Abstract
It is currently estimated that 11 million Canadians are living with diabetes or prediabetes. Although hyperglycemia is associated with serious complications, it is well established that improved glycemic control reduces the risk of microvascular complications and can also reduce cardiovascular (CV) complications over the long term. The UKPDS and ADVANCE landmark trials have resulted in diabetes guidelines recommending an A1C target of ≤ 7.0% for most patients or a target of ≤ 6.5% to further reduce the risk of nephropathy and retinopathy in those with type 2 diabetes (T2D), if it can be achieved safely. However, half of the people with T2D in Canada are not achieving these glycemic targets, despite advances in diabetes pharmacological management. There are many contributing factors to account for this poor outcome; however, one of the major factors is the delay in treatment advancement, particularly a resistance to insulin initiation and intensification. To simplify the process of initiating and titrating insulin in T2D patients, a group of Canadian experts reviewed the evidence and best clinical practices with the goal of providing guidance and practical recommendations to the diabetes healthcare community at large. This expert panel included general practitioners (GPs), nurses, nurse practitioners, endocrinologists, dieticians, pharmacists, and a psychologist. This article summarizes the panel recommendations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Basal Insulin Initiation
Do We Still Need Insulin?
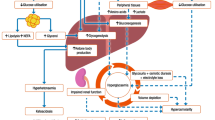
Type 2 diabetes (T2D) is a progressive disorder characterized by multiple pathophysiological defects. The core defects include insulin resistance in the muscle and liver and impaired insulin secretion due to β-cell failure [1, 2]. The progressive nature of the disease is such that it requires therapy to be intensified over time to compensate for the ongoing β-cell deficiency [2,3,4]. At the time of T2D diagnosis, more than 50% of β-cells have already been lost, and continue to decline at an average rate of 5% per year [1, 2, 5]. Therefore, the use of insulin is an appropriate option at any point in the management of T2D to replace the insulin that the pancreas is unable to produce sufficiently [1, 6]. In fact, when the maximum output of insulin has decreased to 15% or 20% of normal, non-insulin anti-hyperglycemic agents can no longer sustain glycemic control and insulin supplementation becomes a necessity [5]. The usual starting point for insulin therapy in T2D is with basal insulin owing to its simplicity and lower risk of hypoglycemia [7].
When and in Whom to Initiate Insulin in T2D
The panel recommendations as to when and in whom to initiate insulin are summarized in Table 1.
What are the Barriers to Insulin Initiation?
Clinical inertia, defined as the failure on the part of the provider to advance therapy when required, adversely affects timely management of T2D [9,10,11,12]. Insulin is often initiated late in the course of the disease, after failure with multiple antihyperglycemic agents, and at glycemic values well above the recommended targets [11,12,13,14,15]. In Canada, mean A1C levels are > 8.5% and mean diabetes duration is ≥ 9 years before initiation of basal insulin in T2D patients [13, 15]. A UK retrospective study of pharmacologically treated T2D patients on one, two, or three oral antihyperglycemic agents reported that the median time to insulin initiation was > 7 years with an A1C ≥ 7.0% and the mean A1C levels at initiation was > 9.0% [12].
There are many barriers that contribute to this delay in initiation and intensification of insulin in T2D. It is important to emphasize that many of these barriers reflect the attitudes and beliefs of both patient and provider. Identifying and addressing both provider and patient beliefs and attitudes are therefore essential to mitigate those barriers (Tables 2 and 3).
What is Your Role in Insulin Therapy?
Success in overcoming patient barriers relies greatly on listening to the patient and proactively addressing their fears and concerns [55, 56]. Open dialogue with the patient throughout the continuum of diabetes management, with an emphasis on the positive benefits of insulin therapy, will significantly enhance the outcomes for patients with diabetes. See Table 4 for review of action points with your patient.
Basal Insulin Dose and Titration Recommendations
In light of the persistent barriers contributing to delays in diabetes management with insulin, there is an urgent need for a simplified and practical approach to the initiation and intensification of insulin. Complex regimens and unrealistic targets can worsen the patient’s engagement in the process and ultimately the patient’s well-being [3, 4, 57]. Simplification allows for empowerment by engaging the patient in doable tasks, which provides the context for behavior shaping (next step goals) and self-efficacy (confidence in the face of barriers) [58].
What Do We Want in a Basal Insulin Recommendation?
-
A starting dose that can be safely applied and individualized.
-
A titration schedule that is simple and can be safely patient-driven, with a fasting blood glucose (FBG) target that can be individualized. Patient-driven titration schedules are as effective as provider-driven titration schedules [19, 59,60,61,62,63,64] and engage the patient, which in turn can lower barriers to insulin therapy [4, 65, 66].
-
Clear instructions to the patient on how the dose will be titrated, to manage expectations which will empower the patient and improve adherence to therapy [3, 4, 16, 66].
