Abstract
Background
Rituximab is a biologic medicine widely used for the treatment of autoimmune diseases and lymphoma. Several biosimilars of rituximab have been developed and marketed with the expiration of the originator rituximab’s patent; thus, systematic combination and analysis of the latest data on the efficacy and safety of biosimilars and the demonstration of the interchangeability of biosimilar agents are required.
Objective
The objective of this study was to collate available data from head-to-head randomized controlled trials (RCTs) and evaluate the efficacy and safety of biosimilar rituximab compared with the reference drug in patients with rheumatoid arthritis (RA) and non-Hodgkin’s lymphoma (NHL).
Methods
The PubMed, EMBASE, Cochrane Library, and Google Scholar databases were searched to identify head-to-head RCTs that directly compare the efficacy and safety of biosimilar rituximab and its originator. The efficacy outcome for RA was the American College of Rheumatology (ACR) response rates and the outcome for NHL was the response rate. The occurrence of adverse events (AEs) and anti-drug antibodies (ADAs) were evaluated for the safety outcome. Data on the pharmacokinetic profile were also included as a secondary outcome.
Results
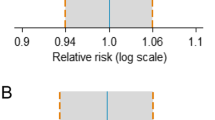
Eleven head-to-head RCTs with 3163 patients were included (1744 patients with RA and 1419 patients with NHL). Biosimilars of rituximab showed similar efficacy in the clinical response in both RA and NHL. The pooled risk ratio (RR) of the ACR 20% response rate (ACR20) response in patients with RA at weeks 24 and 48 was 0.99 (p = 0.70, 95% confidence interval [CI] 0.92–1.06) and 1.04 (p = 0.73, 95% CI 0.83–1.31), respectively. The pooled RR of the overall response at week 24 in NHL patients was 1.02 (p = 0.31, 95% CI 0.98–1.07). No significant differences were found in the formation of ADAs (RR 0.86, p = 0.20, 95% CI 0.68–1.08) or AEs (RR 1.04, p = 0.30, 95% CI 0.97–1.12).
Conclusion
This systematic review and conventional meta-analysis demonstrated the overall similarity of the long-term efficacy and safety of biosimilar rituximab to those of originator rituximab in RA and NHL patients by combining direct evidence from head-to-head trials.
PROSPERO registration No. CRD42019125138.







Similar content being viewed by others
References
FDA. Rituxan: highlights of prescribing information. 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/103705s5454lbl.pdf. Accessed 3 Mar 2019.
EMA. MabThera: summary of product characteristics. 2019. https://www.ema.europa.eu/en/documents/product-information/mabthera-epar-product-information_en.pdf. Accessed 22 Mar 2019.
EvaluatePharma® world preview 2018, outlook to 2024. 2018. http://www.evaluate.com/thought-leadership/pharma/evaluatepharma-world-preview-2018-outlook-2024. Accessed 20 Mar 2019.
Kozlowski S, Woodcock J, Midthun K, Sherman RB. Developing the nation’s biosimilars program. N Engl J Med. 2011;365(5):385–8. https://doi.org/10.1056/NEJMp1107285.
Baer WH II, Maini A, Jacobs I. Barriers to the access and use of rituximab in patients with non-Hodgkin’s lymphoma and chronic lymphocytic leukemia: a physician survey. Pharmaceuticals. 2014;7(5):530.
Gulacsi L, Brodszky V, Baji P, Kim H, Kim SY, Cho YY, et al. Biosimilars for the management of rheumatoid arthritis: economic considerations. Expert Rev Clin Immunol. 2015;11:S43–52. https://doi.org/10.1586/1744666x.2015.1090313.
Nabhan C, Parsad S, Mato AR, Feinberg BA. Biosimilars in oncology in the United States: a review. JAMA Oncol. 2018;4(2):241–7. https://doi.org/10.1001/jamaoncol.2017.2004.
