment of patients with PsA. |
Introduction
Psoriatic arthritis (PsA) is a complex and heterogeneous inflammatory disease, characterized by psoriasis, peripheral joint disease, and inflammation of the axial skeleton, enthesitis, dactylitis, and nail lesions [1]. Both musculoskeletal and skin/nail symptoms contribute to the disease burden of PsA and can have significant negative impacts on patients’ health-related quality of life [2]. Indeed, PsA has been reported to affect both patients’ physical and emotional health, as well as limiting their functional capacity, work abilities, and social participation [2].
Shared decision-making between patients and healthcare professionals (HCPs) is pivotal for the management of PsA [3]; however, there is limited information on the patient’s perspective. In the few studies that have explored the patient’s perspective of the impact and severity of PsA, it has been suggested that discrepancies between the perspectives of patients and HCPs have been observed, with patients generally perceiving their disease as more severe than HCPs [4,5,6]. Furthermore, patient–HCP misalignment in satisfaction with PsA disease control has been associated with increased disease activity and disability [7]. Overall, it is important to consider patients’ perceptions and priorities relating to the management of PsA to ensure that their expectations are met [8].
To meet this need, a global online survey was conducted to evaluate the impact of PsA on daily life from the patient’s perspective and to evaluate if there are any differences related to geographical locations [9]. The overall findings from this survey suggested that patient perceptions of disease impact may vary between countries [9]; however, treatment satisfaction and the preferences of patients, which are important to consider when deciding on a treatment strategy, were not reported. In this analysis, we used survey data to compare the perceptions of disease management, including overall health, disease severity, symptoms, and disease impacts, as well as treatment satisfaction and preferences between patients with PsA in North America and Europe, and between countries within each region.
Methods
Survey Design
The survey methodology has been described previously [9]. In brief, an online survey was conducted by The Harris Poll on behalf of Pfizer Inc between November 2, 2017, and March 12, 2018, among 1286 patients with PsA in Australia, Brazil, Canada, France, Spain, Taiwan, the UK, and the US. Eligible patients were aged ≥ 18 years, with a self-reported diagnosis of PsA of > 1 year, who had visited a rheumatologist or dermatologist in the past 12 months, and had reported using ≥ 1 conventional synthetic/biologic disease-modifying antirheumatic drug (DMARD) for PsA. All eligible participants were required to provide consent before continuing to the core survey content.
The survey included questions related to psoriasis experience, quality of life, and general feelings towards PsA; experiences pre-diagnosis and at initial diagnosis; treatment attitudes and experiences; and feelings towards changes in medication regimens. For each country, a custom set of demographic questions was followed by a dynamic number of core questions (approximately 35–60 questions in total), which varied depending on patient responses. No qualitative questions were included in the survey.
Analysis of Survey Data
The original survey was designed to support regional and cross-country comparisons. This analysis focused on data reported by patients relating to demographics and disease characteristics, perceptions of current overall health, disease severity, symptoms of PsA, and impacts of PsA on social and work life, as well as treatment satisfaction and preferences. Patient responses were compared between North America (US and Canada) and Europe (France, Spain, and the UK), between Canada and the US, and between each of the three European countries.
Descriptive statistics and binomial (Chi-square) tests for two proportions are reported [10, 11]. Raw data were not weighted at the individual country level, and are therefore only representative of the individuals who completed the survey; in the previously reported analysis of the full global data set, a post-weight was applied to adjust for the relative size of each country’s adult population within the total adult population across all countries surveyed [9]. All percentages reported and statistical comparisons were calculated and analyzed based on the weighted global data. Statistical significance was defined as p < 0.05, and all differences described within the text were statistically significant unless stated otherwise.
It should be noted that some country-specific survey data have previously been reported as part of the overall findings from the full global data set [9].
Compliance with Ethics Guidelines
Patients were recruited from online market research panels made up of members who agreed to participate in this type of research. Qualified respondents provided informed consent to complete the research. The surveys were noninterventional and were not conducted as a clinical study. All respondents agreed to participate but ethics approval was not required.
