Abstract
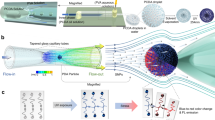
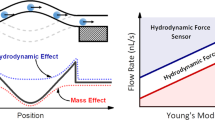
The wall shear stress (WSS) that a moving fluid exerts on a surface affects many processes including those relating to vascular function. WSS plays an important role in normal physiology (e.g. angiogenesis) and affects the microvasculature’s primary function of molecular transport. Points of fluctuating WSS show abnormalities in a number of diseases; however, there is no established technique for measuring WSS directly in physiological systems. All current methods rely on estimates obtained from measured velocity gradients in bulk flow data. In this work, we report a nanosensor that can directly measure WSS in microfluidic chambers with sub-micron spatial resolution by using a specific type of virus, the bacteriophage M13, which has been fluorescently labeled and anchored to a surface. It is demonstrated that the nanosensor can be calibrated and adapted for biological tissue, revealing WSS in micro-domains of cells that cannot be calculated accurately from bulk flow measurements. This method lends itself to a platform applicable to many applications in biology and microfluidics.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Nge, P. N.; Rogers, C. I.; Woolley, A. T. Advances in microfluidic materials, functions, integration, and applications. Chem. Rev. 2013, 113, 2550–2583.
Zarins, C. K.; Giddens, D. P.; Bharadvaj, B. K.; Sottiurai, V. S.; Mabon, R. F.; Glagov, S. Carotid bifurcation atherosclerosis. Quantitative correlation of plaque localization with flow velocity profiles and wall shear-stress. Circ. Res. 1983, 53, 502–514.
Chatzizisis, Y. S.; Coskun, A. U.; Jonas, M.; Edelman, E. R.; Feldman, C. L.; Stone, P. H. Role of endothelial shear stress in the natural history of coronary atherosclerosis and vascular remodeling: Molecular, cellular, and vascular behavior. J. Am. Coll. Cardiol. 2007, 49, 2379–2393.
Katritsis, D.; Kaiktsis, L.; Chaniotis, A.; Pantos, J.; Efstathopoulos, E. P.; Marmarelis, V. Wall shear stress: Theoretical considerations and methods of measurement. Prog. Cardiovasc. Dis. 2007, 49, 307–329.
Reneman, R. S.; Hoeks, A. P. G. Wall shear stress as measured in vivo: Consequences for the design of the arterial system. Med. Biol. Eng. Comput. 2008, 46, 499–507.
Reneman, R. S.; Arts, T.; Hoeks, A. P. G. Wall shear stressan important determinant of endothelial cell function and structure-in the arterial system in vivo. J. Vasc. Res. 2006, 43, 251–269.
Young, E. W. K.; Beebe, D. J. Fundamentals of microfluidic cell culture in controlled microenvironments. Chem. Soc. Rev. 2010, 39, 1036–1048.
Naughton, J. W.; Sheplak, M. Modern developments in shear-stress measurement. Prog. Aerosp. Sci. 2002, 38, 515–570.
Große, S.; Schröder, W. Mean wall-shear stress measurements using the micro-pillar shear-stress sensor MPS3. Meas. Sci. Technol. 2008, 19, 015403.
Brücker, C.; Spatz, J.; Schröder, W. Feasability study of wall shear stress imaging using microstructured surfaces with flexible micropillars. Exp. Fluids. 2005, 39, 464–474.
Smith, M. L.; Long, D. S.; Damiano, E. R.; Ley, K. Nearwall µ-PIV reveals a hydrodynamically relevant endothelial surface layer in venules in vivo. Biophys. J. 2003, 85, 637–645.
Samady, H.; Eshtehardi, P.; McDaniel, M. C.; Suo, J.; Dhawan, S. S.; Maynard, C.; Timmins, L. H.; Quyyumi, A. A.; Giddens, D. P. Coronary artery wall shear stress is associated with progression and transformation of atherosclerotic plaque and arterial remodeling in patients with coronary artery disease. Circulation 2011, 124, 779–788.
Mao, C. B.; Solis, D. J.; Reiss, B. D.; Kottmann, S. T.; Sweeney, R. Y.; Hayhurst, A.; Georgiou, G.; Iverson, B.; Belcher, A. M. Virus-based toolkit for the directed synthesis of magnetic and semiconducting nanowires. Science 2013, 303, 213–217.
