Abstract
Introduction
Trimetazidine (TMZ) was shown to reduce angina symptoms and increase the exercise capacity in stable angina (SA) patients. A new formulation allowing a once-daily (od) dosage could improve patients’ satisfaction and adherence.
Methods
ODA was a 3-month, observational, multicenter, prospective Russian study in SA patients with persistent symptoms despite therapy. Angina attack frequency, short-acting nitrate (SAN) consumption, adherence to antianginal medications, and overall efficacy and tolerability of TMZ 80 mg od were assessed in a real-world setting.
Results
A total of 3066 patients were included (mean age 62.8, 48% male). After 3 months, TMZ 80 mg od treatment led to a significant (p < 0.001) decrease in angina attack frequency (from 4.7 ± 3.5 to 0.9 ± 1.3/week) and SAN use (from 4.5 ± 3.9 to 0.7 ± 1.3/week). Overall tolerability and effectiveness were rated as “very good” by the majority of physicians. Medication adherence improved significantly, with good adherence reported by 56% of patients (vs. 24% at baseline, p < 0.0001) and non-adherence by 3% (vs. 36% at baseline, p < 0.0001) at month 3. Patient satisfaction with TMZ od was 9.5 [on a scale of 1 to 10 (very satisfied)]. Patients reported improved physical activity: more patients reported no limitations (15% vs. 1% at baseline p < 0.01), slight limitation (46% vs. 5% at baseline, p < 0.001) or moderate limitation (30% vs. 23%, p < 0.01) and fewer patients reported substantial limitation (8% vs. 52% at baseline, p < 0.001) or very marked reduction (1% vs. 19% at baseline, p < 0.01) at month 3.
Conclusion
In this prospective, observational study, TMZ 80 mg od effectively reduced angina attacks and SAN consumption, improved physical activity and adherence and was well tolerated in chronic SA patients.
Trial Registration
ISRCTN registry Identifier, ISRCTN97780949.
Funding
Servier.
Plain Language Summary
Plain language summary available for this article.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Plain Language Summary
The drug trimetazidine has been shown to be efficacious for the treatment of angina, a condition affecting more than 100 million patients worldwide. In the present study, a new formulation of trimetazidine was tested that allows patients to take only one pill per day. The new formulation was found to decrease the number of angina attacks and the use of nitroglycerine, improve physical activity and also increase the number of patients who take their prescribed medicine. The majority of participating physicians rated the new formulation favorably as being efficacious and well tolerated by patients. This new formulation could provide an opportunity to improve patients’ angina symptoms and encourage them to follow their treatment.
Introduction
Chronic stable angina pectoris (SA) is a common manifestation of coronary artery disease (CAD), which affects as many as 112 million people worldwide [1]. Angina symptoms can often be disabling, thus having a major impact on patient quality of life [2] and resulting in considerable financial burden [3].
Existing medical treatments offer an opportunity for alleviation of angina symptoms [4]. Despite this opportunity and the wide range of available treatments, suboptimal angina management has been reported [5,6,7,8]. This could be related to the substantial discordance observed between physicians and patients regarding angina burden assessment and to the underestimation of angina’s impact on patient quality of life by physicians, which could prevent them from intensifying treatment [5, 7, 8].
Another factor contributing to suboptimal angina control could be the nonadherence of patients to their treatment. Approximately 50% of patients with cardiovascular disease and/or its major risk factors have poor adherence to their prescribed medications [9], and although nonadherence is a well-known phenomenon for asymptomatic conditions such as hypertension, it has also been observed in symptomatic conditions like angina [10]. Nonadherence to medication is a complex phenomenon related to several factors [11]. Among other factors, adherence has been reported to be inversely related to the number of medication doses to be taken per day [12], suggesting that simplification of treatment regimens could translate into improved adherence and thus to potential clinical benefit.
A new formulation of trimetazidine (TMZ) 80 mg that allows a once-a-day dosage regimen was developed with the aim of simplifying treatment and thus providing an opportunity for increasing patient adherence.