Results
Patients
This analysis included survey data from 456 patients in North America (Canada, n = 155; US, n = 301) and 417 patients in Europe (France, n = 123; Spain, n = 135; UK, n = 159). Patient demographics and disease characteristics by region and country are shown in Table 1 and Supplementary Table S1, respectively.
Overall, there were differences in demographics and characteristics between patients in North America and Europe, as well as between European countries (Table 1). Compared with patients in Europe, a greater proportion of patients in North America was female (46% Europe vs. 60% North America), and a smaller proportion of patients in North America stated they were employed (78% Europe vs. 70% North America; Table 1). On average, patients in North America were older than those in Europe (mean age, 45.5 vs. 40.0 years), and older when diagnosed with PsA (mean age, 35.0 vs. 31.6 years; Table 1). In both North America and Europe, patients most commonly saw rheumatologists (78 and 68%, respectively) and primary care physicians/general practitioners/internists (44 and 56%, respectively) to help manage their PsA (Table 1). In the past 12 months, similar proportions of patients in North America (90%) and Europe (91%) had seen a rheumatologist for their PsA (Table 1). However, a greater proportion of patients in Europe (71%) than in North America (64%) had seen a dermatologist for their PsA (Table 1); this trend was primarily driven by the responses from patients in Spain (Supplementary Table S1). Compared with patients in Europe, a greater proportion of patients in North America considered their rheumatologist to be their primary physician (72% Europe vs. 83% North America), while fewer patients in North America considered their dermatologist to be their primary physician (28% Europe vs. 17% North America; Table 1).
Several characteristics of patients in each individual country have been described previously [9]. Within North America, a similar proportion of patients in the US and Canada was female (61 vs. 52%) and those in the US were more likely to be in employment (71 vs. 60%; Supplementary Table S1). Patients in Canada were on average older than those in the US (mean age, 49.1 vs. 45.1 years; Supplementary Table S1). In Canada and the US, most patients were seeing a rheumatologist (66 and 79%, respectively) and primary care physicians/general practitioners/internists for their PsA (51 and 44%, respectively; Supplementary Table S1). A greater proportion of patients in the US vs. Canada considered a rheumatologist to be their primary physician (85 vs. 70%); 15 vs. 30% of patients in the US and Canada, respectively, considered their primary physician to be a dermatologist (Supplementary Table S1). In the past 12 months, more patients in the US (91%) had seen a rheumatologist for their PsA, compared with patients in Canada (83%; Supplementary Table S1).
The proportion of patients in employment was greater in Spain (89%) than in France (75%) or the UK (74%; Supplementary Table S1). On average, patients in Spain were younger (mean age, 36.9 years) vs. those in France (40.9 years) or the UK (41.5 years; Supplementary Table S1). In addition, patients in Spain were also diagnosed with PsA at a younger age (mean age, 28.1 years), compared with those in France (32.5 years) or the UK (33.4 years; Supplementary Table S1). Across the three countries, the HCPs most commonly seen by patients to manage their PsA were rheumatologists (61–79%) and primary care physicians/general practitioners/internists (48–63%; Supplementary Table S1). Similar proportions of patients across the three countries considered their primary physician to be a rheumatologist (70–75%) or a dermatologist (25–30%; Supplementary Table S1). In the past 12 months, similar proportions of patients across the three countries had seen a rheumatologist for their PsA (90–92%); however, more patients in Spain (82%) had seen a dermatologist, compared with patients in France or the UK (both 67%; Supplementary Table S1).
Various types of PsA medications had previously been used across the populations from both regions and all countries, but, at the time of the survey, most patients were taking biologic DMARDs and/or oral DMARDs (Table 1 and Supplementary Table S1).
Patients’ Perceptions of their Overall Health and Disease Severity
Compared with patients in Europe, a greater proportion of patients in North America rated their overall health as excellent/good (14% Europe vs. 49% North America; Fig. 1a); however, more patients in North America (27%) rated their PsA disease as severe on the day of the survey than those in Europe (15%; Fig. 1b). A similar proportion of patients reported their overall health as excellent/good in Canada (50%) and the US (49%; Supplementary Fig. S1a); although over twice as many patients in the US (28%) rated their disease as severe on the day of the survey, compared with patients in Canada (12%; Supplementary Fig. S1b). Within the three European countries, although Spain had the smallest proportion of patients reporting their overall health as excellent/good (7%) compared with patients in France (15%) or the UK (19%; Supplementary Fig. S1a), more patients in France (22%) than in Spain (7%) rated their disease as severe on the day of the survey (Supplementary Fig. S1b).
