Abstract
The Systems Approach Framework with an integrated Ecological-Social-Economic assessment was applied to address the issue of zebra mussel (Dreissena polymorpha) farming in the large Oder (Szczecin) Lagoon, southern Baltic Sea. Heavy eutrophication hampers the use of the lagoon and zebra mussel farming is considered as new use and potential measure to improve water quality. Three alternative scenarios were developed in interaction with local stakeholders: 1) the production of mussels as fresh feed and meal on a commercial basis seemed not profitable, because of a limited market for fresh mussels (zoos, aquaculture) and low prices for organic feed. 2) Mussel cultivation to improve transparency and attractiveness of bathing waters near beaches had only a limited potential (0.2 m improvement of Secchi depth). A higher mussel biomass would increase the risk of temporary hypoxia. 3) Mussels farms for improving the environmental status (according to EU Water Framework Directive) by supporting macrophyte restoration were considered as the most promising scenario. Our model simulations suggested that as soon as a compensation for nutrient removal is considered, all mussel farm scenarios could cover the costs. Experiments and literature confirm that the conditions for an environmental friendly farming approach in the lagoon are suitable. Steps towards and problems associated with an implementation, e.g. invasion of Dreissena bugensis (quagga mussel), are discussed. Each step of the Ecological-Social-Economic assessment and major lessons learnt are documented in detail. Altogether, the approach turned out to be very suitable for this issue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Systems Approach Framework (SAF) with its integrated Ecological-Social-Economic assessment is a stepwise, user-friendly methodology with high practical relevance that allows addressing problems and challenges in the coastal zone systematically (Hopkins et al. 2011). It is a major approach to support Integrated Coastal Zone Management (ICZM) (Støttrup et al. 2017) and an Ecosystem Approach to Management (EAM).
Spatially, this case study focusses on the Oder (Szczecin) Lagoon at the German/Polish border in the southern Baltic Sea region. High riverine nutrient loads, entering with the Oder/Odra River, cause poor water quality, low transparency, an unsatisfactory ecological status in the Oder Lagoon and hamper the socio-economic development. The summerly chlorophyll-a (chl-a) concentrations, for example, are four times above the threshold for the good environmental status (Schernewski et al. 2015). Especially during summer, eutrophication effects like cyanobacteria blooms or fish kills due to hypoxia can cause serious economic problems for tourism (Dolch and Schernewski 2003, Wasmund 2002).
In the first application approach, we addressed this issue using the Integrated Coastal Area - River Basin Management (ICARM) approach of the United Nations Environmental Programme (UNEP) (Schernewski et al. 2005; Schernewski 2008). A comprehensive integrated modelling approach was carried out (Behrendt and Dannowsk 2005; Behrendt et al. 2008; Schernewski et al. 2008; Schernewski et al. 2011a). The work was supported by UNEP-GPA (Global Programme of Action for the Protection of the Marine Environment from Land-Based Activities), ministries, authorities and the German-Polish Regional Agenda 21 ‘Stettiner Haff’ (Schernewski et al. 2005; Schernewski et al. 2011b). Conclusions were that even maximum realistic riverine nutrient load reductions in the Odra river basin seemed not to be sufficient to transfer the lagoon into a non-eutrophic state. Beside insufficient political will, the lack of comprehensive water quality objectives for the river as well as the transitional and coastal waters hampered the implementation of nutrient retention and avoidance measures to fulfill the requirements of the EU Water Framework Directive (Schernewski et al. 2008).
In the second approach, the SAF was used to analyze to what extent a comprehensive eutrophication management approach should also include internal nutrient retention and removal measures in the lagoon (Schernewski 2008). This application followed the steps of the Ecological-Social-Economic (ESE) assessment, as suggested by Hopkins et al. (2011) under involvement of ministries, authorities and the German-Polish Regional Agenda 21 ‘Stettiner Haff’. The focus was on the potential of zebra mussel (Dreissena polymorpha) farming for removing nutrients and improving water transparency in the Oder Lagoon. Previous studies indicated that zebra mussels are abundant in the lagoon (Radziejewska et al. 2009; Wolnomiejski and Woźniczka 2008) and possess a high cleaning potential (Newell 2004; Fenske 2005; Schories et al. 2006; Stybel et al. 2009; Zaiko et al. 2009). For this purpose, an ecological model was extended by a mussel module and an economic model. The model simulations showed that mussel farming in the lagoon could only be considered as supporting measure to improve water quality. A maximum size mussel farm established in the lagoon has the potential to remove 1000 t N and 70 t of P, about 2% of the annual nutrient loads (Schernewski et al. 2012). However, this approach suffered from several weaknesses. Most important is that it considered mussel farming only on a large scale and focused on nutrient removal. Concrete and spatially well-defined cultivation scenarios on a local level were lacking and did not allow a full SAF application (Schernewski 2008; McFadden and Schernewski 2014). Further, the positive effects of mussel farming on water transparency and resulting economic and ecological implications were not taken into account.
Objective of this paper is to document a third Ecological-Social-Economic (ESE) application within the SAF that overcomes previous shortcomings. We develop and provide a clear realistic vision about purpose, size, setup and locations of potential mussel farms in the Oder Lagoon. Three scenarios on mussel cultivation are distinguished: 1) to produce mussels as fresh feed and meal on a commercial basis, 2) to use mussel cultivation to improve transparency and attractiveness of bathing waters at beaches and c) to improve the environmental status and support the objectives of the Water Framework Directive (WFD) by supporting macrophyte restoration. Improved and spatially resolved ecological and economic models allow the assessment of specific ecological and economic implications of mussel cultivation. These results serve as a basis for discussions with stakeholders towards an implementation of mussel farms in the Oder Lagoon. Focus and guiding idea of this article is to describe the stepwise process and the interaction with the stakeholders and to assess the general suitability of the SAF as methodology in coastal management. Therefore, the presentation of the results is largely organized according to the SAF succession, rather than following the common structure of a scientific paper.
Study site & methods
The Oder lagoon
With a surface area of 687 km2, the Oder Lagoon is one of the largest lagoons in Europe, shared between Germany (Kleines Haff, 277 km2) and Poland (Wielki Zalew, 410 km2) (Fig. 1). Humans use the lagoon since the early Middle Ages. For example, already in the fifteenth century, fisheries became so intensive that it had to be regulated. Currently, most of the coastal area and the water surface of the Oder Lagoon are under nature protection and reported as EU Natura 2000 site (EU Habitats and Birds Directives). It means that only those human activities are allowed that do not have negative impact on the species or habitat types for which the site has been designated. Along the coastline, tourism is the most important economic factor. Fishing (above 160 professional fishermen with a total catch of 3000 t/a in the entire lagoon) and shipping are other important activities that occupy larger areas of the lagoon (Fig. 1).
The average annual total nutrient load (2010–2014) into the lagoon is about 60,000 t total nitrogen (N) and 3000 t total phosphorus (P) (Pastuszak et al. 2018). The Oder/Odra River discharge dominates the lagoon’s water (average discharge of 530 m3/s) budgets and contributes more than 95% of the total nutrient loads. It is responsible for heavy eutrophication (average annual water transparency of 0.9 m) and frequent blue-green algae blooms in summer. Because of its shallowness (average depth of 3.7 m, maximum natural depth 8.5 m), the lagoon’s average water exchange time is only about 55 days (Radziejewska and Schernewski 2008). Three outlets connect the lagoon to the Baltic Sea. The low average salinity of around 1.5 psu indicates the minor influence of the Baltic Sea (6 psu). As consequence of low salinity, zebra mussels (Dreissena polymorpha) are the only filter-feeding epifaunal bivalve in the lagoon (Wolnomiejski and Witek 2013). They form mussel beds in all parts of the lagoon, with an estimated total biomass of about 68,000 t (Radziejewska et al. 2009, Wolnomiejski and Wozniczka 2008). The mussel was common throughout Northern Germany even before the last Ice Age and re-colonized the Oder Lagoon in the nineteenth century (Fenske 2003; Stybel et al. 2009). Therefore, we consider zebra mussels as naturally occuring species.
