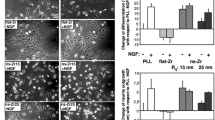
Abstract
In this study, we report that height-controlled vertically etched silicon nano-column arrays (vSNAs) induce strong growth cone-to-substrate coupling and accelerate In vitroneurite development while preserving the essential features of initial neurite formation. Large-scale preparation of vSNAs with flat head morphology enabled the generation of well-controlled topographical stimulation without cellular impalement. A systematic analysis on topography-induced variations on cellular morphology and cytoskeletal dynamics was conducted. In addition, neurite development on the grid-patterned vSNAs exhibited preferential adhesion to the nanostructured region and outgrowth directionality. The arrangement of cytoskeletal proteins and the expression of a focal adhesion complex indicated that a strong coupling existed between the underlying nanocolumns and growth cones. Furthermore, the height-controlled nanocolumn substrates differentially modulated neurite polarization and elongation. Our findings provide an important insight into neuron-nanotopography interactions and their role in cell adhesion and neurite development.

Similar content being viewed by others
References
Dent, E. W.; Gertler, F. B. Cytoskeletal dynamics and transport in growth cone motility and axon guidance. Neuron 2003, 40, 209–227.
Geraldo, S.; Gordon-Weeks, P. R. Cytoskeletal dynamics in growth-cone steering. J. Cell Sci. 2009, 122, 3595–3604.
Schaar, B. T.; McConnell, S. K. Cytoskeletal coordination during neuronal migration. Proc. Natl. Acad. Sci. USA 2005, 102, 13652–13657.
Turney, S. G.; Bridgman, P. C. Laminin stimulates and guides axonal outgrowth via growth cone myosin II activity. Nat. Neurosci. 2005, 8, 717–719.
Gomez, N.; Chen, S. C.; Schmidt, C. E. Polarization of hippocampal neurons with competitive surface stimuli: Contact guidance cues are preferred over chemical ligands. J. R. Soc. Interface 2007, 4, 223–233.
San Miguel-Ruiz, J. E.; Letourneau, P. C. The role of Arp2/3 in growth cone actin dynamics and guidance is substrate depe-ndent. J. Neurosci. 2014, 34, 5895–5908.
Kerstein, P. C.; Nichol IV, R. H.; Gomez, T. M. Mechan-ochemical regulation of growth cone motility. Front. Cell. Neurosci. 2015, 9, 244.
Kulangara, K.; Leong, K. W. Substrate topography shapes cell function. Soft Matter 2009, 5, 4072–4076.
Teixeira, A. I.; Abrams, G. A.; Bertics, P. J.; Murphy, C. J.; Nealey, P. F. Epithelial contact guidance on well-defined micro- and nanostructured substrates. J. Cell Sci. 2003, 116, 1881–1892.
Teixeira, A. I.; McKie, G. A.; Foley, J. D.; Bertics, P. J.; Nealey, P. F.; Murphy, C. J. The effect of environmental factors on the response of human corneal epithelial cells to nanoscale substrate topography. Biomaterials 2006, 27, 3945–3954.
Yim, E. K. F.; Reano, R. M.; Pang, S. W.; Yee, A. F.; Chen, C. S.; Leong, K. W. Nanopattern-induced changes in morphology and motility of smooth muscle cells. Biomaterials 2005, 26, 5405–5413.
Yim, E. K. F.; Darling, E. M.; Kulangara, K.; Guilak, F.; Leong, K. W. Nanotopography-induced changes in focal adhesions, cy-toskeletal organization, and mechanical properties of human mesenchymal stem cells. Biomaterials 2010, 31, 1299–1306.
Watari, S.; Hayashi, K.; Wood, J. A.; Russell, P.; Nealey, P. F.; Murphy, C. J.; Genetos, D. C. Modulation of osteogenic diff-erentiation in hMSCs cells by submicron topographically-patt-erned ridges and grooves. Biomaterials 2012, 33, 128–136.
Foley, J. D.; Grunwald, E. W.; Nealey, P. F.; Murphy, C. J. Cooperative modulation of neuritogenesis by PC12 cells by topography and nerve growth factor. Biomaterials 2005, 26, 3639–3644.
Ferrari, A.; Cecchini, M.; Dhawan, A.; Micera, S.; Tonazzini, I.; Stabile, R.; Pisignano, D.; Beltram, F. Nanotopographic control of neuronal polarity. Nano Lett. 2011, 11, 505–511.