Patients’ perceptions of their a overall health and b disease severity by region. a The percentage of patients reporting their overall health today as excellent/good based on responses to the question: “How would you describe your current overall health today? (excellent/good/fair/poor)”. b The percentage of patients reporting their PsA severity based on responses to the question: “How bad is your psoriatic arthritis today? (mild/moderate/severe)”. All percentages were calculated based on the weighted population of each region. *Significant difference (p < 0.05) with North America. †Significant difference (p < 0.05) with Europe. PsA psoriatic arthritis
Patient-Reported Symptoms Related to PsA
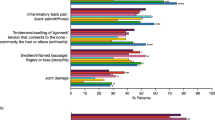
Despite treatment, nearly all patients currently taking prescription medications for PsA still experienced musculoskeletal symptoms (North America: 92%; Europe: 91%), and most still experienced skin/nail symptoms (North America: 62%; Europe: 58%). More patients in North America, compared with patients in Europe, still experienced joint pain (59 vs. 51%, respectively), stiffness (56 vs. 37%), joint tenderness (47 vs. 34%), joint swelling (43 vs. 31%), or enthesitis (31 vs. 24%; Fig. 2a). A greater proportion of patients in North America reported skin patches or plaques, compared with patients in Europe (43 vs. 33%; Fig. 2a). Among patients who had experienced any symptoms in the past 12 months, more patients in North America than in Europe reported joint pain as the most bothersome symptom (37 vs. 26%), while a smaller proportion of patients reported joint swelling (4 vs. 10%) or skin discomfort (7 vs. 11%) as the most bothersome symptom (Fig. 2b).
Patient-reported a symptoms related to PsA despite treatment and b most bothersome symptoms related to PsA, experienced in the past 12 months by region. a The percentage of patients reporting symptoms related to PsA among those currently taking prescription PsA medications based on responses to the question: “Which of the following symptoms do you still experience despite psoriatic arthritis treatment? Please select all that apply.” b The percentage of patients reporting symptoms related to PsA among those who had experienced any symptoms in the past 12 months based on responses to the question: “You mentioned that you have experienced symptoms related to psoriatic arthritis in the past 12 months. Of these symptoms, which is the most bothersome?” All percentages were calculated based on the weighted population of each region. *Significant difference (p < 0.05) with North America. †Significant difference (p < 0.05) with Europe. PsA psoriatic arthritis
Similar proportions of patients in Canada and the US reported that they still experienced different musculoskeletal symptoms despite receiving treatment (Supplementary Fig. S2a). In contrast, more patients in Canada vs. the US reported nail changes (38 vs. 27%; Supplementary Fig. S2a). In Canada, joint pain (38%) and unusual fatigue (12%) were most commonly reported as the most bothersome symptoms experienced in the past 12 months. Patients in the US were more likely to report joint pain (37%) and inflammatory back pain (12%) as the most bothersome symptoms (Supplementary Fig. S2b).
Within Europe, more patients in the UK, compared with Spain, reported stiffness (47 vs. 26%, respectively), joint tenderness (40 vs. 22%), enthesitis (30 vs. 18%), or joint damage (23 vs. 13%); joint swelling was more frequently reported in the UK (42%) than in Spain (31%) or France (19%; Supplementary Fig. S2a). In addition, the proportion of patients reporting joint tenderness was greater in France (37%) than in Spain (22%), but the proportion of patients reporting joint swelling was lower in France (19%) than in Spain (31%; Supplementary Fig. S2a). A greater proportion of patients in the UK reported symptoms of skin patches or plaques, compared with Spain (41 vs. 25%, respectively). Among patients who had experienced any symptoms in the past 12 months, joint tenderness was reported as the most bothersome symptom more commonly among patients in the UK (5%) than in Spain (0%; Supplementary Fig. S2b).