-
Recognition that insulin initiation and titration are two separate behaviors for the patient, each of which needs to be addressed in relation to patient readiness to change.
How to Select a Basal Insulin?
Three generations of basal insulins are available in Canada. The first generation of basal insulin is NPH, a human insulin that has been available for many decades, since 1946. The basal analogues (insulins detemir and glargine (Gla-100)) emerged in the 2000s and provided longer duration of action, improved day-to-day variability, reduced hypoglycemia, especially nocturnal, and did not require resuspension (as does NPH) [67]. A next generation of long-acting basal insulins—insulins glargine 300 U/mL (Gla-300) and degludec—have emerged with an extended action profile, improved safety, and the advantage of being administered in smaller volumes [29]. Table 5 summarizes the main characteristics of the currently available basal insulins. The panel recognizes that the choice of basal insulin may depend on access, cost, and clinical judgment with respect to the patient’s individual needs and lifestyle [29].
How to Dose?
There are several important concepts to remember when dosing basal insulin: (a) the starting dose will be wrong; (b) there is no maximal insulin dose; (c) titration of insulin dose is the key [8]. Each of these concepts needs to be explicitly discussed and understood by the patient in order for titration to be successful. Despite 92% of physicians agreeing that “insulin intensification is an essential element of diabetes management,” 30% of primary care physicians “never or rarely” personally intensified insulin (vs 4% of specialists) in the multinational survey MODIFY [14, 81]. Interestingly, in a recent multinational survey, HCPs generally preferred a gradual and safe approach to titration to avoid hypoglycemia whereas patients are frustrated by time to reach goal [66]. It is therefore important to manage the patient’s expectations.
The starting dose for basal insulin recommended by this panel is 10 U/day. The dose should be incrementally increased on a regular basis using target FBG as the determinant for dose adjustments. At initiation, educating patients that many people will need at least 40–50 units of basal insulin to achieve target FBG is useful for goal setting and behavior shaping. This may help mitigate patient fear/reluctance to up-titrate [8].
Box 1A details the recommendations by the panel for basal insulin dose and titration.
Box 1B provides a summary of key recommendations, including a starting dose and titration schedule.
Basal Insulin Dose and Titration Recommendations (2017)
Box 1A: 2017 recommendations by the panel for basal insulin dose and titration
Panel recommendations | Comments | |
|---|---|---|
The initial dosea | 10 U/day http://guidelines.diabetes.ca/browse/appendices/appendix3 Other considerations: Using FBG as starting point: e.g., if FBG is 16 mmol/L start at 16 U [59] | May need to be lower for some patients—recall that the starting dose should be individualized [14] http://guidelines.diabetes.ca/cdacpg_resources/CPG_Quick_Reference_Guide_WEB.pdf The lower dosages have the advantage of decreasing the risk of a hypoglycemic reaction with the first injection, but make the titration period a bit longer Discuss and negotiate your patient’s expectation |
Fasting SMBG target | Target should be 4.0–7.0 mmol/L for most people Patient/HCP contact recommended at 7.0 mmol/L. HCP may then suggest continuing to 4.0–5.5 mmol/L | Individualize target with a step approach (within 3 months) [14] http://guidelines.diabetes.ca/cdacpg_resources/CPG_Quick_Reference_Guide_WEB.pdf Important to educate that diabetes is a progressive disease and this is a moving target [4] |
Dose adjustments | Select a simple titration algorithm that matches patient lifestyle [57] The following dose adjustment algorithms have been shown to be safe and effective. Select the one that is easiest for the patient to follow: One easy titration algorithm is 1 unit every dayb [19, 63, 64, 66] Other titration algorithms include: 2 units twice weekly based on lowest fasting SMGB value of the last 3 days [26, 27, 62, 86] Every week, based on lowest fasting SMGB value of the last 3 days [26, 63, 64] Other considerations: If (nocturnal) hypoglycemia occurs (BG < 4.0 mmol/L) reduce the dose by 2–4 units, or 10% of the basal dose based on clinical judgement [57] For other considerations, see Table 6 | Measure glucose level at least every morning before breakfastc [57] http://guidelines.diabetes.ca/browse/appendices/appendix3 Remind patient to adjust the basal insulin based on morning glucose not bedtime glucosec [57] Assess for possible hypoglycemia (< 4.0 mmol/L) and decrease titration [52] http://guidelines.diabetes.ca/fullguidelines/chapter14 Recognize that patient fear of hypoglycemia is easily elicited (hypoglycemia is a traumatic stress) and that providers underestimate the psychological impact of nonsevere hypoglycemia [51] Mitigating hypoglycemia: Is there an identifiable cause? [52] http://guidelines.diabetes.ca/fullguidelines/chapter14 Teach patients how to prevent, recognize, and treat hypoglycemia [52] http://guidelines.diabetes.ca/fullguidelines/chapter14 Confirm with patient that it is not “pseudo-hypoglycemia”. Explain what pseudo-hypoglycemiad is and ways to mitigate it [54] If no identifiable and preventable cause is identified, reduce the dose Confirm patient is using an accurate glucometer |
Optimal/maximum basal insulin dose | Educate the patient of their expected dose [3, 57] In most studies: 40 to 50 units is needed [8, 19, 26, 27, 66] Communicate how long it will take them to reach target (e.g., if the expected dose is 60 units at 1 U/day increase, then it will take on average 6 weeks) | Indication that basal insulin is not enough includes: Up-titrations without a corresponding drop on BG (verify patient adherence and check injection sites). http://www.fit4diabetes.com/canada-english/fit-recommendations/ Patient has surpassed 1 U/kg/day of basal insulin without sufficient FBG control [87] FBG in target, but A1C above target |
Frequent Questions and What to Do with Previous Drugs When Initiating Basal Insulin
Tables 6 and 7 outline some of the frequently encountered questions and concerns facing HCPs when initiating and titrating basal insulin.