Xie, J. W.; MacEwan, M. R.; Li, X. R.; Sakiyama-Elbert, S. E.; Xia, Y. N. Neurite outgrowth on nanofiber scaffolds with different orders, structures, and surface properties. ACS Nano 2009, 3, 1151–1159.
Cho, W. K.; Kang, K.; Kang, G.; Jang, M. J.; Nam, Y.; Choi, I. S. Pitch-dependent acceleration of neurite outgrowth on nanos-tructured anodized aluminum oxide substrates. Angew. Chem., Int. Ed. 2010, 49, 10114–10118.
Kang, K.; Choi, S.-E.; Jang, H. S.; Cho, W. K.; Nam, Y.; Choi, I. S.; Lee, J. S. In vitro developmental acceleration of hipp-ocampal neurons on nanostructures of self-assembled silica be-ads in filopodium-size ranges. Angew. Chem., Int. Ed. 2012, 51, 2855–2858.
Jang, K.-J.; Kim, M. S.; Feltrin, D.; Jeon, N. L.; Suh, K.-Y.; Pertz, O. Two distinct filopodia populations at the growth cone allow to sense nanotopographical extracellular matrix cues to guide neurite outgrowth. PLoS One 2010, 5, e15966.
Kang, K.; Yoon, S. Y.; Choi, S.-E.; Kim, M.-H.; Park, M.; Nam, Y.; Lee, J. S.; Choi, I. S. Cytoskeletal actin dynamics are involved in pitch-dependent neurite outgrowth on bead mono-layers. Angew. Chem., Int. Ed. 2014, 53, 6075–6079.
Liu, X. L.; Wang, S. T. Three-dimensional nano-biointerface as a new platform for guiding cell fate. Chem. Soc. Rev. 2014, 43, 2385–2401.
Kim, W.; Ng, J. K.; Kunitake, M. E.; Conklin, B. R.; Yang, P. Interfacing silicon nanowires with mammalian cells. J. Am. Chem. Soc. 2007, 129, 7228–7229.
Hanson, L.; Lin, Z. C.; Xie, C.; Cui, Y.; Cui, B. X. Chara-cterization of the cell–nanopillar interface by transmission electron microscopy. Nano Lett. 2012, 12, 5815–5820.
Xie, X.; Xu, A. M.; Angle, M. R.; Tayebi, N.; Verma, P.; Melosh, N. A. Mechanical model of vertical nanowire cell penetration. Nano Lett. 2013, 13, 6002–6008.
Xu, A. M.; Aalipour, A.; Leal-Ortiz, S.; Mekhdjian, A. H.; Xie, X.; Dunn, A. R.; Garner, C. C.; Melosh, N. A. Quantification of nanowire penetration into living cells. Nat. Commun. 2014, 5, 3613.
Qi, S.; Yi, C. Q.; Ji, S. L.; Fong, C.-C.; Yang, M. S. Cell adhesion and spreading behavior on vertically aligned silicon nanowire arrays. ACS Appl. Mater. Interfaces 2009, 1, 30–34.
Bonde, S.; Berthing, T.; Madsen, M. H.; Andersen, T. K.; Buch-Månson, N.; Guo, L.; Li, X. M.; Badique, F.; Anselme, K.; Nygård, J. et al. Tuning InAs nanowire density for HEK293 cell viability, adhesion, and morphology: Perspectives for nano-wire-based biosensors. ACS Appl. Mater. Interfaces 2013, 5, 10510–10519.
Padmanabhan, J.; Kinser, E. R.; Stalter, M. A.; Duncan-Lewis, C.; Balestrini, J. L.; Sawyer, A. J.; Schroers, J.; Kyriakides, T. R. Engineering cellular response using nanopatterned bulk metallic glass. ACS Nano 2014, 8, 4366–4375.
Persson, H.; Købler, C.; Mølhave, K.; Samuelson, L.; Teg-enfeldt, J. O.; Oredsson, S.; Prinz, C. N. Fibroblasts cultured on nanowires exhibit low motility, impaired cell division, and DNA damage. Small 2013, 9, 4006–4016.
Persson, H.; Li, Z.; Tegenfeldt, J. O.; Oredsson, S.; Prinz, C. From immobilized cells to motile cells on a bed-of-nails: Effects of vertical nanowire array density on cell behaviour. Sci. Rep. 2015, 5, 18535.
Li, Z.; Song, J. H.; Mantini, G.; Lu, M.-Y.; Fang, H.; Falconi, C; Chen, L.-J.; Wang, Z. L. Quantifying the traction force of a single cell by aligned silicon nanowire array. Nano Lett. 2009, 9, 3575–3580.