Patients’ Perceptions of the Impact of PsA
Similar proportions of patients in North America and Europe reported that PsA impacted their social life overall (81 vs. 85%, respectively) and considering specific aspects of their social life (Fig. 3a). In contrast, more patients in Europe (83%) vs. North America (74%) reported that PsA affected their work life. While more patients in North America had taken a sick day from work compared with patients in Europe (50 vs. 42%, respectively), a smaller proportion had taken medical leave from work (22 vs. 44%; Fig. 3b).
Patients’ perceptions of the impact of PsA on their a social life and b work life by region. The percentage of patients reporting a social or work life impact was based on responses to the question: “Have you done any of the following as a result of psoriatic arthritis? Please select all that apply.” All percentages were calculated based on the weighted population of each region. *Significant difference (p < 0.05) with North America. †Significant difference (p < 0.05) with Europe. PsA psoriatic arthritis
Similar proportions of patients in Canada and the US reported that PsA impacted their social life overall (82 vs. 75%, respectively). More patients in the US vs. Canada also stated they had experienced emotional distress (60 vs. 46%) and stopped doing or participating in social activities (51 vs. 34%; Supplementary Fig. S3a). Similarly, more patients in the US vs. Canada reported that PsA impacted their overall work life (75 vs. 70%), although affected aspects of work life varied between countries (Supplementary Fig. S3b). For example, a higher proportion of patients in the US than in Canada had taken a sick day from work (51 vs. 40%); however, a smaller proportion of patients in the US than in Canada had taken medical leave from work (21 vs. 29%; Supplementary Fig. S3b).
Within Europe, more patients in Spain (92%) vs. France or the UK (both 82%) reported that PsA impacted their social life overall. The proportion of patients who reported experiencing social shame or disapproval was greater in Spain (50%) vs. France (35%) or the UK (23%), and greater in France (35%) vs. the UK (23%; Supplementary Fig. S3a). Similar proportions of patients in France, Spain, and the UK reported that PsA impacted their work life (France: 80%; Spain: 87%; UK: 85%); however, there were variations in different aspects of their work life. Patients in France were less likely to take a sick day from work (30%) compared with patients in Spain (48%) or the UK (49%; Supplementary Fig. S3b). The proportion of patients in the UK (36%) whose productivity had decreased was smaller than that in Spain (50%), but the proportion who had quit or been let go from a job was greater in the UK (21%) vs. Spain (10%; Supplementary Fig. S3b). The proportion of patients who had gone on permanent disability was also greater in the UK (23%) than in France (14%) or Spain (10%; Supplementary Fig. S3b).
Treatment Satisfaction and Preferences
Most patients in North America and Europe were satisfied with their current medication for PsA (Fig. 4a). A greater proportion of patients in North America (89%) were “very” or “somewhat” satisfied with their current medication compared with those in Europe (79%; Fig. 4a). Within Europe, the proportion of patients who were very/somewhat satisfied with their medication was greater in Spain (91%) vs. the UK (82%) or France (66%), and greater in the UK (82%) vs. France (66%; Fig. 4a).