Patient Support and Medical Follow-up
How to Ensure Success of Basal Insulin Management?
The success of basal insulin initiation and titration relies not only on identifying and addressing the patient and practitioner barriers but also on contact frequency with the patient. Post-initiation follow-up may occur by many means including via phone, text, email (depending on jurisdiction), cloud, or virtual consult. Regular contact presents an opportunity to provide or revisit diabetes education, to provide support to patients on how to effectively self-manage their disease and to identify any causes of concern [3, 34]. Furthermore, titration should be revisited when the patient is not achieving goal, hypoglycemia occurs, or there is a change in the insulin type or brand (e.g., biosimilar) [87].
The panel provides guidelines for medical follow-up with patients in Box 2.
Panel Recommendations for Medical Follow-up with Diabetes HCPs
Box 2: panel recommendations for medical follow-up with diabetes HCPs [87, 91]
When | What and why |
|---|---|
24–72 h | When initiating insulin or titration Support insulin initiation and reinforce titration |
1–2 week(s) | Patients report BG readings Ensure titration is occurring normally |
1 month | Patients report BG readings Ensure titration is occurring normally (it is encouraged to continue with biweekly contacts thereafter) |
3 months | A1C measurement If not at goal, patient may continue with titration for another 3 months This contact point should occur in person or by virtual consult |
6 months | A1C measurement Follow-up of titration If A1C above target, review glycemic profile and consider adding mealtime insulin |
Within 24 h of hypoglycemia | Educate patient on recognizing, preventing, and treating hypoglycemia If recurrent hypoglycemia occurs, re-evaluate titration schedule or reduce dose (frequent, recurrent hypoglycemia is typically defined as 1–2 lows in 1 week) |
Conclusion
Several factors underlie the importance of the initiative put forth by this expert panel: there is a rising prevalence of diabetes [98]; half of the T2D population is not at target, among which 61% were receiving insulin therapy [99], suggesting delayed insulin initiation and intensification; there are multiple titration algorithms to choose from which adds to the confusion and complexity for patients and providers; and the arrival of new long-acting basal insulins and other pharmacological and technological advances that require consideration. This document was developed by a multidisciplinary panel to address frequently asked questions on insulin initiation and titration, and it establishes simple and practical guidelines for diabetes HCPs for effective initiation and titration of basal insulin, with the intent that it may translate to effective glycemic outcomes in clinical practice.
Compliance with Ethical Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
References
DeFronzo RA, Eldor R, Abdul-Ghani M. Pathophysiologic approach to therapy in patients with newly diagnosed type 2 diabetes. Diabetes Care. 2013;36(Suppl 2):S127–38.
Pratley RE. The early treatment of type 2 diabetes. Am J Med. 2013;126(9 Suppl 1):S2–9.
Ross SA. Breaking down patient and physician barriers to optimize glycemic control in type 2 diabetes. Am J Med. 2013;126(9 Suppl 1):S38–48.
Barag SH. Insulin therapy for management of type 2 diabetes mellitus: strategies for initiation and long-term patient adherence. J Am Osteopath Assoc. 2011;111(7 Suppl 5):S13–9.
Lebovitz HE. Diabetes Review 1999;7(3):139–53.
Palumbo PJ. The case for insulin treatment early in type 2 diabetes. Cleve Clin J Med. 2004;71(5):385–6.
Holman RR, Farmer AJ, Davies MJ, et al. Three-year efficacy of complex insulin regimens in type 2 diabetes. N Engl J Med. 2009;361(18):1736–47.
Cheng AYY. The rules of 3s: insulin use in type 2 diabetes. Can Diabetes. 2011;24(1):3–9.