Albuschies, J.; Vogel, V. The role of filopodia in the recognition of nanotopographies. Sci. Rep. 2013, 3, 1658.
Kim, H.; Kim, I.; Choi, H.-J.; Kim, S. Y.; Yang, E. G. Neuron-like differentiation of mesenchymal stem cells on silicon nanowires. Nanoscale 2015, 7, 17131–17138.
Shalek, A. K.; Robinson, J. T.; Karp, E. S.; Lee, J. S.; Ahn, D.R.; Yoon, M.H.; Sutton, A.; Jorgolli, M.; Gertner, R. S.; Gujral, T. S. et al. Vertical silicon nanowires as a universal platform for delivering biomolecules into living cells. Proc. Natl. Acad. Sci. USA 2010, 107, 1870–1875.
Shalek, A. K.; Gaublomme, J. T.; Wang, L. L.; Yosef, N.; Chevrier, N.; Andersen, M. S.; Robinson, J. T.; Pochet, N.; Neuberg, D.; Gertner, R. S. et al. Nanowire-mediated delivery enables functional interrogation of primary immune cells: Application to the analysis of chronic lymphocytic leukemia. Nano Lett. 2012, 12, 6498–6504.
Hällström, W.; Mårtensson, T.; Prinz, C.; Gustavsson, P.; Montelius, L.; Samuelson, L.; Kanje, M. Gallium phosphide nanowires as a substrate for cultured neurons. Nano Lett. 2007, 7, 2960–2965.
Prinz, C.; Hällström, W.; Mårtensson, T.; Samuelson, L.; Montelius, L.; Kanje, M. Axonal guidance on patterned free-st-anding nanowire surfaces. Nanotechnology 2008, 19, 345101.
Piret, G.; Perez, M.-T.; Prinz, C. N. Support of neuronal growth over glial growth and guidance of optic nerve axons by vertical nanowire arrays. ACS Appl. Mater. Interfaces 2015, 7, 18944–18948.
Xie, C.; Hanson, L.; Xie, W. J.; Lin, Z. L.; Cui, B. X.; Cui, Y. Noninvasive neuron pinning with nanopillar arrays. Nano Lett. 2010, 10, 4020–4024.
Kang, K.; Park, Y.S.; Park, M.; Jang, M. J.; Kim, S.M.; Lee, J.; Choi, J. Y.; Jung, D. H.; Chang, Y.T.; Yoon, M.H. et al. Axon-first neuritogenesis on vertical nanowires. Nano Lett. 2016, 16, 675–680.
Wierzbicki, R.; Købler, C.; Jensen, M. R. B.; Lopacinska, J.; Schmidt, M. S.; Skolimowski, M.; Abeille, F.; Qvortrup, K.; Mølhave, K. Mapping the complex morphology of cell interactions with nanowire substrates using FIB-SEM. PLoS One 2013, 8, e53307.
Berthing, T.; Bonde, S.; Rostgaard, K. R.; Madsen, M. H.; Sørensen, C. B.; Nygård, J.; Martinez, K. L. Cell membrane conformation at vertical nanowire array interface revealed by fluorescence imaging. Nanotechnology 2012, 23, 415102.
Bucaro, M. A.; Vasquez, Y.; Hatton, B. D.; Aizenberg, J. Fine-tuning the degree of stem cell polarization and alignment on ordered arrays of high-aspect-ratio nanopillars. ACS Nano 2012, 6, 6222–6230.
Lee, S.; Kim, D.; Kim, S.-M.; Kim, J.-A.; Kim, T.; Kim, D.-Y.; Yoon, M.-H. Polyelectrolyte multilayer-assisted fabrication of non-periodic silicon nanocolumn substrates for cellular interface applications. Nanoscale 2015, 7, 14627–14635.
Fath, T.; Ke, Y. D.; Gunning, P.; Götz, J.; Ittner, L. M. Primary support cultures of hippocampal and substantia nigra neurons. Nat. Protoc. 2008, 4, 78–85.
Yang, K.; Jung, H.; Lee, H.-R.; Lee, J. S.; Kim, S. R.; Song, K. Y.; Cheong, E.; Bang, J.; Im, S. G.; Cho, S.-W. Multiscale, hierarchically patterned topography for directing human neural stem cells into functional neurons. ACS Nano 2014, 8, 7809–7822.
Brunetti, V.; Maiorano, G.; Rizzello, L.; Sorce, B.; Sabella, S.; Cingolani, R.; Pompa, P. P. Neurons sense nanoscale roughness with nanometer sensitivity. Proc. Natl. Acad. Sci. USA 2010, 107, 6264–6269.