The ecological model and experiments
The ecological model consists of a coupled 3d-hydrographical-biogeochemical model (GETM-ERGOM) linked to a mussel module. GETM-ERGOM has a horizontal resolution of 150 m and 20 vertical layers (sigma-layers). The vertical layers follow the bottom bathymetry and provide a very high vertical resolution in shallow waters. GETM (Burchard and Bolding 2002) calculates the abiotic conditions (temperature, salinity) as well as horizontal and vertical flow fields based on weather forcing (provided by the German Weather Forecast DWD). The resuspension of the sediment, as function of bottom flow velocity and bottom friction is taken into account. During the years 2015–2017 several Acoustic Doppler Current Profiler (ADCP) field data collections, focusing on the south-western lagoon coast, were carried out to validate the flow simulations under different wind conditions.
The biogeochemical model ERGOM considers three inorganic nutrient compounds (ammonium, nitrate, phosphate) and three functional phytoplankton groups (large phytoplankton representing spring diatoms, small phytoplankton representing mainly summerly flagellates and potentially nitrogen-fixing cyanobacteria). All phytoplankton groups are grazed by zooplankton. Dead phyto- and zooplankton becomes detritus, which is re-mineralized into ammonium and phosphate, if oxygen is available. Oxygen consumption and production are taken into account. Processes in the sediment, like denitrification or the release of phosphate under anoxic condition, are included in a simplified way. The mussel module considers phytoplankton removal, oxygen consumption and release of detritus and dissolved nutrients.
Riverine nutrient loads and atmospheric deposition data were provided by LUNG (Federal State Agency for the Environment) and Pastuszak et al. (2018). Our ecological model simulations cover the years 2007 to 2015. The model was applied to Baltic coastal ecosystems and the Oder Lagoon several times before (e.g. Schernewski et al. 2015). Therefore, we considered the ecological model as sufficiently reliable for general scenario simulations and did not carry out a specific model validation for this application. Calibration data for the mussel module were obtained from literature and own laboratory experiments, especially zebra mussel filtration experiments.
To quantify the filtration/clearance of Dreissena, two methods were applied: a) mussels from the lagoon were placed in transparent PVC tubes (length 2 m, diameter 0.4 m) with open ends and deployed in the lagoon; b) mussels from the lagoon were cultivated in an aquarium in the laboratory, filled with lagoon water. The tube approach allowed comparing the mussel’s clearance effect in a semi-closed environment under nearly natural conditions. After a spin-up phase in all experiments, concentrations of chl-a and dissolved nutrients were frequently determined. Parallel experiments without mussels were carried out to determine the specific sinking rates of phytoplankton.
Additionally, literature data was used. The filtration was adapted depending on the abiotic conditions following Schneider (1992) for temperature and McMahon (1996) and Kilgour et al. (1994) for salinity. According to McLaughlan and Aldridge (2013) and Goedkoop et al. (2011), 10.1% (0.93%) of the mussel’s dry weight is nitrogen (phosphorus). Combined with the estimation of Schneider (1992) that the dry weight is 15% of the wet weight, 1 ton mussels contain 15.1 kg nitrogen and 1.4 kg phosphorus.
The integration of a simple mussel module and the high spatial model resolution allows, different to previous approaches (Schernewski et al. 2011a, 2012), a detailed assessments of environmental impacts of single mussel farms on water body and sediment as well as the optimization of mussel farm structures and locations.
Potential zebra mussel supply and farming locations
Areas, generally suitable for mussel farming in the lagoon were analyzed in a previous study by Schadach (2013) using a Geographic Information System. Since no official guidelines for mussel farming in Germany exist, we defined criteria commercial farming locations should fulfill: a) >150 m distance to both sides of shipping routes, b) >150 m to dredging areas along shipping routes, c) no overlap with leisure boat traffic, d) > 500 m distance to bathing sites, e) > 500 m to harbors and 200 m to landings, f) >500 m to camping sites, g) > 50 m to emerse macrophytes, h) >200 m to fisheries boat berths and > 50 to fishing gear, i) water depth above 2 m.
We assumed that a provisional zebra mussel farm in the Oder Lagoon would rely on natural recruitment of the zebra mussel (without augmented seeding of spat on the farm installations). Therefore, to estimate spatial-temporal patterns in the potential larvae supply, we performed pilot plankton surveys over the putative zebra mussel reproduction season in the lagoon. We applied species-specific PCR assay to detect zebra mussel DNA in environmental samples (Ardura et al. 2016), assuming positive correlation between DNA signal and larvae abundance reported for other benthic bivalve species in the region (Ardura et al. 2015).
Plankton samples were collected on 5 stations in the Oder Lagoon (Fig. 4) on 7., 24., 30. May and 4. June 2017 using a plankton net with 55 μm mesh size. The stations were located in areas that were identified as potentially suitable for mussel farming. The water column from about 50 cm above bottom to the surface was sampled (~ 2 m water depth, vertical tow). Between sampling stations, the gear was thoroughly washed to reduce the risk of cross-contamination. Samples were stored cold until aseptic filtration of 3–4 subsamples by 0.45 μm Whatman GF/C filters took place within few hours. Filters were preserved individually in 96% ethanol until DNA analysis was performed according to Ardura et al. (2016).
The economic model, data collection, experiments and surveys
We largely assumed that zebra mussels have a similar nutrient content and composition compared to blue mussels and can serve as a substitute. To confirm this, we carried out a literature study. Additionally, we contracted a certified laboratory (LUFA-ITL in Kiel) to carry out a full meat content analysis of one zebra mussel sample from hard-substrate in the Oder Lagoon compared to a blue mussel sample from the farm in Kiel. The samples were taken on 15. Sept. and analyzed between 16. and 26. Sept.. The major harvesting season starts in late autumn. Altogether 69 parameters (ingredients/nutrients, minerals as well as amino and fatty acids) were analyzed. Water contents between the mussel species were comparable (84–89%) and raw-ash contents similar (1.3%). Detailed results are shown in chapter 3.3.
The economic model was implemented in a Microsoft-Excel spreadsheet. It separates the mussel production into commercial feed production and biomass production for nutrient removal and transparency increase. The model includes concrete assumptions on location, size, density and setup of a mussel farm and allows biomass yield calculations. The following cost variables were taken into account: specific investment, operational, maintenance and capital costs, the economic lifetime, and labor costs. Further costs for chemical and hygienic laboratory tests were considered based on experiences in Krost et al. (2011). Cost data for extractive long-line blue mussel cultivation in Baltic coastal waters, documented in Haas et al. (2015), and empirical data from a blue mussel farm in Kiel Bay (Krost et al. 2011) were adapted and transferred to our non-intensive zebra mussel production. Blue and zebra mussels occupy similar habitats, share very many characteristics and differ only in their salinity requirements. They co-exist and compete in some ecosystems. This indicates that they have comparable core biological traits, which allows approximations for the zebra mussel to be made based on existing comparable blue mussel studies (e.g. Christensen et al. 2003, Gren et al. 2009, Hoagland et al. 2003, Petersen et al. 2012, 2014).
While blue mussels are usually produced for human consumption and have a defined market and developed business model, this is different for zebra mussels. Because of their smaller size, they are not produced commercially and no market exists. Therefore, the assessment of possible customers, retail prices and sales volume where the most challenging task.
