Abstract
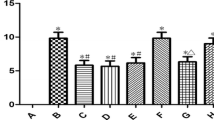
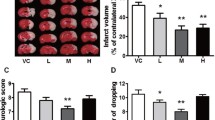
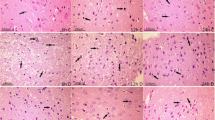
Mitochondrial membrane permeability is closely related to cerebral ischemia/reperfusion (I/R) injury. This paper explored the neuroprotective effect of picroside II (Picr), which inhibits the permeability of mitochondrial permeability transition pore (mPTP) and endonuclease G (EndoG) release from mitochondria into cytoplasm after cerebral I/R in rats. After 2 h of cerebral ischemia and 24 h of reperfusion in rats with different intervention measures, the neurobehavioral function, infarction volume, and reactive oxygen species (ROS) content in brain tissues were observed by modified neurological severity scale (mNSS), triphenyl tetrazolium chloride (TTC) staining, and enzyme-linked immunosorbent assay, respectively. The permeability of mPTP was assayed using spectrophotometry. The morphology and apoptotic cells of brain tissues were observed by hematoxylin-eosin staining and terminal deoxynucleotidyl transferase dUTP nick end labeling assay, respectively. The expressions of EndoG and voltage-dependent anion channel 1 (VDAC1) were determined by immunohistochemical assay and western blot. The Picr group exhibited clear decreases in mNSS scores, ROS content, number of apoptotic cells, mPTP permeability and expression of VDAC1, and EndoG in cytoplasm and nuclei, and the morphology of brain tissue was improved as compared with the model group (P < 0.05). Picr could attenuate cerebral I/R injury by downregulating the expression of VDAC1 and decreasing the permeability of mPTP, thereby inhibiting EndoG release from mitochondria into cytoplasm.









Similar content being viewed by others
References
Alvira CM, Umesh A, Husted C, Ying L, Hou Y, Lyu SC, Cornfield DN (2012) Voltage-dependent anion channel-2 interaction with nitric oxide synthase enhances pulmonary artery endothelial cell nitric oxide production. Am J Respir Cell Mol Biol 47(5):669–678. https://doi.org/10.1165/rcmb.2011-0436OC
Arnoult D, Gaume B, Karbowski M, Sharpe JC, Cecconi F, Youle RJ (2003) Mitochondrial release of AIF and EndoG requires caspase activation downstream of Bax/Bak-mediated permeabilization. EMBO J 22(17):4385–4399. https://doi.org/10.1093/emboj/cdg423
Azarashvili T, Krestinina O, Baburina Y, Odinokova I, Akatov V, Beletsky I, Papadopoulos V (2016) Effect of the CRAC peptide, VLNYYVW, on mPTP opening in rat brain and liver mitochondria. Int J Mol Sci 17(12). https://doi.org/10.3390/ijms17122096
Baines CP (2009) The mitochondrial permeability transition pore and ischemia-reperfusion injury. Basic Res Cardiol 104(2):181–188. https://doi.org/10.1007/s00395-009-0004-8
Ben-Hail D, Shoshan-Barmatz V (2016) VDAC1-interacting anion transport inhibitors inhibit VDAC1 oligomerization and apoptosis. Biochim Biophys Acta 1863(7 Pt a):1612–1623. https://doi.org/10.1016/j.bbamcr.2016.04.002
Chen J, Sanberg PR, Li Y, Wang L, Lu M, Willing AE, Sanchez-Ramos J, Chopp M (2001) Intravenous administration of human umbilical cord blood reduces behavioral deficits after stroke in rats. Stroke 32(11):2682–2688. https://doi.org/10.1161/hs1101.098367
Chu Y, Goldman JG, Kelly L, He Y, Waliczek T, Kordower JH (2014) Abnormal alpha-synuclein reduces nigral voltage-dependent anion channel 1 in sporadic and experimental Parkinson’s disease. Neurobiol Dis 69:1–14. https://doi.org/10.1016/j.nbd.2014.05.003
De PV, Tomasello F, Messina A, Guarino F, Benz R, La MD et al (2007) Determination of the conformation of the human VDAC1 N-terminal peptide, a protein moiety essential for the functional properties of the pore. Chembiochem A Eur J Chem Biol 8(7):744–756. https://doi.org/10.1002/cbic.200700009
Du K, McGill MR, Xie Y, Bajt ML, Jaeschke H (2015) Resveratrol prevents protein nitration and release of endonucleases from mitochondria during acetaminophen hepatotoxicity. Food Chem Toxicol 81:62–70. https://doi.org/10.1016/j.fct.2015.04.014