Mehr SR, Brook RA. Biosimilars in the USA: will new efforts to spur approvals and access spur uptake and cost savings? Pharm Med. 2019;33(1):1–8. https://doi.org/10.1007/s40290-018-00262-z.
EMA. Guideline on similar biological medicinal products. 2014. https://www.ema.europa.eu/en/similar-biological-medicinal-products. Accessed 20 Mar 2019.
FDA. Scientific considerations in demonstrating biosimilarity to a reference product. 2015. https://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM291128.pdf. Accessed 21 Mar 2019.
FDA. Biosimilar product information. 2019. https://www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ApprovalApplications/TherapeuticBiologicApplications/Biosimilars/ucm580432.htm. Accessed 22 Mar 2019.
EMA. Biosimilar medicines: overview. 2019. https://www.ema.europa.eu/en/human-regulatory/overview/biosimilar-medicines-overview#biosimilar-development-and-approval-in-the-eu-section. Accessed 21 Mar 2019.
EMA. Trixima EPAR summary for the public. 2016. https://www.ema.europa.eu/en/medicines/human/EPAR/truxima. Accessed 23 Mar 2019.
FDA. FDA approves first biosimilar for treatment of adult patients with non-Hodgkin’s lymphoma. 2018. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm627009.htm. Accessed 24 Feb 2019.
Leonard E, Wascovich M, Oskouei S, Gurz P, Carpenter D. Factors affecting health care provider knowledge and acceptance of biosimilar medicines: a systematic review. J Manag Care Spec Pharm. 2019;25(1):102–12. https://doi.org/10.18553/jmcp.2019.25.1.102.
Chingcuanco F, Segal JB, Kim SC, Alexander GC. Bioequivalence of biosimilar tumor necrosis factor-α inhibitors compared with their reference biologics: a systematic review. Ann Intern Med. 2016;165(8):565–74. https://doi.org/10.7326/m16-0428.
Komaki Y, Yamada A, Komaki F, Kudaravalli P, Micic D, Ido A, et al. Efficacy, safety and pharmacokinetics of biosimilars of anti-tumor necrosis factor-α agents in rheumatic diseases; a systematic review and meta-analysis. J Autoimmun. 2017;79:4–16. https://doi.org/10.1016/j.jaut.2017.02.003.
Komaki Y, Yamada A, Komaki F, Micic D, Ido A, Sakuraba A. Systematic review with meta-analysis: the efficacy and safety of CT-P13, a biosimilar of anti-tumour necrosis factor-alpha agent (infliximab), in inflammatory bowel diseases. Aliment Pharm Ther. 2017;45(8):1043–57. https://doi.org/10.1111/apt.13990.
Moots RJ, Curiale C, Petersel D, Rolland C, Jones H, Mysler E. Efficacy and safety outcomes for originator TNF inhibitors and biosimilars in rheumatoid arthritis and psoriasis trials: a systematic literature review. BioDrugs. 2018;32(3):193–9. https://doi.org/10.1007/s40259-018-0283-4.
Botteri E, Krendyukov A, Curigliano G. Comparing granulocyte colony-stimulating factor filgrastim and pegfilgrastim to its biosimilars in terms of efficacy and safety: a meta-analysis of randomised clinical trials in breast cancer patients. Eur J Cancer. 2018;89:49–55. https://doi.org/10.1016/j.ejca.2017.10.034.
Amato L, Addis A, Saulle R, Trotta F, Mitrova Z, Davoli M. Comparative efficacy and safety in ESA biosimilars vs. originators in adults with chronic kidney disease: a systematic review and meta-analysis. J Nephrol. 2018;31(3):321–32. https://doi.org/10.1007/s40620-017-0419-5.
Chiumente M, Mengato D, Messori A. Rituximab biosimilar evaluated by network meta-analysis. Haematologica. 2017;102(12):e497–8. https://doi.org/10.3324/haematol.2017.175042.