a Patient satisfaction with their current PsA medication and b statements patients strongly or somewhat agreed with pertaining to their current PsA medication, by region and country. a The percentage of patients who were very/somewhat satisfied with their PsA medication based on responses to the question: “You indicated that you are currently taking prescription medication(s) for psoriatic arthritis. Overall, how satisfied are you with your current psoriatic arthritis medication regimen (very satisfied/somewhat satisfied/somewhat dissatisfied/very dissatisfied)?” b The percentage of patients who strongly/somewhat agreed with the statements pertaining to their PsA medication based on responses to the question: “How much do you agree or disagree with each of the following statements about your prescription psoriatic arthritis medication(s)? (strongly agree/somewhat agree/somewhat disagree/strongly disagree).” All percentages were calculated based on the weighted population of each region or country. North America was compared only with Europe, Canada was compared only with the US, and countries within Europe were compared only with each other. *Significant difference (p < 0.05) with North America. †Significant difference (p < 0.05) with Europe. ‡Significant difference (p < 0.05) with Canada. ‖Significant difference (p < 0.05) with France. #Significant difference (p < 0.05) with the UK. PsA psoriatic arthritis
Across all regions and countries, at least 85% of patients “strongly” or “somewhat” agreed that they took their medication as prescribed (Fig. 4b). The proportion of patients who agreed that they always took their medication as prescribed was greater in North America (95%) than in Europe (88%; Fig. 4b). At least 75% of patients in all regions and countries agreed that their medication controlled their symptoms (Fig. 4b). Despite this, at least 71% of patients in all regions and countries worried that their medication would not work well enough or would stop working (Fig. 4b). More patients in Spain worried that their medication would not work well enough or stop working, compared with patients in the UK (87 vs. 75%, respectively), and were worried that they would experience serious side effects, compared with patients in France and the UK (Spain: 87%; France: 75%; UK: 69%; Fig. 4b). Furthermore, at least 66% of patients across all regions and countries wished they had more medication choices. The proportion of patients who wished they had more medication choice were greater in the US (75%) vs. Canada (66%), and greater in Spain (90%) vs. France (66%) or the UK (69%; Fig. 4b). More patients in Europe vs. North America indicated that they would prefer injection/infusion over oral medication (56 vs. 45%; Fig. 4b). Within Europe, a greater proportion of patients in Spain, compared with France or the UK, would prefer oral medicine over injection/infusion (Spain: 82%; France: 68%; UK: 69%; Fig. 4b).
Despite a majority of patients being satisfied with their medication overall, most also wanted to change an aspect of their medication (Fig. 5). The overall proportions of patients who wanted to change something about their medication was similar between North America and Europe (90 vs. 93%, respectively; Fig. 5). Within North America, a greater proportion of patients in the US (91%) than in Canada (84%) wanted to change something about their medication (Fig. 5). Within Europe, more patients in France (95%) and Spain (99%), compared with the UK (87%), wanted to change something about their medication (Fig. 5).
Aspects of current PsA medication(s) that patients would like to change by region and country. The percentage of patients who reported that they would like to change something about their medication among those currently taking prescription PsA medications was based on responses to the question: “Ideally, what would you most like to change, if anything, about your current psoriatic arthritis prescription medication(s)? Please select all that apply.” Only the eight most reported responses were included. All percentages were calculated based on the weighted population of each region or country. North America was compared only with Europe, Canada was compared only with the US, and countries within Europe were compared only with each other. *Significant difference (p < 0.05) with North America. †Significant difference (p < 0.05) with Europe. ‡Significant difference (p < 0.05) with Canada. ‖Significant difference (p < 0.05) with France. #Significant difference (p < 0.05) with the UK. PsA psoriatic arthritis
More patients in North America vs. Europe wanted to change the cost or coverage available from their healthcare system (30 vs. 20%, respectively); however, a smaller proportion of patients in North America than in Europe wanted to change how often they needed to take their medication (25 vs. 32%; Fig. 5). Within Europe, more patients in France vs. the UK wished to change the number or severity of side effects (48 vs. 34%), while more patients in Spain wanted to change how well their medication relieved musculoskeletal symptoms vs. France (46 vs. 32%), and how often they need to take their medications vs. the UK (40 vs. 27%; Fig. 5).
Discussion
In this analysis of data from a global survey, variations between regions (North America vs. Europe) and countries (Canada vs. the US; France vs. Spain vs. the UK) were seen in patients’ perceptions of overall health, disease severity, symptoms related to PsA, and impact of PsA, as well as treatment satisfaction and preferences. Comparisons of responses from patients in North America and Europe demonstrated that the impact of PsA perceived by patients does not necessarily reflect their perception of their overall health or treatment satisfaction. For example, more patients in North America vs. Europe rated their overall health as excellent or good even though patients in North America were more likely to rate their PsA disease as severe on the day of the survey and to report experiencing joint pain, stiffness, joint tenderness, joint swelling, and skin patches or plaques despite treatment. Furthermore, the proportions of patients reporting an impact of PsA on their social life were similar between North America and Europe.