The aim of the ODA trial was to assess, in a real-world clinical setting, the effectiveness and tolerability of TMZ 80 mg once daily (od), as well as patient adherence to treatment, in chronic stable angina patients with persistent symptoms despite therapy.
Methods
ODA was a 3-month observational, multicenter, prospective study conducted in Russia from March 2017 to June 2017 in a real-world clinical setting. A total of 350 physicians recruited on average 10 patients each. Patients with a confirmed diagnosis of coronary artery disease and chronic stable angina with persistent symptoms despite therapy were included into the study. Inclusion of patients into the study was exclusively determined by the decision of the physician regarding medical meaningfulness and indication for treatment with TMZ 80 mg od. Non-inclusion criteria were: age > 75 years or < 18 years; hypersensitivity to the active substance or to any of the excipients of trimetazidine 80 mg; Parkinson’s disease, parkinsonian symptoms, tremors, restless leg syndrome and other related movement disorders; moderate or severe renal impairment (creatinine clearance < 60 ml/min); stable angina pectoris Canadian Cardiovascular Society (CCS) IV; myocardial infarction (MI) in the last 3 months before inclusion; stroke/transient ischemic attack (TIA) in the last 3 months before inclusion; uncontrolled hypertension (blood pressure over 180/100 mmHg) despite ongoing antihypertensive therapy; pregnancy, breastfeeding.
Patients were treated in line with current recommendations for coronary artery disease management. Data were collected at three visits: at baseline, 1 month (M1) and 3 months (M3).
At each of the study visits, participant data were collected on the number of angina attacks, consumption of short-acting nitrates (SAN) within the week prior to the visit, evaluation of symptomatic status (angina symptoms, CCS classification), patient self-assessment of their daily activity and adherence to the current anti-anginal treatment.
At the last study visit, participants underwent a general assessment for tolerability and effectiveness of TMZ 80 mg od therapy by their physician, who rated tolerability and effectiveness as ‘very good,’ ‘good,’ ‘moderate’ or ‘poor.’
For patient self-assessment of their daily activity, patients were asked to rate how angina impacted their daily activity on a scale of 1–10 (1, no limitations; 10, very marked reduction). Answers were categorized into five categories: no limitation (0), slight limitation (1–2), moderate limitation (3–4), substantial limitation (5–7) and very marked reduction (8–10).
Adherence to all anti-anginal medications was assessed by using a previously published six-item questionnaire [13], with the following definitions: good adherence, patient responded “no” to all questions; moderate adherence, patient responded “yes” to 1–2 questions; non-adherence, patient responded “yes” to three or more questions. As a part of the assessment of adherence to TMZ, patients were asked to rate how satisfied they were with TMZ therapy on a scale of 1 (not satisfied) to 10 (very satisfied) at M1 and M3.
Any adverse drug reactions/events were collected at M1 and M3. The treating physician asked the patient to indicate whether or not an adverse event (serious or not) had occurred. In case of adverse events, adverse drug reactions or special situations that occurred during the study (both serious and non-serious), the physician completed the relevant form, accompanied by laboratory findings, hospitalization reports or other investigation results in connection with the adverse event.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Statistical Analysis
A descriptive statistical analysis was performed, assisted by SAS software, version 9.1. Patients were considered if they had valid data from all visits. All parameters were analyzed using descriptive statistics methods. The number of patients, mean value, standard deviation, minimum and maximum value or proportion by category were specified for each parameter. Differences in the numbers of angina pectoris episodes and in the necessity to use anti-anginal drugs were evaluated by the Wilcoxon’s signed rank test. p < 0.05 was considered significant. The dynamics of the parameters analyzed from visit to visit (i.e., for the blood pressure, the efficacy of therapy, etc.) were studied using both Wilcoxon signed rank test and Student’s paired t test.