Lovshin JA, Zinman B. Diabetes: clinical inertia–a barrier to effective management of T2DM. Nat Rev Endocrinol. 2013;9(11):635–6. https://doi.org/10.1038/nrendo.2013.185.
Phillips LS, Branch WT, Cook CB, et al. Clinical inertia. Ann Intern Med. 2001;135(9):825–34.
Khunti K. Clinical inertia in the management of type 2 diabetes mellitus: a focused literature review. Br J Diabetes. 2015;15(2):65–9.
Khunti K, Wolden ML, Thorsted BL, Andersen M, Davies MJ. Clinical inertia in people with type 2 diabetes: a retrospective cohort study of more than 80,000 people. Diabetes Care. 2013;36(11):3411–7. https://doi.org/10.2337/dc13-0331.
Harris SB, Kapor J, Lank CN, Willan AR, Houston T. Clinical inertia in patients with T2DM requiring insulin in family practice. Can Fam Phys. 2010;56(12):e418–24.
Khunti K, Millar-Jones D. Clinical inertia to insulin initiation and intensification in the UK: a focused literature review. Prim Care Diabetes. 2017;11(1):3–12.
Cheng AY, et al. Barriers to achieve A1C target in patients with T2D: Key learnings from a Canadian personal practice assessment. December 2015; IDF/WDC Meeting 0738-P.
Ng CJ, Lai PS, Lee YK, Azmi SA, Teo CH. Barriers and facilitators to starting insulin in patients with type 2 diabetes: a systematic review. Int J Clin Pract. 2015;69(10):1050–70.
Kunt T, Snoek FJ. Barriers to insulin initiation and intensification and how to overcome them. Int J Clin Pract Suppl. 2009;164:6–10.
Gerstein HC, Bosch J, Dagenais GR, et al. Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med. 2012;367(4):319–28.
Gerstein HC, Yale JF, Harris SB, Issa M, Stewart JA, Dempsey E. A randomized trial of adding insulin glargine vs. avoidance of insulin in people with type 2 diabetes on either no oral glucose-lowering agents or submaximal doses of metformin and/or sulphonylureas. The Canadian INSIGHT (Implementing New Strategies with Insulin Glargine for Hyperglycaemia Treatment) Study. Diabet Med. 2006;23(7):736–42.
Ritzel R, Roussel R, Bolli GB, et al. Patient-level meta-analysis of the EDITION 1, 2 and 3 studies: glycaemic control and hypoglycaemia with new insulin glargine 300 U/ml versus glargine 100 U/ml in people with type 2 diabetes. Diabetes Obes Metab. 2015;17(9):859–67.
Riddle MC, Bolli GB, Ziemen M, Muehlen-Bartmer I, Bizet F, Home PD. New insulin glargine 300 units/mL versus glargine 100 units/mL in people with type 2 diabetes using basal and mealtime insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (EDITION 1). Diabetes Care. 2014;37(10):2755–62.
Yki-Jarvinen H, Bergenstal R, Ziemen M, Wardecki M, Muehlen-Bartmer I, Boelle E, et al. New insulin glargine 300 units/mL versus glargine 100 units/mL in people with type 2 diabetes using oral agents and basal insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (EDITION 2). Diabetes Care. 2014;37(12):3235–43.
Bolli GB, Riddle MC, Bergenstal RM, et al. New insulin glargine 300 U/ml compared with glargine 100 U/ml in insulin-naive people with type 2 diabetes on oral glucose-lowering drugs: a randomized controlled trial (EDITION 3). Diabetes Obes Metab. 2015;17(4):386–94.
Riddle MC, Bolli GB, Home PD, et al. Efficacy and safety of flexible versus fixed dosing intervals of insulin glargine 300 U/mL in people with type 2 diabetes. Diabetes Technol Ther. 2016;18(4):252–7.
Matsuhisa M, Koyama M, Cheng X, et al. New insulin glargine 300 U/ml versus glargine 100 U/ml in Japanese adults with type 1 diabetes using basal and mealtime insulin: glucose control and hypoglycaemia in a randomized controlled trial (EDITION JP 1). Diabetes Obes Metab. 2016;18(4):375–83.
Zinman B, Philis-Tsimikas A, Cariou B, et al. Insulin degludec versus insulin glargine in insulin-naive patients with type 2 diabetes: a 1-year, randomized, treat-to-target trial (BEGIN Once Long). Diabetes Care. 2012;35(12):2464–71.
Gough SC, Bhargava A, Jain R, Mersebach H, Rasmussen S, Bergenstal RM. Low-volume insulin degludec 200 units/ml once daily improves glycemic control similarly to insulin glargine with a low risk of hypoglycemia in insulin-naive patients with type 2 diabetes: a 26-week, randomized, controlled, multinational, treat-to-target trial: the BEGIN LOW VOLUME trial. Diabetes Care. 2013;36(9):2536–42.