Patient-reported symptoms related to PsA, as well as the social and work impact of PsA on patients in individual countries, have been previously reported [9]; however, this is the first time that data on treatment satisfaction and preferences have been reported. Survey responses from patients in Canada and the US were generally similar, but some significant differences were noted. Patients in the US were more likely to rate their disease as severe. The proportions of patients experiencing different musculoskeletal or skin symptoms despite treatment were similar in Canada and the US, although more patients in Canada experienced nail changes. Compared with Canada, the proportion of patients who had taken a sick day from work was greater in the US; however, more patients in Canada than those in the US had taken medical leave from work. Within Europe, there were some significant differences in survey responses between France, Spain, and the UK, but few clear over-arching patterns. The proportions of patients experiencing different musculoskeletal or skin/nail symptoms despite treatment were typically smallest in Spain and greatest in the UK. Spain was the country with the greatest proportion of patients who were very/somewhat satisfied with their medication, but also had the smallest proportion of patients who rated their overall health as excellent/good. Notably, patients in Spain were more likely to have experienced social shame or disapproval and decreased work productivity, while the UK had a greater proportion of patients who had gone on permanent disability than Spain and France, and a greater proportion of patients who had quit or been let go from a job than Spain.
Across all regions and countries, patients were generally satisfied with their PsA medication and agreed that it controlled their disease. However, most patients still experienced symptoms, and most wished they had a greater choice of medications or could change some aspect of their medication (e.g., the number or severity of the side effects or how well it relieved musculoskeletal symptoms). This suggests that there is scope to improve the treatment of PsA.
The results presented here are aligned with the overall findings from the global survey, which reported differences in patient perspectives between eight individual countries [9]. It has been speculated that the observed differences in the perception of disease impact may have been influenced by phenotypical variations in disease severity and different cultural norms and expectations [9]. In addition, access to medications may be determined by the different types of healthcare system in each country, which may contribute to the variations in patient satisfaction observed in this analysis. A greater proportion of patients in North America vs. Europe wanted to change the cost or coverage available from their healthcare system; these differences were not observed between patients in Canada vs. the US, though more patients in the US than Canada wished they had more medication choices. Given that the US is the only country of those surveyed where universal health coverage is not available, these findings were not unexpected. However, even among countries with universal health coverage, access to medications may still vary. A previous study suggested that access to biologic DMARDs, as determined by availability (the number of reimbursed biologic DMARDs), affordability (average annual price of all biologic DMARDs), and acceptability, was better in France than in Spain, the UK, or the US [12]. Accordingly, our findings suggested that the proportion of patients with PsA who wished they had more medication choices was lowest in France (66%); however, we also found that patients in France were the least likely to be satisfied with their PsA medication. Overall, discrepancy in patient perspectives between regions and countries may be due to aspects beyond the disease itself, and there is likely a complex interplay of factors involved.
Consistent with this analysis, differences in patient perspectives about disease severity and treatment preferences have also been reported in other studies. Based on the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey, which included 3426 patients with psoriasis and/or PsA, more patients with PsA in North America compared with European countries rated their disease as severe (60 vs. 40%, respectively) [13]. In the present analysis, patients in North America were also more likely to rate their disease as severe than those in Europe, but the proportions of patients were smaller (North America: 27%; Europe: 15%). In a qualitative multi-country study of 85 patients with PsA who were currently taking a DMARD, more patients in Europe (44%) than in the US (8%) named self-injection as their first choice for mode of administration [14], which was similar to the results reported herein (the proportion of patients who indicated that they would prefer an injection/infusion to an oral medicine was greater in Europe vs. North America).