Murugesan, M.; Abbineni, G.; Nimmo, S. L.; Cao, B. R.; Mao, C. B. Virus-based photo-responsive nanowires formed by linking site-directed mutagenesis and chemical reaction. Sci. Rep. 2013, 3, 1820.
Nam, K. T.; Kim, D. W.; Yoo, P. J.; Chiang, C. Y.; Meethong, N.; Hammond, P. T.; Chiang, Y. M.; Belcher, A. M. Virusenabled synthesis and assembly of nanowires for lithium ion battery electrodes. Science 2006, 312, 885–888.
Chiang, C. Y.; Mello, C. M.; Gu, J.; Silva, E. C. C. M.; Van Vliet, K. J.; Belcher, A. M. Weaving genetically engineered functionality into mechanically robust virus fibers. Adv. Mater. 2007, 19, 826–832.
Niu, Z. W.; Bruckman, M. A.; Harp, B.; Mello, C. M.; Wang, Q. Bacteriophage M13 as a scaffold for preparing conductive polymeric composite fibers. Nano Res. 2008, 1, 235–241.
Domaille, D. W.; Lee, J. H.; Cha, J. N. High density DNA loading on the M13 bacteriophage provides access to colorimetric and fluorescent protein microarray biosensors. Chem. Commun. 2013, 49, 1759–1761.
Suthiwangcharoen, N.; Li, T.; Li, K.; Thompson, P.; You, S. J.; Wang, Q. M13 bacteriophage-polymer nanoassemblies as drug delivery vehicles. Nano Res. 2011, 4, 483–493.
Carrico, Z. M.; Farkas, M. E.; Zhou, Y.; Hsiao, S. H.; Marks, J. D.; Chokhawala, H.; Clark, D. S.; Francis, M. B. N-terminal labelling of filamentous phage to create cancer marker imaging agents. ACS Nano 2012, 6, 6675–6680.
Khalil, A. S.; Ferrer, J. M.; Brau, R. R.; Kottmann, S. T.; Noren, C. J.; Lang, M. J.; Belcher, A. M. Single M13 bacteriophage tethering and stretching. Proc. Nat. Acad. Sci. USA 2007, 104, 4892–4897.
Pacheco-Gómez, R.; Kraemer, J.; Stokoe, S.; England, H. J.; Penn, C. W.; Stanley, E.; Rodger, A.; Ward, J.; Hicks, M. R.; Dafforn, T. R. Detection of pathogenic bacteria using a homogeneous immunoassay based on shear alignment of virus particles and linear dichroism. Anal. Chem. 2012, 84, 91–97.
Sidhu, S. S. Engineering M13 for phage display. Biomol. Eng. 2001, 18, 57–63
Cheng, X.; Joseph, M. B.; Covington, J. A.; Dafforn, T. R.; Hicks, M. R.; Rodger, A. Continuous-channel flow linear dichroism. Anal. Methods 2012, 4, 3169–3173.
Satchell, S. C.; Tasman, C. H.; Singh, A.; Ni, L.; Geelen, J.; von Ruhland, C. J.; O’Hare, M. J.; Saleem, M. A.; van den Heuvel, L. P.; Mathieson, P. W. Conditionally immortalized human glomerular endothelial cells expressing fenestrations in response to VEGF. Kidney Int. 2006, 69, 1633–1640.
Otsu, N. A threshold selection method from gray-level histogram. IEEE Trans. Syst. Man Cybernetics. 1979, 9, 62–66.
Schindelin, J.; Arganda-Carreras, I.; Frise, E. Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B. et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682.
Barbee, K. A.; Mundel, T.; Lal, R.; Davies, P. F. Subcellular distribution of shear stress at the surface of flow-aligned and nonaligned endothelial monolayers. Am. J. Physiol. 1995, 268, H1765–H1772.
Pozrikidis, C. Shear flow over a protuberance on a plane wall. J. Eng. Math. 1997, 31, 29–42.
Arkill, K. P.; Neal, C. R.; Mantell, J. M.; Michel, C. C.; Qvortrup, K.; Rostgaard, J.; Bates, D. O.; Knupp, C.; Squire, J. M. 3D reconstruction of the glycocalyx structure in mammalian capillaries using electron tomography. Microcirculation 2012, 19, 343–351.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lobo, D.P., Wemyss, A.M., Smith, D.J. et al. Direct detection and measurement of wall shear stress using a filamentous bio-nanoparticle. Nano Res. 8, 3307–3315 (2015). https://doi.org/10.1007/s12274-015-0831-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-015-0831-x