Results
A total of 3066 stable angina patients were included in the study. At baseline, mean age was 62.8 ± 7.3 years, 1470 (47.9%) of patients were male, 980 patients (31.9%) had a previous history of MI, and 641 (20.9%) patients had diabetes mellitus (Table 1). Baseline therapy included statins (70%), angiotensin-converting enzyme inhibitors (56%), angiotensin receptor blockers (23%), beta-blockers (BB) (83%), calcium-channel blockers (42%), nitrates (29%) and TMZ tid or bid (37%) (Table 1).
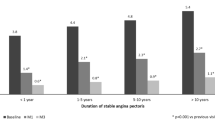
Treatment with TMZ 80 mg od (either initiation of TMZ 80 mg od or switch from TMZ tid/bid) led to a significant decrease in weekly angina attack frequency from 4.7 ± 3.5 to 2.2 ± 2.4 at M1 (p < 0.001) and to 0.9 ± 1.3 at M3 (p < 0.001) (Fig. 1). The average consumption of SAN per week decreased from 4.5 ± 3.9 at baseline to 1.9 ± 2.5 at M1 (p < 0.001) and to 0.7 ± 1.3 at M3 (p < 0.001) (Fig. 2). Additionally, there was a shift toward a better functional status (Fig. 3). CCS classification of patients improved from 83% classified in CCS grades II or III and 17% in CCS grade I at baseline to 68% classified in CCS grade I and only 32% remaining in CCS grades II or III at study end.
The proportion of angina-free patients who did not report angina was 26% at M1 and further increased to 54% at M3.
At the global assessment performed during the last visit, overall effectiveness was rated by physicians as “very good” (69%), “good” (30%) or “moderate” (1%), and overall tolerability of TMZ 80 mg od was rated by physicians as “very good” (75%), “good” (25%) or “moderate” (< 1%).
Physical activity, as self-assessed by patients, was improved (Fig. 4), as evidenced by an increase at M1 in the proportion of patients who reported no limitation (5% at M1 vs. 1% at baseline, p < 0.001), slight limitation (21% at M1 vs. 5% at baseline, p < 0.001) or moderate limitation (43% at M1 vs. 23% at baseline, p < 0.001) and by a decrease in the proportion of patients who reported substantial limitation (29% at M1 vs. 52% at baseline, p < 0.001) or very marked reduction (2% at M1 vs. 19% at baseline, p < 0.0001). At M3, we observed a further increase in the proportion of patients who reported no limitation (15% at M3 vs. 1% at baseline, p < 0.01) and in those with slight limitation (46% at M3 vs. 5% at baseline, p < 0.001) and a further decrease in the proportion of patients who reported substantial limitation (8% at M3 vs. 52% at baseline, p < 0.001) or very marked reduction (1% at M3 vs. 19% at baseline, p < 0.01). The proportion of those with moderate limitation was still significantly higher than at baseline (30% at M3 vs. 23% at baseline, p < 0.01).
Adherence to all anti-anginal medications was also improved (Fig. 5). Good adherence was reported by 56% of patients at M3 vs. 24% at baseline (p < 0.0001), moderate adherence by 41% at M3 vs. 40% at baseline and non-adherence by 3% at M3 vs. 36% at baseline (p < 0.0001). Patient satisfaction with TMZ 80 mg od at M3 was 9.5 [scale from 1 (not satisfied) to 10 (very satisfied)].
Eleven suspected adverse drug reactions were reported in nine patients (0.3%); none of these were classified as severe. The most common were nausea (5 episodes in 3 patients) and dizziness (3 episodes in 2 patients). Two patients reported headache. None of the patients reported parkinsonian symptoms or other movement disorders or any cardiac adverse effects. There were no serious adverse events and no deaths.
A total of 3066 patients completed the study per the protocol; 13 patients (0.42%) discontinued treatment because of patient decision without explanation (0.3%), hospitalization for percutaneous coronary intervention (PCI) procedure (0.1%) and hospitalization for other causes (0.1%).
