Abstract
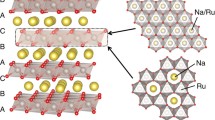
The development of new sodium ion battery (SIB) cathodes with satisfactory performance requires an in-depth understanding of their structure−function relationships, to rationally design better electrode materials. In this work, highly ordered, honeycomb-layered Na3Ni2SbO6 was prepared to elucidate the structural evolution and Na+ kinetics during electrochemical desodiation/sodiation processes. Structural analysis involving in situ synchrotron X-ray diffraction (XRD) experiments, electrochemical performance measurements, and electrochemical characterization (galvanostatic intermittent titration technique, GITT) methods were used to obtain new insights into the reaction mechanism controlling the (de)intercalation of sodium into the host Na3−xNi2SbO6 structure. Two phase transitions occur (initial O′3 phase → intermediate P′3 phase → final O1 phase) upon Na+ extraction; the partial irreversible O′3-P′3 phase transition is responsible for the insufficient cycling stability. The fast Na+ mobility (average 10–12 cm2·s–1) in the interlayer, high equilibrium voltage (3.27 V), and low voltage polarization (50 mV) establish the linkage between kinetic advantage and a good rate performance of the cathode. These new findings provide deep insight into the reaction mechanism operating in the honeycomb cathode; the present approach could be also extended to investigate other materials for SIBs.

Similar content being viewed by others
References
Armand, M.; Tarascon, J. M. Building better batteries. Nature 2008, 451, 652–657.
Yabuuchi, N.; Kubota, K.; Dahbi, M.; Komaba, S. Research development on sodium-ion batteries. Chem. Rev. 2014, 114, 11636–11682.
Zhao, F. P.; Gong, Q. F.; Traynor, B.; Zhang, D.; Li, J. J.; Ye, H. L.; Chen, F. J.; Han, N.; Wang, Y. Y.; Sun, X. H. et al. Stabilizing nickel sulfide nanoparticles with an ultrathin carbon layer for improved cycling performance in sodium ion batteries. Nano Res. 2016, 9, 3162–3170.
Kim, S. W.; Seo, D. H.; Ma, X. H.; Ceder, G.; Kang, K. Electrode materials for rechargeable sodium-ion batteries: Potential alternatives to current lithium-ion batteries. Adv. Energy Mater. 2012, 2, 710–721.
Barpanda, P.; Ati, M.; Melot, B. C.; Rousse, G.; Chotard, J. N.; Doublet, M. L.; Sougrati, M. T.; Corr, S. A.; Jumas, J. C.; Tarascon, J. M. A 3.90 V iron-based fluorosulphate material for lithium-ion batteries crystallizing in the triplite structure. Nat. Mater. 2011, 10, 772–779.
Sun, Y. K.; Chen, Z. H.; Noh, H. J.; Lee, D. J.; Jung, H. G.; Ren, Y.; Wang, S.; Yoon, C. S.; Myung, S. T.; Amine, K. Nanostructured high-energy cathode materials for advanced lithium batteries. Nat. Mater. 2012, 11, 942–947.
Xin, S.; Chang, Z. W.; Zhang, X. B.; Guo, Y. G. Progress of rechargeable lithium metal batteries based on conversion reactions. Natl. Sci. Rev. 2017, 4, 54–70.
Wei, Q. L.; Xiong, F. Y.; Tan, S. S.; Huang, L.; Lan, E. H.; Dunn, B.; Mai, L. Q. Porous one-dimensional nanomaterials: Design, fabrication and applications in electrochemical energy storage. Adv. Mater. 2017, 29, 1602300.
Xiang, X. D.; Zhang, K.; Chen, J. Recent advances and prospects of cathode materials for sodium-ion batteries. Adv. Mater. 2015, 27, 5343–5364.