Onishi Y, Iwamoto Y, Yoo SJ, Clauson P, Tamer SC, Park S. Insulin degludec compared with insulin glargine in insulin-naive patients with type 2 diabetes: a 26-week, randomized, controlled, Pan-Asian, treat-to-target trial. J Diabetes Investig. 2013;4(6):605–12.
Standl E, Owen DR. New long-acting basal insulins: does benefit outweigh cost? Diabetes Care. 2016;39(Supplement 2):S172–9.
Terauchi Y, Koyama M, Cheng X, et al. New insulin glargine 300 U/ml versus glargine 100 U/ml in Japanese people with type 2 diabetes using basal insulin and oral antihyperglycaemic drugs: glucose control and hypoglycaemia in a randomized controlled trial (EDITION JP 2). Diabetes Obes Metab. 2016;18(4):366–74.
Home PD, Bergenstal RM, Bolli GB, et al. New insulin glargine 300 units/mL versus glargine 100 Units/mL in people with type 1 diabetes: a randomized, phase 3a, open-label clinical trial (EDITION 4). Diabetes Care. 2015;38(12):2217–25.
Garber AJ, King AB, Del Prato S, et al. Insulin degludec, an ultra-longacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 2 diabetes (BEGIN Basal-Bolus Type 2): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet. 2012;379(9825):1498–507.
Polinski JM, Smith BF, Curtis BH, et al. Barriers to insulin progression among patients with type 2 diabetes: a systematic review. Diabetes Educ. 2013;39(1):53–65.
Powers MA, Bardsley J, Cypress M, et al. Diabetes self-management education and support in type 2 diabetes. Diabetes Educ. 2017;43(1):40–53. https://doi.org/10.1177/0145721716689694.
Al Hamarneh YN, Charrois T, Lewanczuk R, Tsuyuki RT. Pharmacist intervention for glycaemic control in the community (the RxING study). BMJ Open. 2013;3(9):e003154.
Furler J, O’Neal D, Speight J, et al. Supporting insulin initiation in type 2 diabetes in primary care: results of the Stepping Up pragmatic cluster randomised controlled clinical trial. BMJ. 2017;356:j783.
Kovacs Burns K, Nicolucci A, Holt RI, et al. Diabetes attitudes, wishes and needs second study (DAWN2): cross-national benchmarking indicators for family members living with people with diabetes. Diabet Med. 2013;30(7):778–88.
Peyrot M, Burns KK, Davies M, et al. Diabetes attitudes wishes and needs 2 (DAWN2): a multinational, multi-stakeholder study of psychosocial issues in diabetes and person-centred diabetes care. Diabetes Res Clin Pract. 2013;99(2):174–84.
Funnell MM. Overcoming barriers to the initiation of insulin therapy. Clin Diabetes. 2007;25(1):36–8.
Meece J. Dispelling myths and removing barriers about insulin in type 2 diabetes. Diabetes Educ. 2006;32(1 Suppl):9S–18S.
Skovlund SE, Peyrot M. The diabetes attitudes, wishes, and needs (DAWN) program: a new approach to improving outcomes of diabetes care. Diabetes Spect. 2005;18(3):136–42.
Polonsky WH, Jackson RA. What’s so tough about taking insulin? Addressing the problem of psychological insulin resistance in type 2 diabetes. Clin Diabetes. 2004;22(3):147–50.
Polonsky WH, Fisher L, Guzman S, Villa-Caballero L, Edelman SV. Psychological insulin resistance in patients with type 2 diabetes: the scope of the problem. Diabetes Care. 2005;28(10):2543–5.
Davies MJ, Gagliardino JJ, Gray LJ, Khunti K, Mohan V, Hughes R. Real-world factors affecting adherence to insulin therapy in patients with type 1 or type 2 diabetes mellitus: a systematic review. Diabet Med. 2013;30(5):512–24.
Karter AJ, Subramanian U, Saha C, et al. Barriers to insulin initiation: the translating research into action for diabetes insulin starts project. Diabetes Care. 2010;33(4):733–5.
Lebovitz HE. Type 2 diabetes: an overview. Clin Chem. 1999;45(8 Pt 2):1339–45.
UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837–53.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HAW. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359(15):1577–89.
Kostev K, Dippel FW, Rathmann W. Risk of hypoglycaemia in type 2 diabetes patients under different insulin regimens: a primary care database analysis. Ger Med Sci. 2015;12(13):Doc01.
Polonsky WH, Fisher L, Hessler D, Edelman SV. Identifying the worries and concerns about hypoglycemia in adults with type 2 diabetes. J Diabetes Complic. 2015;29(8):1171–6.
Vallis M, Jones A, Pouwer F. Managing hypoglycemia in diabetes may be more fear management than glucose management: a practical guide for diabetes care providers. Curr Diabetes Rev. 2014;10(6):364–70.