Chiumente M, Messori A. Rituximab biosimilar in rheumatoid arthritis: an enhanced-evidence assessment to evaluate equivalence with the originator based on network meta-analysis. Ther Adv Musculoskelet Dis. 2017;9(10):271–3. https://doi.org/10.1177/1759720x17721151.
Bae SC, Lee YH. Comparative efficacy and safety of biosimilar rituximab and originator rituximab in combination with methotrexate in patients with active rheumatoid arthritis: a Bayesian network meta-analysis. Int J Clin Pharmacol Ther. 2019;57(4):188–96. https://doi.org/10.5414/cp203360.
Thorlund K, Mills EJ. Sample size and power considerations in network meta-analysis. Syst Rev. 2012;1(1):41. https://doi.org/10.1186/2046-4053-1-41.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. https://doi.org/10.1136/bmj.b2700.
EMA. Truxima: assessment report. 2016. https://www.ema.europa.eu/en/documents/assessment-report/truxima-epar-public-assessment-report_en.pdf. Accessed 9 Apr 2019.
Higgins JPT, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:9. https://doi.org/10.1136/bmj.d5928.
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJM, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1–12. https://doi.org/10.1016/0197-2456(95)00134-4.
GRADEpro GDT: GRADEpro Guideline Development Tool [software]. Hamilton: McMaster University (developed by Evidence Prime, Inc.); 2015.
Guyatt GH, Oxman AD, Kunz R, Vist GE, Falck-Ytter Y, Schunemann HJ, et al. What is “quality of evidence” and why is it important to clinicians? BMJ. 2008;336(7651):995–8. https://doi.org/10.1136/bmj.39490.551019.BE.
Felson DT, Anderson JJ, Boers M, Bombardier C, Furst D, Goldsmith C, et al. American college of rheumatology preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum. 1995;38(6):727–35. https://doi.org/10.1002/art.1780380602.
Fransen J, van Riel P. The Disease Activity Score and the EULAR response criteria. Clin Exp Rheumatol. 2005;23(5):S93–9.
Felson DT, LaValley MP. The ACR34 and defining a threshold for response in rheumatic diseases: too much of a good thing. Arthritis Res Ther. 2014;16(1):101. https://doi.org/10.1186/ar4428.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60. https://doi.org/10.1136/bmj.327.7414.557.
Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34. https://doi.org/10.1136/bmj.315.7109.629.
Bredemeier M, de Oliveira FK, Rocha CM. Low- versus high-dose rituximab for rheumatoid arthritis: a systematic review and meta-analysis. Arthritis Care Res (Hoboken). 2014;66(2):228–35. https://doi.org/10.1002/acr.22116.
Park W, Bozic-Majstorovic L, Milakovic D, Berrocal-Kasay A, El-Khouri EC, Irazoque-Palazuelos F, et al. Comparison of biosimilar CT-P10 and innovator rituximab in patients with rheumatoid arthritis: a randomized controlled phase 3 trial. mAbs. 2018;2018:1–10. https://doi.org/10.1080/19420862.2018.1487912.
Suh CH, Yoo DH, Berrocal Kasay A, Chalouhi El-Khouri E, Cons Molina FF, Shesternya P, et al. Long-term efficacy and safety of biosimilar CT-P10 versus innovator rituximab in rheumatoid arthritis: 48-week results from a randomized phase III trial. BioDrugs. 2019;33(1):79–91. https://doi.org/10.1007/s40259-018-00331-4.
Eremeeva A, Chernyaeva E, Ivanov R, Nasonov E, Knyazeva L. Comparison of efficacy and safety of rituximab biosimilar, BCD-020, and innovator rituximab in patients with active rheumatoid arthritis refractory to TNFa inhibitors. Ann Rheum Dis. 2016;75:513–4. https://doi.org/10.1136/annrheumdis-2016-eular.1715.
Yoo DH, Suh CH, Shim SC, Jeka S, Cons-Molina FF, Hrycaj P, et al. A multicentre randomised controlled trial to compare the pharmacokinetics, efficacy and safety of CT-P10 and innovator rituximab in patients with rheumatoid arthritis. Ann Rheum Dis. 2016;76(3):566–70. https://doi.org/10.1136/annrheumdis-2016-209540.
