Abstract
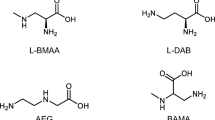
The analysis of β-N-methylamino-L-alanine (BMAA) has been validated according to AOAC international standards by a single laboratory (Glover et al. 2015). Using the same validated method, we add a second laboratory validation optimizing for different equipment. Given publicized concerns about standardizing methods across laboratories and recent reviews indicating superior results using 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate derivatization for the separation of BMAA and its isomers N-(2aminoethyl)glycine (AEG), and 2,4-diaminobuytric acid (DAB) (Bishop and Murch 2019), we add a second laboratory validation to this method demonstrating that the method is robust across laboratories using different equipment. Using the US Food and Drug Administration (FDA 2018) method for evaluating instrument parameters, we calculated a limit of detection (LOD) of 10 pg/ml for BMAA, AEG, and DAB and lower limits of quantification (LLOQ) of 37 pg/ml based on reagent blanks. In biological matrices, a higher LLOQ may be warranted for AEG and DAB. We demonstrate that the endogenous BMAA in mussel tissue can be lost by drying the hydrolyzed preparation and suggest sample preparation parameters be evaluated for robustness.


Similar content being viewed by others
Change history
25 February 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12640-021-00335-z
References
Albano R, Lobner D (2018) Transport of BMAA into neurons and astrocytes by system x c. Neurotox Res 33(1):1–5
Andrýs R, Zurita J, Zguna N, Verschueren K, De Borggraeve WM, Ilag LL (2015) Improved detection of β-N-methylamino-L-alanine using N-hydroxysuccinimide ester of N-butylnicotinic acid for the localization of BMAA in blue mussels (Mytilus edulis). Anal Bioanal Chem 407(13):3743–3750
Banack SA, Cox PA (2003) Distribution of the neurotoxic nonprotein amino acid BMAA in Cycas micronesica. Bot J Linn Soc 143(2):165–168
Banack SA, Murch SJ (2018) Methods for the chemical analysis of β-N-methylamino-LA lanine: what is known and what remains to be determined. Neurotox Res 33(1):184–191
Beach DG, Kerrin ES, Quilliam MA (2015) Selective quantitation of the neurotoxin BMAA by use of hydrophilic-interaction liquid chromatography–differential mobility spectrometry–tandem mass spectrometry (HILIC–DMS–MS/MS). Anal Bioanal Chem 407(28):8397–8409
Beach DG, Kerrin ES, Giddings SD, Quilliam MA, McCarron P (2018) Differential mobility-mass spectrometry double spike isotope dilution study of release of β-methylaminoalanine and proteinogenic amino acids during biological sample hydrolysis. Sci Rep 8(1):1–11
Bell EA (2009) The discovery of BMAA, and examples of biomagnification and protein incorporation involving other non-protein amino acids. Amyotrophic lateral sclerosis 10(sup2):21-5
Betz J, Brown P, Roman M (2011) Accuracy, precision, and reliability of chemical measurements in natural products research. Fitoterapia 82:44–52
Bishop SL, Murch SJ (2019) A systematic review of analytical methods for the detection and quantification of β-N-methylamino-L-alanine (BMAA). Analyst 145(1):13–28
Cohen SA (2012) Analytical techniques for the detection of α-amino-β-methylaminopropionic acid. Analyst 137(9):1991–2005
Cox PA, Banack SA, Murch SJ, Rasmussen U, Tien G, Bidigare RR, Metcalf JS, Morrison LF, Codd GA, Bergman B (2005) Diverse taxa of cyanobacteria produce β-N-methylamino-L-alanine, a neurotoxic amino acid. Proc Natl Acad Sci 102(14):5074–5078
Cox PA, Davis DA, Mash DC, Metcalf JS, Banack SA (2016) Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain. Proc R Soc B Biol Sci 283(1823):e20152397
Davis DA, Mondo K, Stern E, Annor AK, Murch SJ, Coyne TM, Brand LE, Niemeyer ME, Sharp S, Bradley WG, Cox PA (2019) Cyanobacterial neurotoxin BMAA and brain pathology in stranded dolphins. PLoS One 14(3):e0213346
Davis DA, Cox PA, Banack SA, Lecusay PD, Garamszegi SP, Hagan MJ, Powell JT, Metcalf JS, Palmour RM, Beierschmitt A, Bradley WG. (2020) L-Serine Reduces Spinal Cord Pathology in a Vervet Model of Preclinical ALS/MND. Journal of Neuropathology & Experimental Neurology, Jan 21
FDA (2018) Bioanalytical method validation. Guidance for industry. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, Center for Veterinary Medicine. Biopharmaceutics, Washington, DC