Clayton D, Woo V, Yale J-F. Canadian Diabetes Association 2013 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: hypoglycemia. Can J Diabetes 2013;37(suppl 1):S69–71.
Harper W, Clement M, Goldenberg R, et al. Canadian Diabetes Association 2013 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: pharmacologic management of type 2 diabetes. Can J Diabetes 2013;37(suppl 1):S61–68.
Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care. 2013;36(5):1384–95.
Sorli C, Heile MK. Identifying and meeting the challenges of insulin therapy in type 2 diabetes. J Multidiscip Healthc. 2014;7:267–82.
Polonsky WH, Arsenault J, Fisher L, et al. Initiating insulin: how to help people with type 2 diabetes start and continue insulin successfully. Int J Clin Pract. 2017. https://doi.org/10.1111/ijcp.12973.
Arnolds S, Heise T, Flacke F, Sieber J. Common standards of basal insulin titration in type 2 diabetes. J Diabetes Sci Technol. 2013;7(3):771–88.
Sabourin BC, Pursley S. Psychosocial issues in diabetes self-management: strategies for healthcare providers. Can J Diabetes. 2013;37(1):36–40.
Davies M, Storms F, Shutler S, Bianchi-Biscay M, Gomis R. Improvement of glycemic control in subjects with poorly controlled type 2 diabetes: comparison of two treatment algorithms using insulin glargine. Diabetes Care. 2005;28(6):1282–8.
Blonde L, Merilainen M, Karwe V, Raskin P. Patient-directed titration for achieving glycaemic goals using a once-daily basal insulin analogue: an assessment of two different fasting plasma glucose targets—the TITRATE study. Diabetes Obes Metab. 2009;11(6):623–31.
Meneghini L, Koenen C, Weng W, Selam JL. The usage of a simplified self-titration dosing guideline (303 Algorithm) for insulin detemir in patients with type 2 diabetes–results of the randomized, controlled PREDICTIVE 303 study. Diabetes Obes Metab. 2007;9(6):902–13.
Philis-Tsimikas A, Brod M, Niemeyer M, Ocampo Francisco AM, Rothman J. Insulin degludec once-daily in type 2 diabetes: simple or step-wise titration (BEGIN: once simple use). Adv Ther. 2013;30(6):607–22.
Berard L, Harris SB, Yale J-F, Groleau M, Javadi P, Stewart J. Safety and efficacy of a pragmatic self-titration 1 unit/day (INSIGHT) algorithm for insulin glargine 300 U/mL (Gla-300). Can J Diabetes. 2016;40(5):S47.
Yale JF, Berard L, Groleau M, Javadi P, Stewart J, Harris SB. TITRATION: a randomized study to assess 2 treatment algorithms with new insulin glargine 300 units/ml. Can J Diabetes. 2017;41(5):478–84. https://doi.org/10.1016/j.jcjd.2017.06.007.
Peyrot M, Barnett AH, Meneghini LF, Schumm-Draeger PM. Insulin adherence behaviours and barriers in the multinational global attitudes of patients and physicians in insulin therapy study. Diabet Med. 2012;29(5):682–9.
Berard L, Bonnemaire M, Mical M, Edelman S. Insights into optimal basal insulin titration in type 2 diabetes: results of a quantitative survey. Diabetes Obes Metab. 2018;20(2):301–8.
Owens DR, Matfin G, Monnier L. Basal insulin analogues in the management of diabetes mellitus: what progress have we made? Diabetes Metab Res Rev. 2014;30(2):104–19.
TOUJEO™SoloSTAR® Product Monograph May 28, 2015.
FPD. Clinical pharmacology and biopharmaceutics review(s). 2015. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/203313Orig1s000_203314Orig1s000ClinPharmR.pdf.
Marso SP, McGuire DK, Zinman B, et al. Efficacy and safety of degludec versus glargine in type 2 diabetes. N Engl J Med. 2017;377:723–32.
Heise T, Nosek L, Bottcher SG, Hastrup H, Haahr H. Ultra-long-acting insulin degludec has a flat and stable glucose-lowering effect in type 2 diabetes. Diabetes Obes Metab. 2012;14(10):944–50.
Lajara R, Cengiz E, Tanenberg RJ. The role of the new basal insulin analogs in addressing unmet clinical needs in people with type 1 and type 2 diabetes. Curr Med Res Opin. 2017;33(6):1045–55.
Bramlage P, Bluhmki T, Fleischmann H, et al. Determinants of weight change in patients on basal insulin treatment: an analysis of the DIVE registry. BMJ. 2017;5(1):e000301.
Garber AJ. Will the next generation of basal insulins offer clinical advantages? Diabetes Obes Metab. 2014;16(6):483–91.
Owens DR. Clinical evidence for the earlier initiation of insulin therapy in type 2 diabetes. Diabetes Technol Ther. 2013;15:776–85.