Discussion
The present study was the first to assess the use of a new formulation of TMZ in a real-world setting. Addition of TMZ 80 mg od to antianginal treatment was found to significantly reduce the frequency of angina attacks and short-acting nitrate consumption, while overall tolerability and effectiveness were rated favorably by the vast majority of physicians (“very good” by 75%, “good” by 25% for tolerability; “very good” by 69%, “good” by 30% for effectiveness). Moreover, improvement was observed in self-reported patient daily activity and adherence to treatment.
TMZ is a well-established therapy for angina control, whose mechanism of action is distinct from that of other antianginal agents, as it directly targets the myocardial cell [14] optimizing cellular energetics particularly in ischemic conditions. As a result of its unique mechanism of action, TMZ is an important antianginal therapy regardless of the background medications. Numerous controlled clinical trials have assessed the efficacy of TMZ as both monotherapy and part of a combination regimen in the treatment of angina, including a total of 3985 patients with chronic stable angina treated with TMZ 20 mg tid or TMZ MR 35 mg bid for a duration ranging from 2 weeks to 6 months. In monotherapy, TMZ reduced angina attacks and SAN consumption versus placebo significantly [15]. It has shown similar efficacy to conventional antianginal drugs in comparative studies with diltiazem [16] or propranolol [17]. The use of TMZ as a part of a combination strategy is an attractive option in patients whose symptom control is suboptimal. The Trimetazidine in Angina Combination Therapy (TACT) study, a randomized, multicenter, placebo-controlled study, showed that the addition of TMZ to BB or long-acting nitrates for 12 weeks resulted in a significant reduction in angina attacks and SAN consumption and in a significant increase in exercise test duration, time to 1-mm ST-segment depression and time to onset of angina pain [18]. The double-blind, placebo-controlled study TRIMetazidine in POLand (TRIMPOL) II showed that the addition of TMZ to metoprolol over a 12-week period significantly improved exercise test parameters and reduced the number of angina attacks and SAN use [19].
In addition, the data from monotherapy and combination therapy trials are supported by meta-analyses [20, 21]. In the largest meta-analysis, conducted by Danchin et al. [21], which evaluated 218 trials with a total of 19,028 patients, TMZ significantly improved exercise tolerance, weekly angina episodes and use of SAN compared with placebo. This efficacy was comparable to that of other antianginal treatments in patients with stable angina pectoris. All these data provide evidence that TMZ is an efficacious therapy to improve angina control.
The results of the present study provide additional evidence for the effectiveness of TMZ in patients with angina.
Non-adherence to treatment is a well-known problem in several chronic conditions. It has been reported in patients with stable angina [10] and has even been observed in patients with serious conditions, for example, post-myocardial infarction patients, of whom 18% did not fill their cardiac medications even once in the 4 months after discharge from hospital [22]. Adherence is related to the number of medications and the number of doses per day [23] and is inversely related to the latter [12]. Simplification of the medication regimen could thus provide a way to improve adherence, and the new TMZ 80 mg od formulation could be beneficial in this respect. However, in the present study, treatment simplification concerned only a subset of patients, those switching from previous use of TMZ (at a dosage of ≥ 2 doses per day) to TMZ od. Given the fact that TMZ treatment led to a significant reduction in angina and to an improvement in daily activity as perceived by patients, it could be possible that this direct impact of the treatment on their quality of life motivated patients to take their medications and thus contributed to the observed improvement in adherence.
Tolerability of this new formulation in daily practice conditions was very good, in line with data from a recently published randomized clinical trial [24].
Study Limitations
The study has limitations inherent in the nature of its design (open-label, observational), which may have resulted in bias toward overestimating the treatment effect. Another limitation is the lack of a control group, which might have biased the results by overestimating the treatment effect. Medication changes were not assessed throughout the study. Another limitation is the lack of objective assessment of anti-ischemic efficacy and exercise capacity. The tools used to test tolerability and physical activity were not previously validated. Finally, observational trials can lead to an underestimation of adverse events as they were spontaneously reported and not specifically sought.