Fernandes MA, Marques RJ, Vicente JA, Santos MS, Monteiro P, Moreno AJ, Custodio JB (2008) Sildenafil citrate concentrations not affecting oxidative phosphorylation depress H2O2 generation by rat heart mitochondria. Mol Cell Biochem 309(1–2):77–85. https://doi.org/10.1007/s11010-007-9645-9
Godbole A, Varghese J, Sarin A, Mathew MK (2003) Vdac is a conserved element of death pathways in plant and animal systems. Biochim Biophys Acta 1642(2):87–96. https://doi.org/10.1016/S0167-4889(03)00102-2
He F, Duan X, Dai R, Li Y, Lin Q (2017) Protective effect of 4-methoxy benzyl alcohol on the blood-brain barrier after cerebral ischemia reperfusion injury. J Stroke Cerebrovasc Dis 26(6):1258–1265. https://doi.org/10.1016/j.jstrokecerebrovasdis.2017.01.018
Higgins GC, Beart PM, Nagley P (2009) Oxidative stress triggers neuronal caspase-independent death: endonuclease g involvement in programmed cell death-type iii. Cell Mol Life Sci 66(16):2773–2787. https://doi.org/10.1152/ajpregu.00747.2009
Israelson A, Zaid H, Abu-Hamad S, Nahon E, Shoshan-Barmatz V (2008) Mapping the ruthenium red-binding site of the voltage-dependent anion channel-1. Cell Calcium 43(2):196–204. https://doi.org/10.1016/j.ceca.2007.05.006
Keinan N, Pahima H, Ben-Hail D, Shoshan-Barmatz V (2013) The role of calcium in VDAC1 oligomerization and mitochondria-mediated apoptosis. Biochim Biophys Acta 1833(7):1745–1754. https://doi.org/10.1016/j.bbamcr.2013.03.017
Laker RC, Taddeo EP, Akhtar YN, Zhang M, Hoehn KL, Yan Z (2016) The mitochondrial permeability transition pore regulator cyclophilin D exhibits tissue-specific control of metabolic homeostasis. PLoS One 11(12):e0167910. https://doi.org/10.1371/journal.pone.0167910
Lan CH, Sheng JQ, Fang DC, Meng QZ, Fan LL, Huang ZR (2010) Involvement of VDAC1 and Bcl-2 family of proteins in VacA-induced cytochrome c release and apoptosis of gastric epithelial carcinoma cells. J Dig Dis 11(1):43–49. https://doi.org/10.1111/j.1751-2980.2009.00412.x
Lee BI, Lee DJ, Cho KJ, Kim GW (2005) Early nuclear translocation of endonuclease G and subsequent DNA fragmentation after transient focal cerebral ischemia in mice. Neurosci Lett 386(1):23–27. https://doi.org/10.1016/j.neulet.2005.05.058
Liu W, Wang X, Yang S, Huang J, Xue X, Zheng Y, Chen L (2016) Electroacupunctre improves motor impairment via inhibition of microglia-mediated neuroinflammation in the sensorimotor cortex after ischemic stroke. Life Sci 151:313–322. https://doi.org/10.1016/j.lfs.2016.01.045
Lü AJ, Dong CW, Du CS, Zhang QY (2007) Characterization and expression analysis of paralichthys olivaceus voltage-dependent anion channel (vdac) gene in response to virus infection. Fish Shellfish Immunol 23(3):601–613. https://doi.org/10.1016/j.fsi.2007.01.007
McCommis KS, Baines CP (2012) The role of VDAC in cell death: friend or foe? Biochim Biophys Acta 1818(6):1444–1450. https://doi.org/10.1016/j.bbamem.2011.10.025
Perez MJ, Quintanilla RA (2017) Development or disease: duality of the mitochondrial permeability transition pore. Dev Biol 426(1):1–7. https://doi.org/10.1016/j.ydbio.2017.04.018
Schäfer P, Scholz SR, Gimadutdinow O, Cymerman IA, Bujnicki JM, Ruiz-Carrillo A, Pingoud A, Meiss G (2004) Structural and functional characterization of mitochondrial EndoG, a sugar non-specific nuclease which plays an important role during apoptosis. J Mol Biol 338(2):217–228
Shoshan-Barmatz V, Ben-Hail D (2012) VDAC, a multi-functional mitochondrial protein as a pharmacological target. Mitochondrion 12(1):24–34. https://doi.org/10.1016/j.mito.2011.04.001
Shoshan-Barmatz V, De S, Meir A (2017) The mitochondrial voltage-dependent anion channel 1, Ca2+ transport, apoptosis, and their regulation. Front Oncol 7:60. https://doi.org/10.3389/fonc.2017.00060