Steinstraesser A, Schmidt R, Bergmann K, Dahmen R, Becker RH. Investigational new insulin glargine 300 U/ml has the same metabolism as insulin glargine 100 U/ml. Diabetes Obes Metab. 2014;16(9):873–6.
Goldman J, Kapitza C, Pettus J, Heise T. Understanding how pharmacokinetic and pharmacodynamic differences of basal analog insulins influence clinical practice. Curr Med Res Opin. 2017;33(10):1821–31.
de Galan BE. Insulin glargine 300 U/mL in the management of diabetes: clinical utility and patient perspectives. Patient Prefer Adher. 2016;10:2097–106.
Becker RH, Dahmen R, Bergmann K, Lehmann A, Jax T, Heise T. New insulin glargine 300 units. mL-1 provides a more even activity profile and prolonged glycemic control at steady state compared with insulin glargine 100 units. mL-1. Diabetes Care. 2015;38(4):637–43. https://doi.org/10.2337/dc14-0006.
Marso SP, McGuire DK, Zinman B, et al. Design of DEVOTE (trial comparing cardiovascular safety of insulin degludec vs insulin glargine in patients with type 2 diabetes at high risk of cardiovascular events) - DEVOTE 1. Am Heart J. 2016;179:175–83. https://doi.org/10.1016/j.ahj.2016.06.004.
Shah BR, Hux JE, Laupacis A, Zinman B, van Walraven C. Clinical inertia in response to inadequate glycemic control: do specialists differ from primary care physicians? Diabetes Care. 2005;28(3):600–6.
Blonde L, Merilainen M, Karwe V, Raskin P, Group TS. Patient-directed titration for achieving glycaemic goals using a once-daily basal insulin analogue: an assessment of two different fasting plasma glucose targets—the TITRATETM study. Diabetes Obes Metab. 2009;11(6):623–31.
Riddle MC, Rosenstock J, Gerich J. The treat-to-target trial. Randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients. Diabetes Care. 2003;26(11):3080–6.
Yki-Jarvinen H, Kauppinen-Makelin R, Tiikkainen M, et al. Insulin glargine or NPH combined with metformin in type 2 diabetes: the LANMET study. Diabetologia. 2006;49(3):442–51.
Harris S, Yale JF, Dempsey E, Gerstein H. Can family physicians help patients initiate basal insulin therapy successfully? Randomized trial of patient-titrated insulin glargine compared with standard oral therapy: lessons for family practice from the Canadian INSIGHT trial. Can Fam Physician. 2008;54(4):550–8.
Meneghini L, Atkin SL, Gough SC, et al. The efficacy and safety of insulin degludec given in variable once-daily dosing intervals compared with insulin glargine and insulin degludec dosed at the same time daily: a 26-week, randomized, open-label, parallel-group, treat-to-target trial in individuals with type 2 diabetes. Diabetes Care. 2013;36(4):858–64.
Partners HealthCare. Partners guidelines for the treatment of type 2 diabetes in the non-pregnant adult. Partners Healthcare; 2012. http://vdc.partners.org/guidelines/Guidelines_7-10-12.pdf.
Staples H, Yale JF. Starting Insulin. Booklet based on INSIGHT Study conducted 2002–2004. Aventis Pharma.
Glasgow Diabetes Managed Clinical Network. Guidelines for insulin initiation and adjustment in primary care in patients with type 2 diabetes: for the guidance of diabetes specialist nurses. Greater Glasgow and Clyde: NHS; 2010.
Heise T, Meneghini LF. Insulin stacking versus therapeutic accumulation: understanding the differences. Endocr Pract. 2014;20(1):75–83.
Canadian Diabetes Association Clinical Practice Guidelines Expert Committee. Canadian Diabetes Association 2013 clinical practice guidelines for the prevention and management of diabetes in Canada. Can J Diabetes. 2013;37:S1–212.
Canadian Diabetes Association Clinical Practice Guidelines Expert Committee. Appendix 3: Examples of insulin initiation and titration regimens in people with type 2 diabetes. Can J Diabetes 2013;37:S200–S1.
Bergenstal RM, Johnson M, Powers MA, et al. Adjust to target in type 2 diabetes: comparison of a simple algorithm with carbohydrate counting for adjustment of mealtime insulin glulisine. Diabetes Care. 2008;31(7):1305–10.
Buse JB, Wolffenbuttel BH, Herman WH, et al. The DURAbility of basal versus lispro mix 75/25 insulin Efficacy (DURABLE) trial: comparing the durability of lispro mix 75/25 and glargine. Diabetes Care. 2011;34(2):249–55.
Kennedy L, Herman WH, Strange P, Harris A. Impact of active versus usual algorithmic titration of basal insulin and point-of-care versus laboratory measurement of HbA1c on glycemic control in patients with type 2 diabetes: the Glycemic Optimization with algorithms and labs at Point of Care (GOAL A1C) trial. Diabetes Care. 2006;29(1):1–8.