Fresh mussels as animal feed for zoos: The Zoological Garden in Osnabrück hosts about 3600 animals and 295 species of which 25 species are carnivorous. In 2016, we carried out zebra mussel feeding experiments with mongoose (Mungos mungo), oriental small-clawed otter (Aonyx cinerea), raccoon (Procyon lotor) and Arctic fox (Vulpes lagopus) (Klumpe 2016). Aim was to assess the potential use and market for fresh zebra mussels as feed in animal parks. For this purpose, we harvested zebra mussels in the Oder Lagoon and transported the living mussels to Osnabrück. In the zoo, the mussels were kept alive in baskets in a 0.5 m3 storage basin with permanent water circulation and oxygen supply. To be able to maintain the mussels over weeks, they got some feed every two days (Aquamedic, Reef Life planklo).
A comprehensive literature and data review was carried out to estimate how many animal parks and zoos exist in proximity to the Oder Lagoon, that potentially could utilize mussels as feed. We considered data on location, travel distance from the lagoon (using Google maps), size, number and species and of captive animals as well as information about animal species with a known preference for mussels in their natural diet. A 300 km distance range was arbitrarily applied. Based on the data of the Zoological Garden Osnabrück, we estimated their potential annual demand for fresh mussels as feed.
The costs of goods transportation with trucks is accounted for as 0.1 € per ton and km. This value resulted from an online Internet survey. Transportation of fresh mussels require cooling, special containers, additional precautions and trucks returning empty. Therefore, higher truck transport costs of 1.0 € per ton and km were assumed and additional costs of 400 €/t for special transport containers, selection of high quality mussels and additional processing. These estimates take into account the experiences with the Zoological Garden Osnabrück.
Fresh mussels as feed for fish aquaculture: we carried out a review of official fish aquaculture statistical data and focused on a distance of up to 300 km from the Oder Lagoon (northern German federal states of Mecklenburg-Vorpommern, Schleswig-Holstein, Niedersachen, Sachsen, Sachsen-Anhalt, Brandenburg). It is assumed that fresh mussels in average can replace 10% of the protein rich feed and can be sold at an average price of 0.3 €/kg, similar to the price for industrial fresh-mussel usage (cooked, preservatives) (Haas et al. 2015). Again, 1.0 € per ton and km truck transport costs were assumed, as well as the additional costs for transport containers.
Mussel meal: costs for blue mussel meal production are between 0.5–1 €/kg (Thong et al. 2013) and we assumed the average of 0.75 €/kg for zebra mussels, as well. For the blue mussel farm in Kiel with an annual production of 100 t, Krost et al. (2011) report production costs of 1.925 Euros/kg fresh mussels, including 1.25 Euros/kg labor costs. Assuming a larger, more cost effective farm and much reduced labor cost, because mussels do not require regular cleaning and manual sorting if they are not for human consumption, the assumed value of 0.75 €/kg for the production of zebra mussels in Germany under comparable conditions seems realistic. Blue mussel meal has a weight of about 5% of the fresh mussels including shells (Lindahl 2012) and our. The assumed market price for organic certified mussel meal was assumed as 2.0 €/kg (Haas et al. 2015). To be able to produce mussel meal on commercial basis, the annual production has to exceed 500 t/a mussel meal requiring about 10,000 t fresh mussels. The price for fresh blue mussels for meal production of 0.06 €/kg (Haas et al. 2015) was assumed for zebra mussels, as well.
Acceptance of mussel meal as feed: In an earlier master thesis, 52 aquaculture companies were contacted and received a questionnaire with the aim to explore the acceptance of zebra mussel meal as feed in fish aquaculture (Drews 2012). The questionnaire covered questions about size and structure of the fish aquaculture, feed frequency, costs and availability, possible feed alternatives, and their attitude towards mussel meal. 21 companies answered the questionnaire. The focus was on companies producing carp and trout in open pond systems. Additionally, we interviewed feed producers (questions attached to an introductory email) about their readiness to sell zebra mussel meal or fresh mussels. Only 3 out of 7 companies replied (Klumpe 2016). Questions were a) Do you use mussel meal in your products and if yes which species? b) Is mussel meal from your perspective a suitable substitute for fish meal? c) Are there doubts in using mussel meal? d) Do you need further information to consider mussel meal as a product? The questions left space for additional comments. For practical reasons this study was carried out in Austria, but we assume that these results are transferable to our study site.
The stakeholder process
As preparation of the stakeholder involvement activities and for obtaining a better understanding of relevant social, economic, environmental components and dependencies in the region, we drafted a DPSIR (Driver-Pressure-State-Impact-Response) and a CATWOE (Customers-Actors-Transformational process-Worldview-Owners-Environmental constraints) analysis (www.baltcoast.net) and visualized the results.
Altogether two workshops with stakeholders took place. The first stakeholder workshop on 21. Oct. 2015 at the south-western lagoon coast, in Ueckermünde, was attended by 31 persons. Introductory talks provided an overview about regional sustainable development, the role of tourism and the importance of the lagoon. Later, the meeting focused on the poor state of water quality in the lagoon, options to improve it and the potentials of mussel farming. Several general options for mussel farming were introduced. Zebra mussel farms as promising measure to improve water transparency was introduced to the participants and they discussed potentials, ways of implementation and results to be used for the sustainable development of the lagoon.
The second workshop in Sept. 2017 aimed at catching the view of the 18 attending stakeholders with respect to the different mussel farming scenarios. Information about all scenarios was provided in several introductory presentations. To guide the second workshop and to be able to end-up with concrete preferences and implementation suggestions, a computer aided stakeholder preference and planning tool was applied. The methodology is described in detail in Schumacher et al. (2018). The tool consists of a success criteria application and a facilitated weighting exercise. Stakeholders assessed the scenarios with respect to four pre-defined success indicators, which partly resulted from discussions during the first stakeholder meeting. The criteria were: minimal spatial conflicts, high public acceptance, improved quality of life of local inhabitants and sustainability of the scenario. The assessment was done in two groups, facilitated by a moderator. The moderators’ role was to enhance participation, ensure that all views are heard and discussed, to support the development of a joint perception and to enter a joint consensus score into a spreadsheet that visualizes the results. The moderator later compiled and presented the results to the entire stakeholder group.
Ecological-social-economic assessment within the SAF
The SAF includes an Ecological-Social-Economic (ESE) assessment (Hopkins et al. 2011). Table 1 shows a modified ESE assessment with six steps and defined tasks, developed within the BONUS BaltCoast project (www.baltcoast.net). This approach and the described single steps guided this study and the presentation of the results, because testing the suitability and applicability of the ESE for a concrete problem was a major objective.
Results & discussion
Issue identification
First steps in the ESE assessment were listing human activities and associated stakeholder groups in the region. We identified 29 stakeholders from science, governance (federal state ministry, state agency, authority and district representatives as well as local mayor), tourism, fisheries and nature protection NGOs. Additionally, we carried out an institutional mapping to get an overview about institutions related to lagoon and water quality issues. Further, we listed main ecosystem goods and services as well as economic drivers and identified relevant social and economic components. During the first stakeholder workshop on 21. Oct. 2015, the scientists introduced zebra mussel farming as potential measure for improving water transparency and the ecological status of the lagoon. After a discussion, the group agreed to pick up this issue for a detailed study.
In the Oder Lagoon region, water quality aspects were already addressed in earlier studies. Previous iterative attempts and the present approach can be visualized using the DPSIR method (Fig. 2). The earlier attempts showed that it is hard to tackle large areas with complex problems and a large amount of potential stakeholders and to come to concrete results in a limited time. Therefore, the present approach focused spatially on a relatively small area of about 150 km2, the south-western part of the Kleines Haff, including several coastal towns and the city of Ueckemünde. Additionally the focus shifted from environmental problems and possible solutions towards a local utilization of the lagoon, addressing aspects of the EU Blue Growth strategy (European Commission 2012). The 3rd DPSIR analysis of this study and its outcome (Fig. 2) remained controversial within the research group and other alternative DPSIR cycles existed, but the approach helped to guide discussions and to develop a joint view about the issue.