Yoo DH, Suh CH, Shim SC, Jeka S, Molina FFC, Hrycaj P, et al. Efficacy, safety and pharmacokinetics of up to two courses of the rituximab biosimilar CT-P10 versus innovator rituximab in patients with rheumatoid arthritis: results up to week 72 of a phase I randomized controlled trial. BioDrugs. 2017;31(4):357–67. https://doi.org/10.1007/s40259-017-0232-7.
Cohen S, Emery P, Greenwald M, Yin D, Becker JC, Melia LA, et al. A phase I pharmacokinetics trial comparing PF-05280586 (a potential biosimilar) and rituximab in patients with active rheumatoid arthritis. Br J Clin Pharmacol. 2016;82(1):129–38. https://doi.org/10.1111/bcp.12916.
Smolen JS, Cohen SB, Tony HP, Scheinberg M, Kivitz A, Balanescu A, et al. A randomised, double-blind trial to demonstrate bioequivalence of GP2013 and reference rituximab combined with methotrexate in patients with active rheumatoid arthritis. Ann Rheum Dis. 2017;76(9):1598–602. https://doi.org/10.1136/annrheumdis-2017-211281.
Kim WS, Buske C, Ogura M, Jurczak W, Sancho JM, Zhavrid E, et al. Efficacy, pharmacokinetics, and safety of the biosimilar CT-P10 compared with rituximab in patients with previously untreated advanced-stage follicular lymphoma: a randomised, double-blind, parallel-group, non-inferiority phase 3 trial. Lancet Haematol. 2017;4(8):e362–73. https://doi.org/10.1016/S2352-3026(17)30120-5.
Ogura M, Sancho JM, Cho SG, Nakazawa H, Suzumiya J, Tumyan G, et al. Efficacy, pharmacokinetics, and safety of the biosimilar CT-P10 in comparison with rituximab in patients with previously untreated low-tumour-burden follicular lymphoma: a randomised, double-blind, parallel-group, phase 3 trial. Lancet Haematol. 2018;5(11):e543–53. https://doi.org/10.1016/s2352-3026(18)30157-1.
Jurczak W, Moreira I, Kanakasetty GB, Munhoz E, Echeveste MA, Giri P, et al. Rituximab biosimilar and reference rituximab in patients with previously untreated advanced follicular lymphoma (ASSIST-FL): primary results from a confirmatory phase 3, double-blind, randomised, controlled study. Lancet Haematol. 2017;4(8):e350–61. https://doi.org/10.1016/s2352-3026(17)30106-0.
Pfizer. A study of PF-05280586 (rituximab-Pfizer) or MabThera® (rituximab-EU) for the first-line treatment of patients with CD20-positive, low tumor burden, follicular lymphoma (REFLECTIONS B328–06) [ClinicalTrials.gov identifier NCT02213263]. National Institutes of Health, ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT02213263. Accessed 22 Mar 2019.
Faltinsen EG, Storebo OJ, Jakobsen JC, Boesen K, Lange T, Gluud C. Network meta-analysis: the highest level of medical evidence? BMJ Evid Based Med. 2018;23(2):56–9. https://doi.org/10.1136/bmjebm-2017-110887.
Papageorgiou SN, Tsiranidou E, Antonoglou GN, Deschner J, Jäger A. Choice of effect measure for meta-analyses of dichotomous outcomes influenced the identified heterogeneity and direction of small-study effects. J Clin Epidemiol. 2015;68(5):534–41. https://doi.org/10.1016/j.jclinepi.2015.01.00.
Schechtman E. Odds ratio, relative risk, absolute risk reduction, and the number needed to treat—which of these should we use? Value Health. 2002;5(5):431–6. https://doi.org/10.1046/J.1524-4733.2002.55150.x.