Conclusions
In this prospective, observational study, over a 3-month period in daily clinical practice, TMZ 80 mg od effectively reduced angina attacks and nitrate consumption and improved patient's daily physical activity in a chronic SA patient cohort.
References
Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2163–96.
Moran AE, Forouzanfar MH, Roth GA, et al. The global burden of ischemic heart disease in 1990 and 2010: the Global Burden of Disease 2010 study. Circulation. 2014;129:1493–501.
Arnold SV, Morrow DA, Lei Y, et al. Economic impact of angina after an acute coronary syndrome: insights from the MERLIN-TIMI 36 trial. Circ Cardiovasc Qual Outcomes. 2009;2:344–53.
Montalescot G, Sechtem U, Achenbach S, et al. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013;34:2949–3003.
Beltrame JF, Weekes AJ, Morgan C, Tavella R, Spertus JA. The prevalence of weekly angina among patients with chronic stable angina in primary care practices: The Coronary Artery Disease in General Practice (CADENCE) Study. Arch Intern Med. 2009;169:1491–9.
Kureshi F, Shafiq A, Arnold SV, et al. The prevalence and management of angina among patients with chronic coronary artery disease across US outpatient cardiology practices: insights from the Angina Prevalence and Provider Evaluation of Angina Relief (APPEAR) study. Clin Cardiol. 2017;40:6–10.
Qintar M, Spertus JA, Gosch KL, et al. Effect of angina under-recognition on treatment in outpatients with stable ischemic heart disease. Eur Heart J Qual Care Clin Outcomes. 2016;2:208–14.
Shafiq A, Arnold S, Gosch K, et al. Patient and physician discordance in reporting symptoms of angina among stable coronary artery disease patients: Insights from the Angina Prevalence and Provider Evaluation of Angina Relief (APPEAR) study. Am Heart J. 2016;175:94–100.
Kronish I, Ye S. Adherence to cardiovascular medication: lessons learned and future direction. Prog Cardiovasc Dis. 2013;55:590–600.
Kardas P. on behalf of COMPASS investigators. Comparison of once daily versus twice daily oral nitrates in stable angina pectoris. Am J Cardiol. 2004;94:213–6.
Ferdinand KC, Senatore FF, Clayton-Jeter H, et al. Improving medication adherence in cardiometabolic disease: practical and regulatory implications. J Am Coll Cardiol. 2017;69:437–51.
Claxton AJ, Cramer J, Pierce C. A systematic review of the associations between dose regimens and medication compliance. Clin Ther. 2001;23:1296–310.
Girerd X, Radauceanu A, Achard JM, et al. Evaluation of patient compliance among hypertensive patients treated by specialists. Arch Mal Coeur Vaiss. 2001;94:839–42 (Article in French).
Fragasso G, Perseghin G, De Cobelli F, et al. Effects of metabolic modulation by trimetazidine on left ventricular function and phosphocreatine/adenosine triphosphate ratio in patients with heart failure. Eur Heart J. 2006;27:942–8.
Passeron J. Effectiveness of trimetazidine in stable effort angina due to chronic coronary insufficiency. A double-blind versus placebo study. Presse Med. 1986;15:1775–8.
Koylan N, Bilge AK, Adalet K, Mercanoglu F, Buyukozturk K. Comparison of the effects of trimetazidine and diltiazem on exercise performance in patients with coronary heart disease. The Turkish trimetazidine study (TTS). Acta Cardiol. 2004;59:644–50.
Detry JM, Sellier P, Pennaforte S, Cokkinos D, Dargie H, Mathes P. Trimetazidine: a new concept in the treatment of angina. Comparison with propranolol in patients with stable angina. Trimetazidine European Multicenter Study Group. Br J Clin Pharmacol. 1994;37:279–88.