The MAPP survey also highlighted the unmet needs of patients with psoriasis and PsA, with only 25 and 45% of patients who received oral therapies or biologic DMARDs, respectively, reported being very satisfied with their medications [13]. Of all the patients who completed the MAPP survey, 46% perceived that the currently available therapies for psoriasis and PsA can be worse than the disease itself, and 85% felt that there is a need for better medications [13]. Furthermore, almost 60% of patients with PsA who completed the MAPP survey were not being treated for their joint disease, and approximately 15% of patients with PsA had not seen their HCP in the last 12 months [13]; this is surprising, as most patients who are on DMARD treatments usually visit their HCP at least once a year for safety and reimbursement reasons. It is possible that patients who had not seen their HCP were undertreated; however, this is only speculation as the medications that these patients were taking at the time of the survey were not reported. Among those patients who had not seen their HCP, 30% of patients did not believe an HCP could help and 30% did not perceive their symptoms as severe enough, suggesting that there is a mismatch between patient and HCP perceptions of the disease, and better alignment between patient and HCP is needed to ensure optimal PsA management [13].
The limitations of the overall survey methodology have been previously described and include the reliance on the patients’ ability to understand and respond appropriately to the survey questions, to appropriately describe their diagnosis with PsA, and to accurately recall their own symptoms, as well as the exclusion of patients without internet access [9]. Furthermore, it is possible that selection bias may have influenced the results if different patient populations from different countries saw the invite and responded to the survey; however, this type of bias is difficult to confirm or control for. In this present analysis, data for the European population were only collected from three different countries, limiting the generalizability of the results to the whole region. It is notable that more drugs have been approved for the treatment of PsA since this survey was conducted; therefore, the results may not accurately depict the current treatment landscape. Additionally, results were not adjusted for any differences in demographics or disease characteristics, and no adjustment for multiple comparisons were conducted.
Conclusions
Overall, this analysis contributes valuable insights into patient perspectives on their PsA disease control and treatment, and highlights differences in perceptions between patients in North America and Europe, and between different countries. A key theme across all regions and countries was that, despite receiving medications for PsA, many patients still experience symptoms, wish they had a greater choice of medication, and/or would change something about their medication. While these survey findings are subject to selection bias, they do indicate that there is scope to improve the treatment of patients with PsA. Future research on treatments should consider patient preferences and priorities to improve the management of PsA.
References
Ritchlin CT, Colbert RA, Gladman DD. Psoriatic arthritis. N Engl J Med. 2017;376:957–70.
Gudu T, Gossec L. Quality of life in psoriatic arthritis. Expert Rev Clin Immunol. 2018;14:405–17.
Gossec L, Baraliakos X, Kerschbaumer A, et al. EULAR recommendations for the management of psoriatic arthritis with pharmacological therapies: 2019 update. Ann Rheum Dis. 2020;79:700–12.
Dandorfer SWH, Rech J, Manger B, Schett G, Englbrecht M. Differences in the patient’s and the physician’s perspective of disease in psoriatic arthritis. Semin Arthritis Rheum. 2012;42:32–41.
Desthieux C, Granger B, Balanescu AR, et al. Determinants of patient-physician discordance in global assessment in psoriatic arthritis: a multicenter European study. Arthritis Care Res (Hoboken). 2017;69:1606–11.
Eder L, Thavaneswaran A, Chandran V, Cook R, Gladman DD. Factors explaining the discrepancy between physician and patient global assessment of joint and skin disease activity in psoriatic arthritis patients. Arthritis Care Res (Hoboken). 2015;67:264–72.
Furst DE, Tran M, Sullivan E, et al. Misalignment between physicians and patient satisfaction with psoriatic arthritis disease control. Clin Rheumatol. 2017;36:2045–54.
Garrido-Cumbrera M, Hillmann O, Mahapatra R, et al. Improving the management of psoriatic arthritis and axial spondyloarthritis: roundtable discussions with healthcare professionals and patients. Rheumatol Ther. 2017;4:219–31.
Coates LC, Orbai A-M, Azevedo VF, et al. Results of a global, patient-based survey assessing the impact of psoriatic arthritis discussed in the context of the Psoriatic Arthritis Impact of Disease (PsAID) questionnaire. Health Qual Life Outcomes. 2020;18:173.
Rosner B. Fundamentals of biostatistics. 8th ed. Boston: Cengage Learning; 2015.
Kish L. Survey sampling. New York: Wiley; 1965.