Seta T, Takahashi Y, Noguchi Y, Shikata S, Sakai T, Sakai K, et al. Effectiveness of Helicobacter pylori eradication in the prevention of primary gastric cancer in healthy asymptomatic people: A systematic review and meta-analysis comparing risk ratio with risk difference. PLoS One. 2017;12(8):e0183321. https://doi.org/10.1371/journal.pone.0183321.
Citrome L. Relative vs. absolute measures of benefit and risk: what’s the difference? Acta Psychiatr Scand. 2010;121(2):94–102. https://doi.org/10.1111/j.1600-0447.2009.01449.x.
FDA. Clinical review(s): BLA 761088. 2018. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2018/761088Orig1s000MedR.pdf. Accessed 5 Aug 2019.
EMA. Riximyo: assessment report. 2017. https://www.ema.europa.eu/en/documents/assessment-report/riximyo-epar-public-assessment-report_en.pdf. Accessed 5 Aug 2019.
EMA. Immunogenicity assessment of biotechnology-derived therapeutic proteins. 18 May 2017. https://www.ema.europa.eu/en/immunogenicity-assessment-biotechnology-derived-therapeutic-proteins. Accessed 29 Feb 2019.
Strand V, Balsa A, Al-Saleh J, Barile-Fabris L, Horiuchi T, Takeuchi T, et al. Immunogenicity of biologics in chronic inflammatory diseases: a systematic review. BioDrugs. 2017;31(4):299–316. https://doi.org/10.1007/s40259-017-0231-8.
EMA. Similar biological medicinal products containing monoclonal antibodies; non-clinical and clinical issues. 2012. https://www.ema.europa.eu/en/similar-biological-medicinal-products-containing-monoclonal-antibodies-non-clinical-clinical-issues. Accessed 3 Mar 2019.
FDA. FDA considerations in demonstrating interchangeability with a reference product. https://www.fda.gov/BiologicsBloodVaccines/GuidanceComplianceRegulatoryInformation/Guidances/General/ucm444891.htm. Accessed 3 Mar 2019.
Mulcahy AW, Hlavka JP, Case SR. Biosimilar cost savings in the United States: initial experience and future potential. Rand Health Q. 2018;7(4):3.
Gulacsi L, Brodszky V, Baji P, Rencz F, Pentek M. The rituximab biosimilar CT-P10 in rheumatology and cancer: a budget impact analysis in 28 European countries. Adv Ther. 2017;34(5):1128–44. https://doi.org/10.1007/s12325-017-0522-y.
Treadwell JR, Uhl S, Tipton K, Shamliyan T, Viswanathan M, Berkman ND, et al. Assessing equivalence and noninferiority. J Clin Epidemiol. 2012;65(11):1144–9. https://doi.org/10.1016/j.jclinepi.2012.05.001.
Acknowledgements
This research was supported by the Chung-Ang University Research Scholarship Grants, 2018 (Soohyun Lee) and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2018R1D1A1B07046564).
Author information
Authors and Affiliations
Contributions
SL and EK designed the study. SL and HL carried out data extraction and data analysis. SL wrote the initial manuscript. All the authors contributed to the revision of the article and approved the submission of the final manuscript.
Corresponding author
Ethics declarations
Funding
This research is supported by the 2019 Chung-Ang University Research Scholarship Grants (Soohyun Lee) and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2018R1D1A1B07046564).
Conflicts of interest
Soohyun Lee, Heeyoung Lee, and Eunyoung Kim declare that they have no competing interests related to this study.
Ethical approval
Ethical approval was not applicable for this systematic review and meta-analysis.
Data availability
All the materials used in this systematic review and meta-analysis have been fully referenced.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, S., Lee, H. & Kim, E. Comparative Efficacy and Safety of Biosimilar Rituximab and Originator Rituximab in Rheumatoid Arthritis and Non-Hodgkin’s Lymphoma: A Systematic Review and Meta-analysis. BioDrugs 33, 469–483 (2019). https://doi.org/10.1007/s40259-019-00376-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40259-019-00376-z