System design and scenarios
Schadach (2013) classified areas, which are suitable for mussel farming, based on 12 criteria (shipping routes, dredging areas, recreational boating, harbors, bathing sites, camping sites, nature protection core areas, fishery harbors, location of devices, sediments, water depth and flow velocity). Four priority area classes where distinguished, where 1 indicated most suitable areas and 4 least suitable areas (Fig. 3). This map focused on areas suitable for commercial farming. During our discussion process, we defined three alternative spatially explicit and realistic scenarios for mussel farming. Consequently, we introduced additional criteria for all scenarios, like a) sandy bottoms, b) sufficient flow velocity to avoid long-term accumulation of faeces and pseudo-faeces on the sediment surface and ensure sufficient phytoplankton transport to nourish mussels, c) historic mussel beds in the surrounding to ensure a high likelihood of mussel larvae in the water and natural settling on farms as well as d) no legal or spatial planning restrictions and e) no risk of pollution (organic contaminants, heavy metals).
Scenario 1 - commercial mussel farm
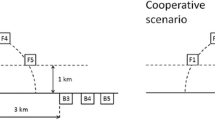
Objective was to establish a mussel farm that removes nutrients, increases water transparency, provides feed (fresh mussels and meal) in the most efficient way and can be maintained as a profitable business (Fig. 4). Additional pre-conditions were high phytoplankton concentrations as well as a low risk of damages due to drifting ice. We assumed 15 farm units of 2.25 ha each, covering an area of 34 ha and a harvested production of 1000 t of mussels per year. Further, we assumed a mussel biomass and annual yield of 1.5 kg/m3 water (bi-annual harvesting), as well as cultivation ropes down to 2 m in a water depth between 3 and 5 m. The general setup is shown in Fig. 11.
Scenario 2 - beach mussel farm to support bathing-tourism
Objective was to locate a mussel farm left and right of a beach, so that the prevailing coast-parallel currents flow through the farm and the transported waters are becoming more transparent. As a result, the bathing area would have an increased water transparency and would become more attractive for tourists. At the same time, near the mussel farm the emerged and submerged macrophyte areas would benefit from improved transparency, spread, stabilize the sediments and in turn further increase water transparency (Fig. 4). Present experiences show that macrophytes will not spread into the bathing area, because of mechanical damage by bathers. Pre-condition is a production density that excludes any negative impacts on water body and sediment (low oxygen concentrations) and locations with stable coast-parallel current pattern. We assumed a mussel farm producing 1500 t of mussels per year covering an area of 18 ha, with a mussel biomass of 5 kg/m3 water as well as cultivation ropes down to 1.7 m in a water depth between 2 and 3 m. Since mussels are harvested only after 2 years and in late autumn, the total biomass actively involved in filtration can be up to 3000 t.
Scenario 3 – Environmental mussel farm for nature restoration
Objective was to establish a mussel farm that induces a self-reinforcing cycle with increasing environmental quality, nature restoration and supports the implementation of the Water Framework Directive (Fig. 4). Idea was that increasing local water transparency enables the extension and restoration of submerged macrophyte belts, which in turn stabilize the sediments and further increase water transparency. Assumption was to establish several non-intensive (1.5 kg/m2 mussel biomass) farms covering a total area of 1.12 km2, spread over an area of about 4 km2 in shallow waters (< 2.5 m) near the south-eastern coast of the Kleines Haff. Very likely, these areas were still covered by macrophytes a century ago.
The system design step in the ESE includes the definition of administrative and virtual system boundaries, the development of scenarios, and the development of a conceptual model. The conceptual model links the simulation models with the scenarios and the framework conditions (Fig. 5). Its development was an iterative process including the identification of the state variables and processes needed to address the issue on one side and the availability of data, modelling and financial resources as well as expertise on the other side.
The next ESE steps were ‘system formulation’ (Table 1) and ‘system assessment’. The latter provided concrete model simulations, assessments and visualizations for each scenario as basis for stakeholder discussions.
System Formulation & Assessment: The commercial mussel farm
Our commercial scenario assumed that zebra mussels have a similar nutrient content and composition compared to blue mussels and can serve as an adequate substitute. Our comparative laboratory nutrition analysis of the two samples confirmed this. Zebra mussels (blue mussels) contained 8.4% (7.9%) protein in fresh weight, 0.8% (1.5%) fat, 0.14% (0.12%) phosphorus, 1.3% (1.3%) raw ash and 89.3% (84.1%) water. The sum of saturated fatty acids is 35.3% (29.2%), the sum of single unsaturated fatty acids 35.8% (25.1%) and sum of multiple unsaturated fat acids 28.6% (45.7%). In general, both species showed a comparable quality as feed. Some of the differences may be natural variability and may result from different locations, growing conditions and water contents. Our literature survey (Drews 2012) and recent results, obtained within DBU-project EBAMA, suggest that Baltic blue-mussel meal seem to be a very good substitute for fishmeal, this is especially true with respect to important ingredients, like vitamin E or fatty acid quantity and composition (Krost, pers. com.).
Revenue: In 2014, the total annual costs of the Zoo Osnabrück for altogether 68 t of protein-rich animal feed were about 118,000 € (e.g. fish, meat and living animals). For protein rich feed, the zoo paid an average price of 1.67 €/kg. In 2014, about 37,000 €/a were spent to buy altogether 28 t feed fish (sprat, smelt, roach, herring and mackerel). The zoo feeding experiments showed that mongooses (Mungos mungo) and the oriental small-clawed otters (Aonyx cinerea) immediately accepted zebra mussels as food, raccoons (Procyon lotor) even showed preference for zebra mussels and Arctic foxes (Vulpes lagopus) accepted it with some reluctance. The results allowed the extrapolation that fresh zebra mussels can substitute about 10% of all protein-rich feed in the zoo at costs of 11,270 €/a. The annual consumption could be 6.7 t in the entire zoo at acceptable costs of 1.68 €/kg. Fresh mussels turned out to have, compared to other feed, the important benefit that they occupy zoo-animals. Beyond a transportation distance of 1200 km, they exceed the value of the mussels and the revenue became zero (Fig. 6a).
Commercial mussel farming: a Revenue and potential sales volume in dependence of fresh mussel transport distance; b mussel production costs and revenues from sales to zoos, fish aquaculture and for mussel meal production in dependence of the produced amount of fresh mussels; c Comparison between costs and accumulated revenue per ton produced mussels. The dotted line indicates the production costs minus the financial compensation for removing nutrients with the harvest
Altogether 254 animal parks and zoos were identified within 300 km around the lagoon. The potential demand of fresh mussels estimated as 1.4 t/a (within 10 km), 4.5 t/a (10–50 km), 18.5 t/a (50–100 km), 94 t/a (100–150 km), 26 t/a (150–200 km), 58 t/a (200–250 km), 137 t/a (250–300 km). Altogether we calculated a potential market of 340 t/a within 300 km. The large zoos in Hamburg (31 t/a) and Berlin (48 t/a) were identified as most important single potential customers (Fig. 6a). The distance-dependent sales volume and the transportation combined gave an idea, how much fresh mussel biomass could be sold at a certain price. Above about 5000 tons, the revenue became negative (Fig. 6b).
In 2014, altogether 7841 t of aquaculture fish were produced in 32 closed and 40 open fish aquaculture systems within 300 km from the Oder Lagoon. 38% of the fishes were carp species and 17% trout species. Other important fish species were catfish, pikeperch and sturgeon. Most species are known to feed on mussels, at least temporary (Schadach 2013). Compared to zoological gardens the sale to and potential revenue from fish aquaculture is low. Existing protein-rich feed, like fishmeal, is still relatively cheap (below 1500 €/metric ton in 2017/2018; https://datacatalog.worldbank.org) and largely prevents the usage of fresh mussels as alternative.