Lyu, Z. Y.; Yang, L. J.; Xu, D.; Zhao, J.; Lai, H. W.; Jiang, Y. F.; Wu, Q.; Li, Y.; Wang, X. Z.; Hu, Z. Hierarchical carbon nanocages as high-rate anodes for Li- and Na-ion batteries. Nano Res. 2015, 8, 3535–3543.
Kim, H.; Kim, H.; Ding, Z.; Lee, M. H.; Lim, K.; Yoon, G.; Kang, K. Recent progress in electrode materials for sodium-ion batteries. Adv. Energy Mater. 2016, 6, 1600943.
Han, M. H.; Gonzalo, E.; Singh, G.; Rojo, T. A comprehensive review of sodium layered oxides: Powerful cathodes for Na-ion batteries. Energy Environ. Sci. 2015, 8, 81–102.
Deng, X.; Liu, X. R.; Yan, H. J.; Wang, D.; Wan, L. J. Morphology and modulus evolution of graphite anode in lithium ion battery: An in situ AFM investigation. Sci. China Chem. 2014, 57, 178–183.
You, Y.; Yu, X. Q.; Yin, Y. X.; Nam, K. W.; Guo, Y. G. Sodium iron hexacyanoferrate with high Na content as a Na-rich cathode material for Na-ion batteries. Nano Res. 2015, 8, 117–128.
Wang, X. P.; Hu, P.; Niu, C. J.; Meng, J. S.; Xu, X. M.; Wei, X. J.; Tang, C. J.; Luo, W.; Zhou, L.; An, Q. Y. et al. New-type K0.7Fe0.5Mn0.5O2 cathode with an expanded and stabilized interlayer structure for high-capacity sodium-ion batteries. Nano Energy 2017, 35, 71–78.
Boucher, F.; Gaubicher, J.; Cuisinier, M.; Guyomard, D.; Moreau, P. Elucidation of the Na2/3FePO4 and Li2/3FePO4 intermediate superstructure revealing a pseudouniform ordering in 2D. J. Am. Chem. Soc. 2014, 136, 9144–9157.
Guignard, M.; Didier, C.; Darriet, J.; Bordet, P.; Elkaim, E.; Delmas, C. P2-NaxVO2 system as electrodes for batteries and electron-correlated materials. Nat. Mater. 2013, 12, 74–80.
Wang, P. F.; Yao, H. R.; Zuo, T. T.; Yin, Y. X.; Guo, Y. G. Novel P2-type Na2/3Ni1/6Mg1/6Ti2/3O2 as an anode material for sodium-ion batteries. Chem. Commun. 2017, 53, 1957–1960.
Berthelot, R.; Carlier, D.; Delmas, C. Electrochemical investigation of the P2-NaxCoO2 phase diagram. Nat. Mater. 2011, 10, 74–80.
Guo, J. Z.; Wang, P. F.; Wu, X. L.; Zhang, X. H.; Yan, Q. Y.; Chen, H.; Zhang, J. P.; Guo, Y. G. High-energy/power and low-temperature cathode for sodium-ion batteries: In situ XRD study and superior full-cell performance. Adv. Mater. 2017, 29, 1701968.
Wang, P. F.; You, Y.; Yin, Y. X.; Wang, Y. S.; Wan, L. J.; Gu, L.; Guo, Y. G. Suppressing the P2-O2 phase transition of Na0.67Mn0.67Ni0.33O2 by magnesium substitution for improved sodium-ion batteries. Angew. Chem., Int. Ed. 2016, 55, 7445–7449.
Singh, G.; Tapia-Ruiz, N.; Lopez del Amo, J. M.; Maitra, U.; Somerville, J. W.; Armstrong, A. R.; Martinez de Ilarduya, J.; Rojo, T.; Bruce, P. G. High voltage Mg-doped Na0.67Ni0.3–xMgxMn0.7O2 (x = 0.05, 0.1) Na-ion cathodes with enhanced stability and rate capability. Chem. Mater. 2016, 28, 5087–5094.
