Abstract
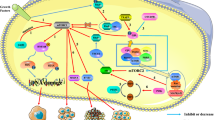
Deregulation of phosphatidylinositol-3-kinase (PI3K)/protein kinase B (Akt)/mammalian target of rapamycin (mTOR) signaling pathway contributes to prostate cancer development and progression. Here, we compared the in vitro effects of the dual PI3K/mTOR inhibitor (XL765) with those observed with the sole PI3K (XL147) or mTOR (rapamycin) inhibition in 2 non-tumor prostate epithelial cell lines, 8 prostate cancer cell lines, and 11 prostate cancer cell derivatives. We demonstrated that the XL765 treatment showed superior and proliferative effects of XL147 or rapamycin. The XL765 effects were associated to increasing the chromosome region maintenance 1 (CRM1)-mediated nuclear localization of glycogen synthase kinase 3 beta (GSK3β) and Foxo-1a with higher induction of apoptosis when compared to those observed in XL147 and rapamycin treatments. IC50 values were calculated in phosphatase and tensin homologue deleted on chromosome 10 (PTEN)-positive and PTEN-negative cell lines as well as after PTEN transfection or PTEN downmodulation by siRNA strategy revealing that the presence of this protein was associated with reduced sensitivity to PI3K and mTOR inhibitors. The comparison of IC50 values was also calculated for androgen-dependent and -independent cell lines as well as after androgen receptor (AR) transfection or the AR downmodulation by siRNA strategy revealing that androgen independence was associated with enhanced responsiveness. Our results provide a rationale to use the dual PI3K/Akt/mTOR inhibitors in hormone-insensitive prostate cancer models due to the overactivity of PI3K/Akt/mTOR in this disease condition.







Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58(2):71–96.
Qu Y, Dai B, Ye D, Kong Y, Chang K, Jia Z, et al. Constitutively active AR-V7 plays an essential role in the development and progression of castration-resistant prostate cancer. Sci Rep. 2015;5:7654.
Schalken J, Fitzpatrick JM. Enzalutamide: targeting the androgen signalling pathway in metastatic castration-resistant prostate cancer. BJU Int. 2015.
Calabrò F, Sternberg CN. Current indications for chemotherapy in prostate cancer patients. Eur Urol. 2007;51:17–26.
Nakabayashi M, Sartor O, Jacobus S, Regan MM, McKearn D, Ross RW, et al. Response to docetaxel/carboplatin-based chemotherapy as first- and second-line therapy in patients with metastatic hormone-refractory prostate cancer. BJU Int. 2008;101:308–12.
Reuter CW, Morgan MA, Ivanyi P, Fenner M, Ganser A, Grünwald V. Carboplatin plus weekly docetaxel as salvage chemotherapy in docetaxel-resistant and castration-resistant prostate cancer. World J Urol. 2010;28:391–8.
Figg WD, Chau CH, Madan RA, Gulley JL, Gao R, Sissung TM, et al. Phase II study of satraplatin and prednisone in patients with metastatic castration-resistant prostate cancer: a pharmacogenetic assessment of outcome and toxicity. Clin Genitourin Cancer. 2013;11(3):229–37.
Murillo H, Huang H, Schmidt LJ, Smith DI, Tindall DJ. Role of PI3K signaling in survival and progression of LNCaP prostate cancer cells to the androgen refractory state. Endocrinology. 2001;142:4795–805.
Liu Z, Zhu G, Getzenberg RH, Veltri RW. The upregulation of PI3K/Akt and MAP kinase pathways is associated with resistance of microtubule-targeting drugs in prostate cancer. J Cell Biochem. 2015. doi:10.1002/jcb.25091.
Feilotter HE, Nagai MA, Boag AH, Eng C, Mulligan LM. Analysis of PTEN and the 10q23 region in primary prostate carcinomas. Oncogene. 1998;16(13):1743–8.
Giri D, Ittmann M. Inactivation of the PTEN tumor suppressor gene is associated with increased angiogenesis in clinically localized prostate carcinoma. Hum Pathol. 1999;30(4):419–24.