The revenue functions did not consider the productions costs. Fig. 6c shows the production costs in comparison. The production costs are always higher than the possible revenue from sale. The cost and revenue functions meet at a production volume of 800 tons at a price of about 830 Euros/t. However, we have to assume that not the total production has a size and quality that is suitable for zoos. In our calculations, we did not take into that not the entire production but possibly only 80% can be sold at this price. A mussel production for fish meal above 5000 tons would be profitable even without other sale options (Fig. 6c). The compensation fee for removing nutrients alone would be able to cover the farming costs. It means that mussel farming is a cost-effective measure to remove nutrients. Therefore, it could receive its funding as a measure within the WFD.
Even in deeper water and with vertical cultivation ropes of 2 m length, a production of 5000 tons mussels would require about 1.7 km2 mussel cultivation area. In our approach, assuming farm units of 2.25 ha, about 74 units would be required. The units would have to be spread over the lagoon to ensure a sufficient nourishment. This seems not realistic.
The survey among 21 Austrian fish aquaculture companies showed that they would accept mussel meal as a replacement for fishmeal, preferably without shells. Because of the high acceptance, the similarities between fish and mussel meal and the possibility to store it, the market is practically not limited. Further, the surveyed feed producers considered mussel meal as highly interesting as long as a sufficient and continuous supply of mussels for the production could be ensured. The companies considered trading fresh mussels on a commercial large-scale basis as not realistic.
Independently from the produced amount, the production of zebra mussels seems to be no suitable business model. However, as soon as mussel farmers get a compensation for the removal of nutrients, the production of more than 100 tons would become profitable. This is shown in Fig. 6c (dotted red line), where the compensation for nutrient removal is included in the cost function.
System Formulation & Assessment: The beach mussel farm
The beach mussel scenario required reliable flow and transport model simulations. A comparison between data collected with an Acoustic Doppler Current Profiler, floating on the water surface, and model simulations (depth averaged data, integrated over 10 min) showed a very good agreement outside the coastal wind shelter, namely 1 km and more off the shoreline. Figure 7a-c presents near-shore flow data compared to model simulations for 3 different days with offshore wind situations. In general, the agreement between data and simulated flow velocities is good, but the direction sometimes differs. Wind shelter due to coastal vegetation was not taken into account by the model. Further, the floating current profiler was not able to collect data for the first few decimeters near the water surface. Therefore, in shallow waters the depth-averaged flow data might not be reliable. Despite these uncertainties, the model simulations seemed sufficiently reliable for our purpose.
During summer (May-Sept.), close to Ueckermünde beach, the model suggested an average flow parallel to the coast, mainly towards south-east, and dominating slow current velocities below 3 cm/s (Fig. 7d). Relatively steady coast-parallel slow current velocities were a suitable precondition for an effective water filtration by mussels and a potentially strong positive effect on water transparency in front of the beach.
In summer, the artificial sandy beach of Ueckermünde (800 m length and about 43,500 m2 area) is intensively used by visitors (Fig. 8a). The model simulation assumed up to 3000 tons of mussels, distributed beside both sides and in front of the beach (Fig. 8b). Because of mussel filtration activity, the model suggested an improved water transparency (Secchi depth) of up to 0.4–0.5 m (Fig. 8c). This means instead of 0.5–0.6 m the summerly Secchi depth would be about 1.0 m.
The model suggested that these 3000 tons of mussels cultivated in a relatively high density in a shallow area with a water depth below 2.5 m would cause about 20 days with oxygen concentrations below 1 mg/l near the bottom (Fig. 9a). Since this would be a risk for the environment, especially the benthic flora and fauna, and maybe counterproductive with respect to fostering bathing tourism, the cultivated mussel biomass had to be reduced to an acceptable level. According to the model, a mussel biomass of 1500 tons would avoid negative effects on bottom oxygen concentrations and would be environmentally sustainable, but would increase Secchi depth only by 0.19 m (Fig. 9a).
Ecological and economic aspects of a mussel farms near Ueckermünde beach: a) comparison of running costs (without investment costs) and potential accumulated revenue from different sources in dependency of the cultivated mussel biomass; b) model simulation results reflecting the dependency between water transparency (Secchi-depth) and days with oxygen concentration below 1 mg/l caused by the cultivated mussel biomass
Although the spatial resolution of the ecological model was compared to earlier model applications refined to 150 m grid cells, the model is not able to represent the coastline smoothly. This means it does not allow to optimize the allocation of the mussel farms. Therefore, we can assume that in reality a spatially optimized farming could generate the predicted effects with a lower mussel biomass.
In 2016, about 121,000 tourist overnight stays were recorded for Ueckermünde (Statistisches Jahrbuch 2017, https://www.laiv-mv.de). Taking into account tourists overnight stays in the surrounding of the city and in private accommodations, as well as day tourists, we estimated a number of 300,000 tourist days per year. To cover the costs for mussel farming around the beach, it was assumed that a fee is added to the tourist tax and for day visitors is added to the parking fee at the beach. Ueckermünde beach has a large parking area of about 1.5 ha. According to an empirical survey, tourists are willing to spend 1 € per day additionally for an improved water transparency of 1 m (Hirschfeld, pers. com.). Based on these assumptions, an improved water transparency by 0.19 m could generate about 57,000 € per year for supporting a 1500 t mussel farm.
Further, 25% of the tourists stated that they would come more often if the water transparency (Secchi depth) would be 1 m better (Hirschfeld, pers. com.). On average, overnight tourists spend 73 € per day in Mecklenburg-Vorpommern including overnight costs and daily expenses (Statistisches Jahrbuch 2017, https://www.laiv-mv.de). Using this value and assuming a 1 m better water transparency, the increased number of tourists would generate an additional income of about 5.4 mill €/a. Assuming that 50% of the 7% added value tax for overnight stays is used to support mussel farming this would generate an additional income for mussel farmers of about 190,000 €. Based on these calculations, but assuming an increased water transparency of only 0.19 m, a 1500 t mussel farm could be supported with 36,000 €/a additionally.
The accumulated revenue and the costs (per t of mussels) depending on the cultivated mussel biomass (standing stock) are shown in Fig. 9b. An environmental fee together with additional income from increased tourism is not able to cover the costs for a mussel farm. Adding a financial compensation for removing nutrients to the other two source of income would result in annual revenues of altogether 309 €/t and costs of 178 €/t mussels, assuming a 1500 t mussel farm. In this case a farmer would make a profit of 131 €/t mussels per year. Theoretically, already a small farm with a production of 300 t could make a profit. The cost function only includes running costs for maintenance, operation and labor. Including investment and capital cost, the total costs would be about 20% higher. However, all calculations are hypothetical, depend on several assumption and include simplifications. The provided numbers can hardly be considered as reliable, but served as starting point for discussions with stakeholders.