Chazov EI, Lepakchin VK, Zharova EA, et al. Trimetazidine in Angina Combination Therapy—the TACT study: trimetazidine versus conventional treatment in patients with stable angina pectoris in a randomized, placebo-controlled, multicenter study. Am J Ther. 2005;12:35–42.
Szwed H, Sadowski Z, Elikowski W, et al. Combination treatment in stable effort angina using trimetazidine and metoprolol: results of a randomized, doubleblind, multicentre study (TRIMPOL II). TRIMetazidine in POLand. Eur Heart J. 2001;22:2267–74.
Ciapponi A, Pizarro R, Harrison J. Trimetazidine for stable angina. Cochrane Database Syst Rev. 2005;4: CD003614.
Danchin N, Marzilli M, Parkhomenko A, Ribeiro JP. Efficacy comparison of trimetazidine with therapeutic alternatives in stable angina pectoris: a network meta-analysis. Cardiology. 2011;120:59–72.
Jackevicius CA, Li P, Tu JV. Prevalence, predictors, and outcomes of primary nonadherence after acute myocardial infarction. Circulation. 2008;117:1028–36.
Cramer JA. Effect of partial compliance on cardiovascular medication effectiveness. Heart. 2002;88:203–6.
Pozdnyakov YM; study investigators. Clinical acceptability of trimetazidine modified-release 80 mg once daily versus trimetazidine modified-release 35 mg twice daily in stable angina pectoris. Cardiol Ther. 2018;7:61–70.
Acknowledgements
The authors would like to thank the study participants.
Funding
Sponsorship for this study was provided by Servier, Moscow, Russian Federation. Editorial assistance, article processing charges and open access fee were funded by Servier, France. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing and Editorial Assistance
Writing and editorial assistance was provided by Dr. Diana Toli of Servier.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole and have given their approval for this version to be published.
Disclosures
Maria G. Glezer, scientific coordinator of this study, received honoraria for lectures from Servier, Moscow, Russian Federation. Vladimir A. Vygodin has nothing to disclose.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Data Availability
The data sets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
ODA Study Investigators
Nekrasova E. L. Khlopina, I. A., Nubret K. S., Agafonova Y. A., Ternavsky S. P., (Arkhangelsk), Bardukova O. V., Kutyakova N. M. Berestennikova Y.V., Ustyugova, V. V., Filatova T. A., Khalturina I. G., (Barnaul), Ishchenko, K. A., Karutskaja O. A., Panikar, V. I., Bolkhovitina O. A., Bondarev E. I., Eremin, I. N., (Belgorod), Kadashova S. I., Frolova O. M., Petukhova I. L., (Bryansk), Suvorov A. V., Zhuchkova Y.V., (Velikiy Novgorod), Kuhl M. A., Rodionova L. V., Musurok T. P., (Vladivostok), Ivkova I. A., Ermakov A.S., Stukalova E. M., (Vladimir), Chumakov V. N., Rubezhanskaja E.Y., Kotlova A. A., Pushkareva A. V., Dmitrieva L. V., Bel’kina E. V., Solov’eva O. E., Panyulaytenene S. L., Sarkisyan O. G., (Volgograd), Konovalova G. V., Kuznetsova S. S., Nikolaeva S. V., (Vologda), Trubitsyna I. V., Alekseeva O. A., Kozmina