Elnathan, R.; Delalat, B.; Brodoceanu, D.; Alhmoud, H.; Harding, F. J.; Buehler, K.; Nelson, A.; Isa, L.; Kraus, T.; Voelcker, N. H. Maximizing transfection efficiency of vertically aligned silicon nanowire arrays. Adv. Funct. Mater. 2015, 25, 7215–7225.
Liu, D. D.; Yi, C. Q.; Wang, K. Q.; Fong, C.-C.; Wang, Z. K.; Lo, P. K.; Sun, D.; Yang, M. S. Reorganization of cytoskeleton and transient activation of Ca2+ channels in mesenchymal stem cells cultured on silicon nanowire arrays. ACS Appl. Mater. Interfaces 2013, 5, 13295–13304.
Kuo, S.W.; Lin, H.I.; Hui-Chun Ho, J.; Shih, Y.-R. V.; Chen, H.F.; Yen, T.J.; Lee, O. K. Regulation of the fate of human mesenchymal stem cells by mechanical and stereo-topog-raphical cues provided by silicon nanowires. Biomaterials 2012, 33, 5013–5022.
Nair, B. G.; Hagiwara, K.; Ueda, M.; Yu, H. H.; Tseng, H.-R.; Ito, Y. High density of aligned nanowire treated with polydopamine for efficient gene silencing by siRNA according to cell membrane perturbation. ACS Appl. Mater. Interfaces 2016, 8, 18693–18700.
Bugnicourt, G.; Brocard, J.; Nicolas, A.; Villard, C. Nanoscale surface topography reshapes neuronal growth in culture. Langmuir 2014, 30, 4441–4449.
Lowery, L. A.; van Vactor, D. The trip of the tip: Understanding the growth cone machinery. Nat. Rev. Mol. Cell Biol. 2009, 10, 332–343.
Vitriol, E. A.; Zheng, J. Q. Growth cone travel in space and time: The cellular ensemble of cytoskeleton, adhesion, and membrane. Neuron 2012, 73, 1068–1081.
Geiger, B.; Spatz, J. P.; Bershadsky, A. D. Environmental sensing through focal adhesions. Nat. Rev. Mol. Cell Biol. 2009, 10, 21–33.
Koch, D.; Rosoff, W. J.; Jiang, J. J.; Geller, H. M.; Urbach, J. S. Strength in the periphery: Growth cone biomechanics and substrate rigidity response in peripheral and central nervous system neurons. Biophys. J. 2012, 102, 452–460.
Gilles, S.; Winter, S.; Michael, K. E.; Meffert, S. H.; Li, P. G.; Greben, K.; Simon, U.; Offenhäusser, A.; Mayer, D. Control of cell adhesion and neurite outgrowth by patterned gold nanoparticles with tunable attractive or repulsive surface properties. Small 2012, 8, 3357–3367.
Lamoureux, P.; Ruthel, G.; Buxbaum, R. E.; Heidemann, S. R. Mechanical tension can specify axonal fate in hippocampal neurons. J. Cell Biol. 2002, 159, 499–508.
Bard, L.; Boscher, C.; Lambert, M.; Mège, R.-M.; Choquet, D.; Thoumine, O. A molecular clutch between the actin flow and N-cadherin adhesions drives growth cone migration. J. Neurosci. 2008, 28, 5879–5890.
Athamneh, A. I. M.; Suter, D. M. Quantifying mechanical force in axonal growth and guidance. Front. Cell. Neurosci. 2015, 9, 359.
Suter, D. M.; Miller, K. E. The emerging role of forces in axonal elongation. Prog. Neurobiol. 2011, 94, 91–101.
Chazeau, A.; Garcia, M.; Czöndör, K.; Perrais, D.; Tessier, B.; Giannone, G.; Thoumine, O. Mechanical coupling between transsynaptic N-cadherin adhesions and actin flow stabilizes dendritic spines. Mol. Biol. Cell 2015, 26, 859–873.
Acknowledgements
This work was supported by the Basic Science Research Program and the Pioneer Research Center Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (Nos. NRF-2013R1A1A107-6103, NRF-2012R1A3A2026403, and NRF-2012-000-9664), and also by the GIST Research Institute (GRI) in 2016.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kim, SM., Lee, S., Kim, D. et al. Strong contact coupling of neuronal growth cones with height-controlled vertical silicon nanocolumns. Nano Res. 11, 2532–2543 (2018). https://doi.org/10.1007/s12274-017-1878-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-017-1878-7