Wang, P. F.; Yao, H. R.; Liu, X. Y.; Zhang, J. N.; Gu, L.; Yu, X. Q.; Yin, Y. X.; Guo, Y. G. Ti-substituted NaNi0.5Mn0.5-xTixO2 cathodes with Reversible O3-P3 phase transition for high-performance sodium-ion batteries. Adv. Mater. 2017, 29, 1700210.
Xie, Y. Y.; Wang, H.; Xu, G. L.; Wang, J. J.; Sheng, H. P.; Chen, Z. H.; Ren, Y.; Sun, C. J.; Wen, J. G.; Wang, J. et al. In operando XRD and TXM study on the metastable structure change of NaNi1/3Fe1/3Mn1/3O2 under electrochemical sodium-ion intercalation. Adv. Energy Mater. 2016, 6, 1601306.
Wang, P. F.; You, Y.; Yin, Y. X.; Guo, Y. G. An O3-type NaNi0.5Mn0.5O2 cathode for sodium-ion batteries with improved rate performance and cycling stability. J. Mater. Chem. A 2016, 4, 17660–17664.
Delmas, C.; Fouassier, C.; Hagenmuller, P. Structural classification and properties of the layered oxides. Phys. B+C 1980, 99, 81–85.
Vassilaras, P.; Ma, X. H.; Li, X.; Ceder, G. Electrochemical properties of monoclinic NaNiO2. J. Electrochem. Soc. 2012, 160, A207–A211.
Doeff, M. M.; Peng, M. Y.; Ma, Y. P.; De Jonghe, L. C. Orthorhombic NaxMnO2 as a cathode material for secondary sodium and lithium polymer batteries. J. Electrochem. Soc. 1994, 141, L145–L147.
Politaev, V. V.; Nalbandyan, V. B.; Petrenko, A. A.; Shukaev, I. L.; Volotchaev, V. A.; Medvedev, B. S. Mixed oxides of sodium, antimony (5+) and divalent metals (Ni, Co, Zn or Mg). J. Solid State Chem. 2010, 183, 684–691.
Schmidt, W.; Berthelot, R.; Sleight, A. W.; Subramanian, M. A. Solid solution studies of layered honeycomb-ordered phases O3–Na3M2SbO6 (M = Cu, Mg, Ni, Zn). J. Solid State Chem. 2013, 201, 178–185.
Zvereva, E. A.; Stratan, M. I.; Ovchenkov, Y. A.; Nalbandyan, V. B.; Lin, J. Y.; Vavilova, E. L.; Iakovleva, M. F.; Abdel-Hafiez, M.; Silhanek, A. V.; Chen, X. J. et al. Zigzag antiferromagnetic quantum ground state in monoclinic honeycomb lattice antimonates A3Ni2SbO6 (A = Li, Na). Phys. Rev. B 2015, 92, 144401.
Viciu, L.; Huang, Q.; Morosan, E.; Zandbergen, H. W.; Greenbaum, N. I.; McQueen, T.; Cava, R. J. Structure and basic magnetic properties of the honeycomb lattice compounds Na2Co2TeO6 and Na3Co2SbO6. J. Solid State Chem. 2007, 180, 1060–1067.
Politaev, V. V.; Nalbandyan, V. B. Subsolidus phase relations, crystal chemistry and cation-transport properties of sodium iron antimony oxides. Solid State Sci. 2009, 11, 144–150.
Yuan, D. D.; Liang, X. M.; Wu, L.; Cao, Y. L.; Ai, X. P.; Feng, J. W.; Yang, H. X. A honeycomb-layered Na3Ni2SbO6: A high-rate and cycle-stable cathode for sodium-ion batteries. Adv. Mater. 2014, 26, 6301–6306.
Zvereva, E. A.; Evstigneeva, M. A.; Nalbandyan, V. B.; Savelieva, O. A.; Ibragimov, S. A.; Volkova, O. S.; Medvedeva, L. I.; Vasiliev, A. N.; Klingeler, R.; Buechner, B. Monoclinic honeycomb-layered compound Li3Ni2SbO6: Preparation, crystal structure and magnetic properties. Dalton Trans. 2012, 41, 572–580.