System formulation & assessment: The environmental mussel farm
In the EU Water Framework Directive (European Commission 2000), macrophytes serve as biological quality elements for surface waters. An improved status of macrophytes of the lagoon, is an environmental policy objective, because it would indicate an improved ecological status. Scenario 3 was sub-divided and altogether three model simulations were carried out, with a total mussel biomass of 540 t, 1621 t and 5065 t (Fig. 10a). With 1.5 kg mussels per m2 water surface, the average mussel density was low. As consequence, the farms covered large areas of 0.36, 1.1 and 3.5 km2. The increase in Secchi depth was 4.6 cm or 7.2% (summer average over 4 simulated years), considering the large farm. The available light above the sediment would increase by 45%. Because of low mussel densities, all farm-sizes would not affect the oxygen conditions above the sediment. The number of days with oxygen concentrations below 1 mg/l in average would remain below 1 day per year. Fig. 10b shows that the area with improved growing conditions for macrophytes is about 5 times larger than the area covered by mussels. Prevailing currents transport the transparent water along the shoreline and therefore into areas where macrophytes potentially could grow. The cost function is comparable to the beach scenario (Fig. 9b). The running costs of all assumed farms could potentially be covered by a financial compensation for nutrient removal.
a Effect of farms with different mussel biomass (1.5 kg mussel/m2) on average water transparency (Secchi-depth) during summer. b Spatial impact of a farm with 1621 t mussel biomass, covering an area of 1.1 km2, on Secchi depth. The farm is assumed to be located in shallow waters (< 2 m) at the south-western coast of the Oder Lagoon
Two major questions remained: would macrophytes recover and re-settle areas with higher light availability at the bottom without additional supporting measures? If yes, what are the critical light conditions to initiate growth and a spatial spread? Our field surveys and a literature study proves that different submerse macrophyte species are still present in the western lagoon and single macrophyte stands were found in a water depth of up to 1.8 m. This is true for the eastern, Polish part as well (Brzeska et al. 2015). Recent studies by Nowak et al. (2008) and Blindow et al. (2016) documented that germinable diaspores of several species are present in the sediments of all observed German Baltic coastal water. Nowak et al. concluded that diaspores have the potential to restore macrophyte communities. This can happen even decades after the stands were lost. The average nutrient levels in the Kleines Haff (western Oder Lagoon) have declined from about 250 μmol/l total nitrogen (9 μmol/l total phosphorus) in the late 1980s to about 100 μmol/l total nitrogen (5 μmol/l total phosphorus) between 2010 and 2015. However, this had no significant effects on water transparency (about 0.6 m Secchi depth, summer average 2010–2015) and chl-a concentration (about 70 μg/l summer average 2010–2015). Obviously, the lagoon shows a hysteresis effect and does not react to nutrient load reductions. There seems to be a potential for an improvement and the restoration of macrophyte stands. A good likelihood exists, that mussel farms could initiate a local restoration and can be considered as a supportive measure in the WFD. However, a concrete experimental farm is required to collect information on the required light conditions at the bottom, recovery behavior, and additional side effects of the farm e.g. on currents, turbulence, resuspension and shading.
System Assessment & Implementation
On 9. Sept. 2017, on a second workshop with 18 local and regional stakeholders the three scenarios and the results of the economic and ecological modelling were presented and the computer-aided preference and planning tool was applied. The number of participants declined, compared to the first meeting, because of the more specific topic. The tool application was successful in initiating an active, guided discussion on the mussel farm scenarios, where every participant’s view was equally taken into account. With 63%, the environmental farm was favored, compared to 19% for beach mussel farm and 18% for the commercial mussel farm. In the given time frame, only one group managed to finish tool application fully. The composition of the group had influence on the results, but the tendency was similar. Despite favoring scenario 3, the attendees were positive about all scenarios and called for an experimental implementation. An experimental farm of at least 1 ha was jointly suggested to gather the data required for more reliable economic and ecological model simulation as well as the analysis of possible positive and negative side-effects of mussel farms.
To ensure that one of the scenario can be implemented, two questions had to be answered: a) are zebra mussel larvae naturally available at the locations where mussel farming is considered and b) is it possible to grow the mussels on material and in a technical stetup that was originally designed for blue mussels (Petersen et al. 2012, 2014). Experiments that addressed these questions were already carried out prior and during the SAF application process, to avoid a delay in case of a mussel farm implementation.
In many cases, blue mussel larvae are caught at suitable places and are entered into growing devices and cultivated in other places. In our scenarios, we assume that the mussel larvae attach themselves and grow naturally in our farms, to save costs and to reduce the environmental impact of farming. Our Dreissena molecular study suggests presence of larvae at all sampled locations from 24. May onward, (Fig. 2). Earlier, on 7. May, positive molecular signal was detected at 3 of 5 sampled stations. This indicates a good recruitment potential, confirms our visual observation that Dreissena occupies suitable habitats fast and in high numbers everywhere in the lagoon and it allows a natural spat collection on the cultivation facilities.
A study by Schulze-Böttcher (2014) in Lake Usedom, an enclosed bay connected to the Oder Lagoon, showed that zebra mussels easily grow on common artificial nets (Fig. 11) in a density of 21,600–31,700 ind./m2. Within 4 months, the mussels increased their shell length from 1 to 1.2 cm to 1.8–1.9 cm. However, competition with bryozoans, moss animals, reduced the mussel growth. Grov (2015) concluded for Lake Usedom, that bio-deposition of faeces and pseudofaeces had only a minor effect on bio-available nutrients and organic carbon. The experiences of the cultivation experiments in Lake Usedom can be transferred to other parts of the Oder Lagoon and in more exposed open parts of the lagoon, problems with bryozoans are less likely. Against this background, the implementation of a farm is a realistic option. However, it is known from literature that zebra mussels may alter their environment e.g. by increasing the availability of phosphate (Wojtal-Frankiewicz and Frankiewicz 2011) or by affecting sediments (e.g. Christensen et al. 2003; Pollet et al. 2015). In our approach, we used the modelled oxygen depletion as indicator for negative impacts on the environment. We adjusted the mussel cultivation density, so that no increase in the number of days with hypoxia above the sediment occured.
An unexpected, new problem that hampers the implementation of a mussel farm is the closely related quagga mussel (Dreissena bugensis) that was recently observed in the eastern Oder Lagoon (Woźniczka et al. 2016). This new species, indigenous to the Dnieper River drainage of Ukraine, is currently invading the Oder Lagoon. A cultivation of Dreissena polymorpha would automatically favor the development of Dreissena bugensis. This may cause legal and permission problems for mussel farms e.g. according to EU-Regulation No 1143/2014 on the prevention and management of the introduction and spread of invasive alien species (European Union 2014). However, the stakeholders were not concerned about the quagga mussel occurrence, because it resembles the zebra mussel in behavior and appearance, and is only slightly bigger. From a pragmatic point of view, the quagga mussel may even be more suitable for farming.
As consequence of the unclear legal situation in the Oder Lagoon, and to test the general concept, in 2017, an experimental mussel farm was established in neighboring Greifswald Bay. Here, higher salinity (about 7 PSU) only allows the cultivation of blue mussels. Aim is to test a low-cost, nature friendly farming concept for producing mussel meal (as animal feed) and for removing nutrients, similar to the scenarios for the Oder Lagoon. Here, a cultivation system is tested that can be lowered to avoid damage by ice during winter (Fig. 11).
Conclusion
We are not aware of existing larger-scale zebra mussel cultivation approaches world-wide. However, zebra mussel farming seems to be a suitable measure for removing nutrients, raising water transparency and improving ecosystem quality, but its quantitative potential in the Oder Lagoon is limited. In smaller water bodies with less external nutrient loads, like lakes or reservoirs, zebra mussel cultivation may be a suitable water quality management option (McLaughlan and Aldridge 2013), but practical field experiences are lacking.
In the Oder Lagoon mussel cultivation could be implemented for concrete local purposes, like small scale feed production, improving water transparency at beaches or as environmental measure to enable macrophyte recovery. Zebra mussel cultivation in the lagoon would not be profitable as a business, because the market for fresh feed mussels is limited (animal parks, aquaculture) and mussel meal production would require a large production. Zebra mussel farms would produce feed of high quality that could serve as replacement for fishmeal. It would meet the standard for organic certified production and could support organic fish aquaculture (Naturland, https://www.naturland.de). The demand and price for such a product may increase in future and may make a commercial production realistic. Our model simulations suggest that as soon as a compensation for nutrient removal is considered, all mussel farm scenarios could cover the costs. Further, our experiments confirm that the conditions for an environmental friendly farming approach in the lagoon are suitable.
From a practical point of view, the positive effects of mussel farms on water transparency are at the moment most important. Even to establish mussel farms in the surrounding of beaches to improve bathing water transparency, was perceived as a worth considering option by our local and regional stakeholders. However, the potential water transparency improvement (Secchi depth) seems limited to about 20 cm. A higher mussel biomass may cause negative impacts on the environment and increase the risk of hypoxia.