M. E., Babautseva N. A. Kochetkova I. V., Anokhin A. A., Vovk E. V. (Voronezh), Buyko, A. N., Malahatka E. S., Iskra M. A., Shilonosov S. N., Moiseenko G. V., Tomm, Y. S., Stopyatuk N. A. Grigorieva E. G. (Yekaterinburg), Zaitseva A. A., Zaitseva N. A. Sirotkina I. A. (Ivanovo), Kiyanichenko, R. L., Grigoriev S. P., Medvedeva N. V. (Izhevsk), Svatkov M. Y., Golubeva L. V., Kozhevnikova L. A., Chernova A. A., Dolbilkin A. Y., Palvinskaya A.Y., Baranova T. V., Habeeva T. S., Khodus D. A., Ermolaeva G. N., Arbatskaja I. V. (Irkutsk), Idiatullina V. R., Egorova M. E, Galimova I. R., Kuklina M. V., Mamatadzhiev A. A., Rahmatullina R. N., Kim Z. F., Stepanova V. S. (Kazan), Gerr I. E., Levchenko G. I., (Kaliningrad), Dubinkina S. E., Zoob R. A., Krokhmal, O. N., (Kaluga), Lisina S. V., Kudryavtseva N. G., Sitnikov D. V., Lunegov D. G., Evdokimova N. A. Ivanova I. V., (Kemerovo), Manilova A.V., Bessergeneva O. L., Mokhova E. E., (Kirov), Kulchytska O. V., Shulikina O. V., Ananenko V. G., Ivanova O. T. (Krasnodar), Alentyeva M. D., Didur M. E., Zemzyulina K. A., Kartina N. P. Kapustina I. A., Skurikhina N. N., Novoselova G. I., Gracheva T. A., Shiryaev I. V., Avdeeva, O. V., Fedorova O. S., (Krasnoyarsk), Filatkina E. A., Pankova N. V. Mikhailova A.V., (Kursk), Samoilenko T. V. Zhurkina N. V. Riazantseva E. N., (Lipetsk), Mokrousova, L. V., Kulieva A. Z., Rychkova, T. N., (Moscow), Skripka T. A., Krasnevich S. Y., Fedorova L. F., Abdalova S. A., Tremaskina M. S., Mushaeva Z. N., Sladkova T. A., Gabisova P. S., Kononova T. V., Maslova V. V., Kazakova A. N., Khamitsaeva E. O., Ratnikova O. S., Ivanova N. A. Savel’eva E. V., Obikhodova I. V., Puakseva G. I., Gunin, A. A., Skvortsova I. Y., Chugunova I. I., Shestopalova N. A. Kutina, O. A., Ponomareva Y. V., Nikolina T. P., Sokolova S. I., Fastovshchuk T. N., Pak, L. I., Kazaryan A.S. Sabitova O. V., Kuznetsova T. V., Kudzaeva Z. K., Pomiluyko A. A., Kudryavtsev A. V., Gabaraeva T. M., Titova, E. A., Levtsova E. A., Faiman, N.I., Chernomorets V. V., Evseeva O. A., Lopata N.S. Lobankova L. A., Komarova O. V., Tsybulnikova E. M., Kutz, O. A., Kalimeeva O. S., Nudikova I. G., Chibrikina M. V., Sarkisova K. A., Kalakova R. V., Erezeeva N. V. Shirganova G. S., Chmil A. A., Kafanova M. S., Ermolaeva S. V., Koshrokova R. A., Orozalieva G. M., Barnashova E. S., Akhadov S. V., Podmoskova E. V., (Moscow and Moscow region), Borisova A. B., Chirkunova E. V., Chernova, T. A., (Naberezhnye Chelny), Kosova N. V. Guseva O. V., Kuzovkova, S. A., Mulin, I. V., Larina O. V., Mingaleva, S. V., Tsvetkova L. A., Vysotskaja I. K., Degtereva, T. Y., (Nizhny Novgorod), Reshetova L. I., Koblyakova I. I. Chigisova A. N., (Novokuznetsk), Krivenko L. A., Matveeva M. A., Kozlova L. A., Yakhontova O. O., Musina G. R., Kuznetsova E. A. (Novosibirsk), Adamov V. I., Podolnaya, S. P., Mukhin A. V., Giley I. A., Savelyeva N. M. (Omsk), Novikova A. V., Makhonina Y. N., Shemetova T. S., (Orel), Tarasova L. V., Galushko I. V., Nasakina L. I., (Orenburg), Chaadaeva, M. I., Grechishkina