McMenamin ME, Soung P, Perera S, Kaplan I, Loda M, Sellers WR. Loss of PTEN expression in paraffin-embedded primary prostate cancer correlates with high Gleason score and advanced stage. Cancer Res. 1999;59(17):4291–6.
Mak P, Li J, Samanta S, Chang C, Jerry DJ, Davis RJ, et al. Prostate tumorigenesis induced by PTEN deletion involves estrogen receptor β repression. Cell Rep. 2015;10(12):1982–91.
Yeh JJ, Sellers WR. Akt and prostate tumorigenesis. Curr Surg. 2000;57(5):502.
Festuccia C, Gravina GL, Muzi P, Pomante R, Ventura L, Vessella RL, et al. Bicalutamide increases phospho-Akt levels through Her2 in patients with prostate cancer. Endocrinol Relat Cancer. 2007;14:601–11.
Graff JR, Konicek BW, McNulty AM, Wang Z, Houck K, Allen S, et al. Increased AKT activity contributes to prostate cancer progression by dramatically accelerating prostate tumor growth and diminishing p27Kip1 expression. J Biol Chem. 2000;275(32):24500–5.
Festuccia C, Gravina GL, Muzi P, Millimaggi D, Dolo V, Vicentini C, et al. Akt down-modulation induces apoptosis of human prostate cancer cells and synergizes with EGFR tyrosine kinase inhibitors. Prostate. 2008;68:965–74.
Meric-Bernstam F, Akcakanat A, Chen H, Do KA, Sangai T, Adkins F, et al. PIK3CA/PTEN mutations and Akt activation as markers of sensitivity to allosteric mTOR inhibitors. Clin Cancer Res. 2012;18(6):1777–89.
Osman I, Scher HI, Drobnjak M, Verbel D, Morris M, Agus D, et al. HER-2/neu (p185neu) protein expression in the natural or treated history of prostate cancer. Clin Cancer Res. 2001;7:2643–7.
Gravina GL, Festuccia C, Angelucci A, Poletti A, Capuano D, Vicentini C, et al. Long-term presence of androgens and anti-androgens modulate EGFreceptor expression and MAP-kinase phosphorylation in androgen receptor-prostate positive cancer cells. Int J Oncol. 2004;25:97–104.
Vignot S, Faivre S, Aguirre D, Raymond E. mTOR-targeted therapy of cancer with derivatives. Ann Oncol. 2005;16(4):525–37.
Hisamatsu T, Mabuchi S, Matsumoto Y, Kawano M, Sasano T, Takahashi R, et al. Potential role of mTORC2 as a therapeutic target in clear cell carcinoma of the ovary. Mol Cancer Ther. 2013;12(7):1367–77.
Majumder PK, Febbo PG, Bikoff R, Berger R, Xue Q, McMahon LM, et al. mTOR inhibition reverses Akt-dependent prostate intraepithelial neoplasia through regulation of apoptotic and HIF-1-dependent pathways. Nat Med. 2004;10(6):594–601.
Courtney KD, Manola JB, Elfiky AA, Ross R, Oh WK, Yap JT, et al. A phase I study of everolimus and docetaxel in patients with castration-resistant prostate cancer. Clin Genitourin Cancer. 2015;13(2):113–23.
Edelman G et al. A phase I dose-escalation study of XL147 (SAR245408), a PI3K inhibitor administered orally to patients (pts) with advanced malignancies. J Clin Oncol. 2010;28(15s):3004.
Braña, et al. A phase 1 dose-escalation study of the safety, pharmacokinetics and pharmacodynamics of XL765 (SAR245409), a PI3K/TORC1/TORC2 inhibitor administered orally to patients with advanced malignancies. J Clin Oncol 28(15s):3030
Reynolds CP, Kang MH, Carol H, Lock R, Gorlick R, Kolb EA, et al. Initial testing (stage 1) of the phosphatidylinositol 3’ kinase inhibitor, SAR245408 (XL147) by the pediatric preclinical testing program. Pediatr Blood Cancer. 2013;60(5):791–8.
Foster P, Yamaguchi K, Hsu PP, Qian F, Du X, Wu J, et al. The selective PI3K inhibitor XL147 (SAR245408) inhibits tumor growth and survival and potentiates the activity of chemotherapeutic agents in preclinical tumor models. Mol Cancer Ther. 2015;14(4):931–40.