Most promising in our stakeholders’ view are mobile mussel farms, which increase water transparency in shallow areas (water depth below 2 m) and initiate a recovery of submerse macrophytes, without disturbing already established submerse macrophytes. These mussel farms could potentially be moved to other places once the macrophytes are re-established. A temporary installation was perceived as a major advantage, because it limits potential local negative effects of mussel farms on sediments and the re-use in a different place would reduce costs. However, this has not been tested so far and additional field experiments are necessary to confirm the suitability of such an approach.
The work at the Oder Lagoon shows that the SAF is applicable on different spatial scales, and that iterative applications can be useful. In this study, we focused on a local approach. It is based on very concrete alternative scenarios (sets of measures) for well-defined purposes, covering a focus area of only 150 km2. This helped us to avoid problems occurring during previous applications (Schernewski et al. 2012), like selecting a manageable number of stakeholders, attracting stakeholders or adapting the group because of a shifting thematic focus. Aspects like differing horizontal and vertical administrative structures and responsibilities in a cross-border region were no problem. We did not have to deal with the challenge of external forcing factors (e.g. changes in legislation, deepening of shipping channels) controlling an issue and the decoupling of origin of a problem and its manifestation (e.g, river basins as source of pollution), like in previous applications.
On a local level, it was easy to map stakeholders and institutions and because of the limited number of potentially interested stakeholders, it was possible to keep the group open for additional interested persons. The concrete setup allowed us to attract stakeholders to attend the workshops and they showed interest, ownership and responsibility. Further, it allowed us to agree on concrete suggestions towards an implementation. A local approach is much better manageable with limited resources and less time consuming. It allows, to complete an Ecological-Social-Economic assessment within two years, an important aspect in keeping stakeholders involved.
In running the workshops, the application of supporting methods were beneficial, like the preference and planning tool, or an ecosystem service and sustainability indicator application. Noteworthy is that the stakeholders were positive about mussel-farming and asked for a test-implementation, despite the generally negative attitude of major German national agencies (UBA, BfN) towards mussel-farming. However, both agencies were identified as key-stakeholders and UBA was generally involved in the process.
On a local level, the provision of ecological and economic model assessments for each scenario is imperative. For many local actors, economic considerations are more familiar and convincing than mere ecological considerations. Further, the perspective that an implementation follows the discussion, maintains their interest.
Our major lessons learnt are: 1) SAF applications carried out within projects with limited time and resources should preferably focus on a local level (municipality). 2) Local applications should be conducted within a short time, ideally one or two years, to keep stakeholders attracted, to keep the group stable, to fit into election periods, and to be able, to plan steps towards a possible implementation. 3) The provision of concrete and realistic alternative scenarios and the provision of ecological and economic assessments for each scenario allows a faster and more concrete stakeholder involvement process and in turn a faster SAF application. 4) A concrete pathway towards implementation and a funding vision needs to be provided.
The local application of the SAF can be regarded as general example for other case studies, even for those addressing very different issues. The developed scenarios for mussel farming should meet the interests to other Baltic inner coastal waters, like the Curonian Lagoon or the Vistula Lagoon, and the scenarios as well as the ESE assessment, can largely be transferred, as well. A SAF application dealing with spatially larger and/or more complex issues (e.g. spatial planning) certainly would require a formal political commitment and a more formal stakeholder involvement process to be able to follow all steps of a SAF process active over a longer period of several years.
References
Ardura A, Zaiko A, Martinez JL, Samuiloviene A, Semenova A, Garcia-Vazquez (E 2015) eDNA and specific primers for early detection of invasive species - a case study on the bivalve Rangia cuneata, currently sprreading in Europe. Mar Environ Res 112: 48–55
Ardura A, Zaiko A, Borrell YJ, Samuiloviene A, Garcia-Vazquez E (2016) Novel tools for early detection of a global aquatic invasive, the zebra mussel Dreissena polymorpha. Aquat Conserv Mar Freshwat Ecosyst. https://doi.org/10.1002/aqc.2655
Behrendt H, Dannowsk R (eds) (2005) Nutrients and heavy metals in the Odra river system. Weißensee Verlag, Berlin
Behrendt H, Opitz D, Kolanek A, Korol R, Stronska M (2008) Changes of the nutrient loads of the Odra River during the last century – their causes and consequences. J Water Land Dev 12:127–144
Blindow I, Dahlke S, Dewart A, Flügge S, Hendreschke M, Kerkow A, Meyer J (2016) Long-term and interannual changes of submerged macrophytes and their associated diaspore reservoir in a shallow southern Baltic Sea bay: influence of eutrophication and climate. Hydrobiologia 778:121. https://doi.org/10.1007/s10750-016-2655-4
Brzeska P, Wozniczka A, Pelechaty M, Blindow I (2015) New records of chara connivens p. Salzmann ex a. Braun 1835-an extremely rare and protected species in polish brackish waters. Acta Soc Bot Pol 84(1). https://doi.org/10.5586/asbp.2015.010
Burchard H, Bolding K (2002) Getm - a general estuarine transport model. Scienti_c documentation, Tech. Rep. EUR 20253 EN
Christensen PB, Glud RN, Dalsgaard T, Gillespie P (2003) Impacts of longline mussel farming on oxygen and nitrogen dynamics and biological communities of coastal sediments. Aquaculture 218:567–588
Dolch T, Schernewski G (2003) Hat Wasserqualität eine Bedeutung für Touristen? Eine Studie am Beispiel des Oderästuars. Berichte Forschungs- und Technologiezentrum Westküste der Universität Kiel 28:197–205
Drews B (2012) Die Muschel Dreissena polymorpha als Futterbestandteil bedeutsamer Speisefische aus Aquakulturen mit Schwerpunkt auf ökologisch wirtschaftenden Produzenten und im Hinblick auf die Substitution von Wildfischfrodukten. Masterarbeit an der Universität Rostock. 64p
European Commission (2000) Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for community action in the field of water policy. http://ec.europa.eu/environment/water/water-framework/index_en.html
European Commission (2012) Blue Growth opportunities for marine and maritime sustainable growth (COM/2012/0494 final), https://ec.europa.eu/maritimeaffairs/policy/blue_growth_en
European Union (2014) Regulation No 1143/2014 of the European Parliament and of the Council of 22 October 2014 on the prevention and management of the introduction and spread of invasive alien species. https://eur-lex.europa.eu/
Fenske C (2003) Die Wandermuschel (Dreissena polymorpha) im Oderhaff und ihre Bedeutung für das Küstenzonenmanagement. - Dissertation an der Universität Greifswald. http://spicosa-inline.databases.euccd.de/files/documents/00000098_Fenske_DieWandermuschelimOderhaff.pdf. Accessed 3 Sept 2018