O. A., Hamrina G. A., (Penza), Oshchepkova O. V., Senina S. D., Graf, E. B., Kozhevnikova I. M., Lukina S. F., Vinokurova E. V., Sobol A. T., Nevolina N. K., Zdankovich O. T., (Perm), Luzhnova Z. N., Mamedova A. A., (Pskov), Osipova V. A., Zheltova V. L., Shahramanyan L. N., (Pyatigorsk), Bulygina E. D., Panchenko N. N., Tolstokoneva T. P., Batalo G. V., Mazrukho M. K., Chepurnenko S. A. (Rostov-on-don), Alexandrova N. I. Starostina N. N., Grushetskaya, I. S. (Ryazan), Novikova O. Y., Krylova O. A., Arhipova L. V., Kostomarova S. V., Khudyakova O. V., Nasyrova M. A., Yakimova N. N., Kositsyna G. V., Ovchinnikova, E. A., Ryazanova E. I., Kuzmin V. P., (Samara), Basharin A. D., Bulycheva L. V., Soesheva A. R., Timaeva G. R., Burenina N. K., Nikolov N. I. Udaltsova I. V., Zhenihova Z. M., Pukhova E. S. And Shmeleva E. A., Luchko A. I., Ermolenko A. M., Melekhova E. Y., Kadyseva Y. V., Nechvolod E. V., Al-Atar, E. M., Sereda E. J., Grigorieva N. A. Algina, V. G., Borisov A. E., Sabolova Z. S., Novozhenina A. Y., Silantyeva L. A., Litsis N. N., (Saint Petersburg), Gamayunova N. V., Velikanova E. E., Gulyaeva N. Y., Zhuravleva O. A., Chichvarina E.G., Kalinina E.G., Bidzhieva O. S., Vasilieva N. V. Novokreschenova N. V. (Saratov), Osmanova G. R., Lugovtsova S. L., Gadomskaya E. N., (Sevastopol), Pastukhova E. A., Krapivina L. V., Kireeva I. B., (Simferopol), Mironova E. E.B., Nikiforova O. G., Vorfolomeeva A. E., (Smolensk), Orekhova E. I., Evdokimova O. K., (Sochi), Naumova E. A., Shtets A. V., Novikova M. V., Moiseeva L. F., Larina O. I., Trusilkina V. N., (Stavropol), Arinushkina E. A., Manokhin P. V., Sotnikova, T. A., (Stary Oskol), Khazova M. P., Gubareva E. P., Gladysheva Y. I. (Tambov), Khalikova M. D., Bochkareva O. S., Andreeva O. V., (Tver), Dmitrieva N. V. Maklakova M. P., Kapustina L. A., (Tolyatti), Kuzmina I. Y., Mzhelskaya A. A., Demenko, L. P., (Tomsk), Yunusova K.I. Strunevskaya Y. V., Tkacheva O. M., (Tula), Dehont L. Y., Kulikova Y. I., Nadtochiy, T. A., (Tyumen), Harrasova G. Z., Ueldanov U. R., Akhmametyeva Y. N., Badykova E. A., Zainitdinova D. R., Timerbulatov T. R., Zaerova L. M., Nurlygayanova A. M., Vardikyan A. G. (Ufa), Zhukova M. V., Zhuravleva E.G., Sabanina E. N., (Khabarovsk), Pogodina, T. N., Cherepanova N. A., Zakhezina L. V., Petrovф V. G., Charykova I. V. (Chelyabinsk), Chinkova V. A., Kuritsyna A. A., Kos’kina A. A. (Yaroslavl).
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Enhanced digital features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.6833636.
The members of ODA investigators are listed in acknowledgements.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Glezer, M.G., Vygodin, V.A. & on behalf of ODA investigators. Anti-Anginal Effectiveness and Tolerability of Trimetazidine Modified Release 80 Mg Once Daily in Stable Angina Patients in Real-World Practice. Adv Ther 35, 1368–1377 (2018). https://doi.org/10.1007/s12325-018-0756-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-018-0756-3