Shoshan-Barmatz V, Keinan N, Abu-Hamad S, Tyomkin D, Aram L (2010) Apoptosis is regulated by the VDAC1 N-terminal region and by VDAC oligomerization: release of cytochrome c, AIF and Smac/Diablo. Biochim Biophys Acta 1797(6–7):1281–1291. https://doi.org/10.1016/j.bbabio.2010.03.003
Studencka M, Schaber J (2017) Senoptosis: non-lethal DNA cleavage as a route to deep senescence. Oncotarget 8:30656–30671
Tomasello F, Messina A, Lartigue L, Schembri L, Medina C, Reina S, Thoraval D, Crouzet M, Ichas F, de Pinto V, de Giorgi F (2009) Outer membrane vdac1 controls permeability transition of the inner mitochondrial membrane in cellulo during stress-induced apoptosis. Cell Res 19(12):1363–1376. https://doi.org/10.1038/cr.2009.98
Tomasello M, Guarino F, Reina S, Messina A, Depinto V (2013) The voltage-dependent anion selective channel 1 (VDAC1) topography in the mitochondrial outer membrane as detected in intact cell. PLoS One 8(12):e81522. https://doi.org/10.1371/journal.pone.0081522.g001
Warne J, Pryce G, Hill JM, Shi X, Lenneras F, Puentes F, Selwood DL (2016) Selective inhibition of the mitochondrial permeability transition pore protects against neurodegeneration in experimental multiple sclerosis. J Biol Chem 291(9):4356–4373. https://doi.org/10.1074/jbc.M115.700385
Weisthal S, Keinan N, Ben-Hail D, Arif T, Shoshan-Barmatz V (2014) Ca(2+)-mediated regulation of VDAC1 expression levels is associated with cell death induction. Biochim Biophys Acta 1843(10):2270–2281. https://doi.org/10.1016/j.bbamcr.2014.03.021
Xu Z, Zhang J, David KK, Yang ZJ, Li X, Dawson TM, Dawson VL, Koehler RC (2010) Endonuclease g does not play an obligatory role in poly(adp-ribose) polymerase-dependent cell death after transient focal cerebral ischemia. Am J Physiol Regul Integr Comp Physiol 299(1):R215–R221. https://doi.org/10.1152/ajpregu.00747.2009
Yu Z, Liu N, Li Y, Xu J, Wang X (2013) Neuroglobin overexpression inhibits oxygen-glucose deprivation-induced mitochondrial permeability transition pore opening in primary cultured mouse cortical neurons. Neurobiol Dis 56:95–103. https://doi.org/10.1016/j.nbd.2013.04.015
Zaid H, Abuhamad S, Israelson A, Nathan I, Shoshanbarmatz V (2005) The voltage-dependent anion channel-1 modulates apoptotic cell death. Cell Death Differ 12(7):751–760. https://doi.org/10.1038/sj.cdd.4401599
Zhan M, Brooks C, Liu F, Sun L, Dong Z (2013) Mitochondrial dynamics: regulatory mechanisms and emerging role in renal pathophysiology. Kidney Int 83(4):568–581. https://doi.org/10.1038/ki.2012.441
Zhang H, Zhai L, Wang T, Li S, Guo Y (2017) Picroside II exerts a neuroprotective effect by inhibiting the mitochondria cytochrome C signal pathway following ischemia reperfusion injury in rats. J Mol Neurosci 61(2):267–278. https://doi.org/10.1007/s12031-016-0870-0
Zhdanov DD, Fahmi T, Wang X, Apostolov EO, Sokolov NN, Javadov S, Basnakian AG (2015) Regulation of apoptotic endonucleases by EndoG. DNA Cell Biol 34(5):316–326. https://doi.org/10.1089/dna.2014.2772
Acknowledgements
We would like to thank Editage [www.editage.cn] for English language editing.
Funding
This work was supported by the National Natural Science Foundation of China (81274116) and The Clinical Medicine + X Project Fund of Qingdao University Medical College (2017-32).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal-related experiments were performed using the protocols approved by our institute (The Ethics Committee of Qingdao University Medical College, QUMC 2011-09)
Conflicts of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Li, S., Wang, T., Zhai, L. et al. Picroside II Exerts a Neuroprotective Effect by Inhibiting mPTP Permeability and EndoG Release after Cerebral Ischemia/Reperfusion Injury in Rats. J Mol Neurosci 64, 144–155 (2018). https://doi.org/10.1007/s12031-017-1012-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-017-1012-z