Bergstra SA, Branco JC, Vega-Morales D, et al. Inequity in access to bDMARD care and how it influences disease outcomes across countries worldwide: results from the METEOR-registry. Ann Rheum Dis. 2018;77:1413–20.
Lebwohl MG, Bachelez H, Barker J, et al. Patient perspectives in the management of psoriasis: results from the population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis Survey. J Am Acad Dermatol. 2014;70:871–81.
Aletaha D, Husni ME, Merola JF, et al. Treatment mode preferences in psoriatic arthritis: a qualitative multi-country study. Patient Prefer Adherence. 2020;14:949–61.
Acknowledgements
The authors would like to thank all the participants for completing the survey.
Funding
This study was sponsored by Pfizer Inc. Medical writing support was funded by Pfizer Inc. Pfizer Inc contributed to the conception/design of the study, data/statistical analyses, and data interpretation. The journal’s Rapid Service Fee for this article was also funded by Pfizer Inc.
Medical Writing, Editorial, and Other Assistance
Medical writing support, under the guidance of the authors, was provided by Justine Juana, BHSc, CMC Connect, McCann Health Medical Communications and was funded by Pfizer Inc, New York, NY, USA in accordance with Good Publication Practice (GPP3) guidelines (Ann Intern Med. 2015; 163:461–4).
Authorship
All named authors meet the International Committee of Medical Journal Editors criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval of the version to be submitted. Please note that Jade Moser was unable to give her final approval of the version to be submitted due to maternity leave, but has met all the other International Committee of Medical Journal Editors criteria for authorship.
Author Contributions
PR, JC, JM, RQS, LF, and MK were involved in the conception and design of the study/analyses. RQS and JM were involved in patient recruitment and/or data acquisition. JM performed the data and statistical analyses. JC and JM contributed to the interpretation of the data. PR, LC, VA, JC, JM, RQS, LF, and MK critically revised the manuscript for important intellectual content. PR, LC, VA, JC, RQS, LF, and MK provided final approval for publication.
Prior Presentation
Data reported herein have been presented, in part, as a poster presentation at the 31st Annual French Congress of Rheumatology of the Societe Francaise de Rheumatologie (2018).
Disclosures
Pascal Richette has received consulting fees from AbbVie, Celgene, Eli Lilly, Janssen, MSD, Novartis, Pfizer Inc, and UCB. Laura C. Coates has received research funding from AbbVie, Amgen, Celgene, Eli Lilly, Novartis, and Pfizer Inc, and has received honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Eli Lilly, Galapagos, Gilead, Janssen, Medac, Novartis, Pfizer Inc, and UCB. Valderilio F. Azevedo has received research funding from AbbVie, Bristol-Myers Squibb, Eli Lilly, Genentech, GSK, Pfizer Inc, and UCB, and has received consulting and speaker fees from AbbVie, Celltrion, Eli Lilly, Janssen, Pfizer Inc, and Roche. Jade Moser is an employee of The Harris Poll. Ruben Queiro-Silva has received research grants from AbbVie, Janssen, and Novartis, and has received consulting and speaker fees from Celgene, Janssen, Novartis, Pfizer Inc, and UCB. Lara Fallon, Joseph C. Cappelleri and Meriem Kessouri are employees and shareholders of Pfizer Inc.
Compliance with Ethics Guidelines
Patients were recruited from online market research panels made up of members who agreed to participate in this type of research. Qualified respondents provided informed consent to complete the research. The surveys were noninterventional and were not conducted as a clinical study. All respondents agreed to participate but ethics approval was not required.
Data Availability
The datasets generated during and/or analyzed during the current study are available from Pfizer via Vivli on reasonable request. Subject to certain criteria, conditions and exceptions, Pfizer may also provide access to the related individual de-identified participant data. See https://www.pfizer.com/science/clinical-trials/trial-data-and-results for more information.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Richette, P., Coates, L.C., Azevedo, V.F. et al. Patient Perception of Medical Care for Psoriatic Arthritis in North America and Europe: Results from a Global Patient Survey. Rheumatol Ther 9, 823–838 (2022). https://doi.org/10.1007/s40744-022-00435-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40744-022-00435-y