Charbonnel B, DeFronzo R, Davidson J, et al. Pioglitazone use in combination with insulin in the prospective pioglitazone clinical trial in macrovascular events study (PROactive19). J Clin Endocrinol Metab. 2010;95(5):2163–71.
Arnolds S, Dellweg S, Clair J, et al. Further improvement in postprandial glucose control with addition of exenatide or sitagliptin to combination therapy with insulin glargine and metformin: a proof-of-concept study. Diabetes Care. 2010;33(7):1509–15.
Canadian Diabetes Association. Diabetes charter for Canada 2016. http://www.diabetes.ca/getmedia/513a0f6c-b1c9-4e56-a77c-6a492bf7350f/diabetes-charter-backgrounder-national-english.pdf.aspx.
Leiter LA, Berard L, Bowering CK, et al. Type 2 diabetes mellitus management in Canada: is it improving? Can J Diabetes. 2013;37(2):82–9.
Acknowledgements
Funding
This publication was supported by Sanofi-Aventis Canada (Laval, Quebec), who funded editorial and managerial support in the preparation of this publication, provided by a third party, HIT Global Consulting Services Inc. The article processing charges were also funded by Sanofi-aventis Canada (Laval, Quebec).
Medical Writing and Editorial Assistance
The manuscript was designed, drafted, and edited by the panel with the help of medical writer, Maria Ferraiuolo of HIT Global Consulting Services Inc. The authors, individually and collectively, were responsible for content and editorial decisions.
Authorship
The manuscript was conceived by the panel of experts recognized here as the co-authors. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published. All authors had full access to all of the data used for this article and take complete responsibility for the integrity and accuracy of the data analysis.
Disclosures
Lori Berard has received consultancy/advisory board honorarium from Sanofi, Novo Nordisk, Eli Lilly, BD, and MontMed. Noreen Antonishyn has received consultancy/advisory board honorarium from Sanofi. Kathryn Arcudi has received consultancy/advisory board honorarium from Janssen, Abbott Nutrition, Astra Zeneca, Eli Lilly, and Sanofi. Sarah Blunden has received consultancy/advisory board honorarium from Ascencia, Abbott Diabetes, Eli Lilly, Medtronic, Roche Diagnostics, and Sanofi. Alice Cheng has received consultancy/advisory board honorarium, speaker honorarium, or research support from Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Janssen, Merck, Novo Nordisk, Sanofi, Servier, and Takeda. Ronald Goldenberg has received consultancy/advisory board honorarium, speaker honorarium, or research support from Eli Lilly, Novo Nordisk, and Sanofi. Stewart Harris has received consultancy/advisory board honorarium or research support from Sanofi, Novo Nordisk, AstraZeneca, Bi/Lilly, Merck, and Janssen. Shelley Jones has received consultancy/advisory board honorarium from Abbott, AZ, Janssen, Eli Lilly, Merck, Novo Nordisk, and Sanofi. Upender Mehan has received consultancy/advisory board honorarium or unrestricted funding from Sanofi, AstraZeneca, Amgen, Janssen, Novo Nordisk, and Dairy Farmers of Canada. James Morrell has received consultancy/advisory board honorarium or research support from AstraZeneca, Novo Nordisk, Island Health, Janssen, Eli Lilly, Abbott, and MontMed. Robert Roscoe has received consultancy/advisory board honorarium or speaker honorarium/other from Novo Nordisk, Abbott Diabetes Care, Janssen, AstraZeneca, Merck Canada, Sanofi Canada, Banting & Best Diabetes Program, New Brunswick Government, Becton–Dickinson Canada, Novo Nordisk Canada, Bayer Healthcare, BMS AstraZeneca, Pfizer Canada, Eli Lilly Canada, Medtronic Canada, Abbott Diabetes Care, Roche Diagnostics, Canadian Pharmacists Association, Canadian Diabetes Association, and New Brunswick Pharmacists Association. Rick Siemens has received consultancy/advisory board honorarium from Sanofi, AstraZeneca, Novo Nordisk, Lilly, Janssen, and Merck. Michael Vallis has received consultancy/advisory board honorarium, speaker honorarium, or research support from Sanofi, Novo Nordisk, Abbvie, Valeant, Merck, CSL Behring, and Pfizer. Jean-François Yale has received consultancy/advisory board honorarium or research support from Sanofi, Novo Nordisk, Eli Lilly, and Mylan.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to https://doi.org/10.6084/m9.figshare.5822136.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Berard, L., Antonishyn, N., Arcudi, K. et al. Insulin Matters: A Practical Approach to Basal Insulin Management in Type 2 Diabetes. Diabetes Ther 9, 501–519 (2018). https://doi.org/10.1007/s13300-018-0375-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-018-0375-7