Schmidt, W.; Berthelot, R.; Etienne, L.; Wattiaux, A.; Subramanian, M. A. Synthesis and characterization of O3-Na3LiFeSbO6: A new honeycomb ordered layered oxide. Mater. Res. Bull. 2014, 50, 292–296.
Liu, J.; Yin, L.; Wu, L. J.; Bai, J. M.; Bak, S. M.; Yu, X. Q.; Zhu, Y. M.; Yang, X. Q.; Khalifah, P. G. Quantification of honeycomb number-type stacking faults: Application to Na3Ni2BiO6 cathodes for Na-ion batteries. Inorg. Chem. 2016, 55, 8478–8492.
Li, Z. Y.; Gao, R.; Sun, L. M.; Hu, Z. B.; Liu, X. F. Designing an advanced P2-Na0.67Mn0.65Ni0.2Co0.15O2 layered cathode material for Na-ion batteries. J. Mater. Chem. A 2015, 3, 16272–16278.
Han, M. H.; Gonzalo, E.; Casas-Cabanas, M.; Rojo, T. Structural evolution and electrochemistry of monoclinic NaNiO2 upon the first cycling process. J. Power Sources 2014, 258, 266–271.
Wang, X. F.; Liu, G. D.; Iwao, T.; Okubo, M.; Yamada, A. Role of ligand-to-metal charge transfer in O3-type NaFeO2–NaNiO2 solid solution for enhanced electrochemical properties. J. Phys. Chem. C 2014, 118, 2970–2976.
Nanba, Y.; Iwao, T.; de Boisse, B. M.; Zhao, W. W.; Hosono, E.; Asakura, D.; Niwa, H.; Kiuchi, H.; Miyawaki, J.; Harada, Y. et al. Redox potential paradox in NaxMO2 for sodium-ion battery cathodes. Chem. Mater. 2016, 28, 1058–1065.
Bucher, N.; Hartung, S.; Franklin, J. B.; Wise, A. M.; Lim, L. Y.; Chen, H. Y.; Weker, J. N.; Toney, M. F.; Srinivasan, M. P2-NaxCoyMn1-yO2 (y = 0, 0.1) as cathode materials in sodium-ion batteries-effects of doping and morphology to enhance cycling stability. Chem. Mater. 2016, 28, 2041–2051.
Chou, S. L.; Wang, J. Z.; Liu, H. K.; Dou, S. X. Rapid synthesis of Li4Ti5O12 microspheres as anode materials and its binder effect for lithium-ion battery. J. Phys. Chem. C 2011, 115, 16220–16227.
Guo, S. H.; Yu, H. J.; Jian, Z. L.; Liu, P.; Zhu, Y. B.; Guo, X. W.; Chen, M. W.; Ishida, M.; Zhou, H. S. A high-capacity, low-cost layered sodium manganese oxide material as cathode for sodium-ion batteries. ChemSusChem 2014, 7, 2115–2119.
Cui, Z. H.; Guo, X. X.; Li, H. Equilibrium voltage and overpotential variation of nonaqueous Li–O2 batteries using the galvanostatic intermittent titration technique. Energy Environ. Sci. 2015, 8, 182–187.
Acknowledgements
This work was supported by the National Key R&D Program of China (No. 2016YFA0202500), the National Natural Science Foundation of China (NSFC) (Nos. 51772301 and 21773264), and the “Strategic Priority Research Program“ of the Chinese Academy of Sciences (No. XDA09010100) and the Chinese Academy of Sciences (CAS).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, PF., Yao, HR., You, Y. et al. Understanding the structural evolution and Na+ kinetics in honeycomb-ordered O′3-Na3Ni2SbO6 cathodes. Nano Res. 11, 3258–3271 (2018). https://doi.org/10.1007/s12274-017-1863-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-017-1863-1