Gravina GL, Marampon F, Petini F, Biordi L, Sherris D, Jannini EA, et al. The TORC1/TORC2 inhibitor, Palomid 529, reduces tumor growth and sensitizes to docetaxel and cisplatin in aggressive and hormone-refractory prostate cancer cells. Endocrinol Relat Cancer. 2011;18(4):385–400.
Pienta KJ, Abate-Shen C, Agus DB, Attar RM, Chung LW, Greenberg NM, et al. The current state of preclinical prostate cancer animal models. Prostate. 2008;68:629.
Tsaur I, Makarević J, Juengel E, Gasser M, Waaga-Gasser AM, Kurosch M, et al. Resistance to the mTOR-inhibitor RAD001 elevates integrin α2- and β1-triggered motility, migration and invasion of prostate cancer cells. Br J Cancer. 2012;107(5):847–55.
van Bokhoven A, Varella-Garcia M, Korch C, Johannes WU, Smith EE, Miller HL, et al. Molecular characterization of human prostate carcinoma cell lines. Prostate. 2003;57(3):205–25.
Gao F, Al-Azayzih A, Somanath PR. Discrete functions of GSK3α and GSK3β isoforms in prostate tumor growth and micrometastasis. Oncotarget. 2015;6(8):5947–62.
Liu ZC, Wang HS, Zhang G, Liu H, Chen XH, Zhang F, et al. AKT/GSK-3β regulates stability and transcription of snail which is crucial for bFGF-induced epithelial-mesenchymal transition of prostate cancer cells. Biochim Biophys Acta. 2014;1840(10):3096–105.
Morgan TM, Koreckij TD, Corey E. Targeted therapy for advanced prostate cancer: inhibition of the PI3K/Akt/mTOR pathway. Curr Cancer Drug Targets. 2009;9(2):237–49.
Wang Y, Zhou D, Chen S. SGK3 is an androgen-inducible kinase promoting prostate cancer cell proliferation through activation of p70 S6 kinase and up-regulation of cyclin D1. Mol Endocrinol. 2014;28(6):935–48.
Dal Col J, Dolcetti R. GSK-3beta inhibition: at the crossroad between Akt and mTOR constitutive activation to enhance cyclin D1 protein stability in mantle cell lymphoma. Cell Cycle. 2008;7(18):2813–6.
Mori K, Hirao E, Toya Y, Oshima Y, Ishikawa F, Nose K, et al. Competitive nuclear export of cyclin D1 and Hic-5 regulates anchorage dependence of cell growth and survival. Mol Biol Cell. 2009;20(1):218–32.
De Cesare M, Cominetti D, Doldi V, Lopergolo A, Deraco M, Gandellini P, Friedlander S, Landesman Y, Kauffman MG, Shacham S, Pennati M, Zaffaroni N. Anti-tumor activity of selective inhibitors of XPO1/CRM1-mediated nuclear export in diffuse malignant peritoneal mesothelioma: the role of survivin. Oncotarget. 2015 Apr 18.
Cheng Y, Holloway MP, Nguyen K, McCauley D, Landesman Y, Kauffman MG, et al. XPO1 (CRM1) inhibition represses STAT3 activation to drive a survivin-dependent oncogenic switch in triple-negative breast cancer. Mol Cancer Ther. 2014;13(3):675–86.
Gravina GL, Marampon F, Sherris D, Vittorini F, Di Cesare E, Tombolini V, et al. Torc1/Torc2 inhibitor, Palomid 529, enhances radiation response modulating CRM1-mediated survivin function and delaying DNA repair in prostate cancer models. Prostate. 2014;74(8):852–68.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gravina, G.L., Mancini, A., Scarsella, L. et al. Dual PI3K/mTOR inhibitor, XL765 (SAR245409), shows superior effects to sole PI3K [XL147 (SAR245408)] or mTOR [rapamycin] inhibition in prostate cancer cell models. Tumor Biol. 37, 341–351 (2016). https://doi.org/10.1007/s13277-015-3725-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3725-3