Gerić M, Gajski G, Domijan AM, Garaj-Vrhovac V, Filipič M, Žegura B (2019) Genotoxic effects of neurotoxin ß-N-methylamino-l-alanine in human peripheral blood cells. Chemosphere 214:623–632
Glover W, Baker TC, Murch SJ, Brown P (2015) Determination of β-N-methylamino-L-alanine, N-(2-aminoethyl) glycine, and 2, 4-diaminobutyric acid in food products containing cyanobacteria by ultra-performance liquid chromatography and tandem mass spectrometry: single-laboratory validation. J AOAC Int 98(6):1559–1565
Horwitz W (2002) AOAC guidelines for single laboratory validation of chemical methods for dietary supplements and botanicals. AOAC International, Gaithersburg, pp 12–19
Horwitz W, Albert R (2006) The Horwitz ratio (HorRat): a useful index of method performance with respect to precision. J AOAC Int 89(4):1095–1109
Janson S (2002) Cyanobacteria in symbiosis with diatoms. In: Cyanobacteria in symbiosis. Springer, Dordrecht, pp 1–10
Kerrin ES, White RL, Quilliam MA (2017) Quantitative determination of the neurotoxin β-N-methylamino-L-alanine (BMAA) by capillary electrophoresis–tandem mass spectrometry. Anal Bioanal Chem 409(6):1481–1491
Masseret E, Banack S, Boumédiène F, Abadie E, Brient L, Pernet F, Juntas-Morales R, Pageot N, Metcalf J, Cox P, Camu W (2013) Dietary BMAA exposure in an amyotrophic lateral sclerosis cluster from southern France. PLoS One 8(12)
McClure FD, Lee JK (2003) Computation of HORRAT values. J AOAC Int 86(5):1056–1058
Murch SJ, Cox PA, Banack SA (2004) A mechanism for slow release of biomagnified cyanobacterial neurotoxins and neurodegenerative disease in Guam. Proc Natl Acad Sci 101(33):12228–12231
Pharmacopeia, U. S. (2011) USP34-NF29. General chapter on validation of compendial procedures, 1225
Pierozan P, Karlsson O (2019) Mitotically heritable effects of BMAA on striatal neural stem cell proliferation and differentiation. Cell Death Dis 10(7):478
Réveillon D, Abadie E, Séchet V, Masseret E, Hess P, Amzil Z (2015) β-N-methylamino-l-alanine (BMAA) and isomers: distribution in different food web compartments of Thau lagoon, French Mediterranean Sea. Mar Environ Res 110:8–18
Réveillon D, Séchet V, Hess P, Amzil Z (2016a) Production of BMAA and DAB by diatoms (Phaeodactylum tricornutum, Chaetoceros sp., Chaetoceros calcitrans and, Thalassiosira pseudonana) and bacteria isolated from a diatom culture. Harmful Algae 58:45–50
Réveillon D, Séchet V, Hess P, Amzil Z (2016b) Systematic detection of BMAA (β-N-methylamino-l-alanine) and DAB (2, 4-diaminobutyric acid) in mollusks collected in shellfish production areas along the French coasts. Toxicon 110:35–46
Sieroslawska A, Rymuszka A (2019) Assessment of the cytotoxic impact of cyanotoxin beta-N-methylamino-L-alanine on a fish immune cell line. Aquat Toxicol 212:214–221
Tymm FJ, Bishop SL, Murch SJ (2019) A single laboratory validation for the analysis of underivatized β-N-methylamino-L-alanine (BMAA). Neurotox Res 11:1–23
Vega A, Bell EA (1967) α-Amino-β-methylaminopropionic acid, a new amino acid from seeds of Cycas circinalis. Phytochemistry 6(5):759–762
Wu X, Wu H, Gu X, Zhang R, Ye J, Sheng Q (2019) Biomagnification characteristics and health risk assessment of the neurotoxin BMAA in freshwater aquaculture products of Taihu Lake Basin, China. Chemosphere 229:332–340
Acknowledgments
We wish to thank Ken Matusuk and Freeman Van Brunt, Thermo Scientific, for mass spectrometer instrument support. We also thank J. Metcalf for contributing the cyanobacterial material, Pearse McCarron for the NRC mussel tissue, and S. Bishop for laboratory assistance. We thank the William Stamps Farish Fund for funding to support this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human and Animal Rights and Informed Consent
The human fluids were samples collected for other laboratory experiments and collections were in accordance with an Institutional Review Board and included written informed consent.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Banack, S.A. Second Laboratory Validation of β-N-Methylamino-L-Alanine, N-(2aminoethyl)Glycine, and 2,4-Diaminobuytric Acid by Ultra-Performance Liquid Chromatography and Tandem Mass Spectrometry. Neurotox Res 39, 107–116 (2021). https://doi.org/10.1007/s12640-020-00208-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00208-x