Fenske C (2005) Renaturierung von Gewässern mit Hilfe der Wandermuschel Dreissena polymorpha (Pallas 1771). Rostocker Meeresbiologische Beiträge 14:55–68
Goedkoop W, Naddafi R, Grandin U (2011) Retention of N and P by zebra mussels (Dreissena polymorpha Pallas) and its quantitative role in the nutrient budget of eutrophic Lake Ekoln. Sweden, Biol Invasions 13:1077. https://doi.org/10.1007/s10530-011-9950-9
Gren I-M, Lindahl O, Lindqvist M (2009) Values of mussel farming for combating eutrophication: an application to the Baltic Sea. Ecol Eng 35(5):935–945
Grov M (2015) Untersuchungen zur Kultivierung filtrierender Organismen im Usedomer See. Master of Science Thesis, Landschaftsökologie und Naturschutz, University of Greifswald
Haas S, Rößner Y, Schröder J, Bronnmann J, Jooss F, Loy, JP, Schulz C (2015) Konzeptionierung einer umweltverträglichen, marinen Aquakultur in schleswig-holsteinischen Ostseeküstengewässern. Konzeptstudie im Auftrag des Ministeriums für Energiewende, Landwirtschaft, Umwelt und ländliche Räume des Landes Schleswig-Holstein. Abschlussbericht 118p
Hoagland P, Kite-Powell HL, Jin D (2003) Business planning handbook for the ocean aquaculture of blue mussels. Marine Policy Center, Woods Hole Oceanographic Institution
Hopkins TS, Bailly D, Støttrup JG (2011) A systems approach framework for coastal zones. Ecology and Society 16(4):25
Kilgour BW, Mackie GL, Baker MA, Keppel R (1994) Effects of salinity on the condition and survival of zebra mussels (Dereissena polymorpha). Estuaries 17:385. https://doi.org/10.2307/1352671
Klumpe T (2016) Möglichkeiten der kommerziellen Nutzung von Zebra-Muscheln. Masterarbeit an der Universität Rostock. 71p
Krost P, Rehm S, Kock M, Piker L (2011) Leitfaden für nachhaltige marine Aquakultur. CRM-Coastal research & managenet GbR, Kiel, 64p
Lindahl O (2012) Mussel farming as an environmental measure in the Baltic. Final report project 2181, BalticSea 2020 Foundation. www.balticsea2020.org
McFadden L, Schernewski G (2014) Critical reflections on a systems approach application in practice: a Baltic lagoon case study. Reg Environ Chang 14:2115–2126. https://doi.org/10.1007/s10113-012-0337-y
McLaughlan C, Aldridge DC (2013) Cultivation of zebra mussels (Dreissena polymorpha) within their invaded range to improve water quality in reservoirs. Water Res 47(13):4357–4369
McMahon RF (1996) The physiological ecology of the Zebra mussel, Dreissena polymorpha, in North America and Europe. Integr Comp Biol 36(3):339–363
Newell RIE (2004) Ecosystem influences of natural and cultivated populations of suspension feeding bivalve mollusks: a review. J Shellfish Res 23:51–61
Nowak P, Steinhardt T, von Ammon U, Rohde H, Schoor A, Holzhausen A, Schaible R, Schubert H (2008) Diaspore bank analysis of Baltic coastal waters. Botany Letters 165(1):159–173
Pastuszak M, Bryhn A, Hakanson L, Stalnacke P, Zalewski M, Wodzinowski T (2018) Reduction of nutrient emission from polish territory to the Baltic Sea (1988-2017) confronted with real environmental needs and international requirements. Oceanol Hydrobiol Stud 47(2):140–166
Petersen JK, Timmermann K, Carlsson M, Holmer M, Maar M, Lindahl O (2012) Mussel farming can be used as a mitigation tool–a reply. Mar Pollut Bull 64:452–454
Petersen JK, Hasler B, Timmermann K, Nielsen P, Tørring DB, Larsen MM, Holmer M (2014) Mussels as a tool for mitigation of nutrients in the marine environment. Mar Pollut Bull 82:137–143
Pollet T, Cloutier O, Nozais C, McKindsey CW, Archambault P (2015) Metabolic activity and functional diversity changes in sediment prokaryotic communities organically enriched with mussel biodeposits. PLoS. https://doi.org/10.1371/journal.pone.0123681
Radziejewska T, Schernewski G (2008) The Szczecin (Oder-) Lagoon. In: Schiewer U (ed) Ecology of Baltic coastal waters. Springer, Berlin, pp 115–129
Radziejewska T, Fenske C, Wawrzyniak-Wydrowska B, Riel P, Woźniczka A, Gruszka P (2009) The zebra mussel (Dreissena polymorpha) and the benthic community in a coastal Baltic lagoon: another example of enhancement? Mar Ecol 30:138–150
Schadach M (2013) Muschelfarmen im Stettiner Haff. Masterarbeit an der Universität Rostock. 81p
Schernewski G (2008) First steps towards an implementation of coastal management: from theory to regional practise. Rostock Meeresbiolog Beitr 19:131–148
Schernewski G, Löser N, Sekscinska A (2005) Integrated Coastal Area and River basin Management (ICARM) - the Oder/Odra case study. Coastline Reports 6, EUCC - The Coastal Union, Leiden, ISSN 0928-2734: 43–54. http://www.eucc-d.de/coastline-reports-6-2005.html
Schernewski G, Behrendt H, Neumann T (2008) An integrated river basin-coast-sea modelling scenario for nitrogen management in coastal waters. J Coast Conserv 12:53–66
Schernewski G, Neumann T, Opitz D, Venohr M (2011a) Long-term eutrophication history and functional changes in a large Baltic river basin - estuarine system. Transitional Water Bulletin 5(2):95–123. https://doi.org/10.1285/i1825229Xv5n2p95
Schernewski G, Neumann T, Maack S, Venohr M (2011b) Gewässereutrophierung – vom globalen Problem zum regionalen Lösungsansatz. In Fränzle O, Müller F, Schröder W (eds) Handbuch der Umweltwissenschaften - Grundlagen und Anwendung der Ökosystemforschung, Wiley –VCH Verlag 20. Erg. Lfg. 3/11: 1-20
Schernewski G, Stybel N, Neumann T (2012) Managing eutrophication: cost-effectiveness of Zebra mussel farming in the Oder (Szczecin) Lagoon. Ecology and Society 17(2):4. https://doi.org/10.5751/ES-04644-170204
Schernewski G, Friedland R, Carstens M, Hirt U, Leujak W, Nausch G, Neumann T, Petenati T, Sagert S, Wasmund N, von Weber M (2015) Implementation of European marine policy: new water quality targets for German Baltic waters. Mar Policy 51:305–321. https://doi.org/10.1016/j.marpol.2014.09.002
Schneider DW (1992) A bioenergetics model of Zebra mussel, Dreissena polymorpha, Growth in the Great Lakes. Can J Fish Aquat Sci 49:1406–1416
Schories D, Selig U, Schygula C (2006) Nutzung mariner Organismen zur Senkung der Nährstoff-Belastung in den Küstengewässern an der Deutschen Ostseeküste – Potenziale und Grenzen. Rostocker Meeresbiolog Beitr 15:87–104
Schulze-Böttcher K (2014) Potential und aktueller Stand der Kultivierung von Dreissena polymorpha zur Verbesserung des ökologischen Zustands des Usedomer Sees. Masters of Science Thesis, Landschaftsökologie und Naturschutz, University of Greifswald
Schumacher J, Schernewski G, Bielecka M, Loizidou XI, Loizidou M (2018) Methodologies to support coastal management - a participation and preference tool and its application. Mar Policy 94:150–157
Støttrup JG, Dinesen GE, Janßen H, Gillgren C, Schernewski G (2017) Re-visiting ICM theory and practice: lessons learned from the Baltic Sea region. Ocean Coast Manag 139:64–76
Stybel N, Fenske C, Schernewski G (2009) Mussel cultivation to improve water quality in the Szczecin lagoon. J Coast Res SI 56:1459–1463
Thong TN, van Deurs MA, Ravn-Jonsen L, Roth E (2013) Assessment of financial feasibility of farming blue mussel in the great belt by the “smart farm system”. IME report 15/13. Department of Environmental and Business Economics (IME), University of Southern Denmark
Wasmund N (2002) Harmful algal blooms in coastal waters of the South-Eastern Baltic Sea. In: Schernewski G, Schiewer U (eds) Baltic coastal ecosystems: structure, function and coastal zone management. CEEDES-Series, Berlin, Springer
Wojtal-Frankiewicz A, Frankiewicz P (2011) The impact of pelagic (Daphnia longispina) and benthic (Dreissena polymorpha) filter feeders on chlorophyll and nutrient concentration. Limnologica 41(3):191–200
Wolnomiejski N, Witek Z (2013) The Szczecin lagoon ecosystem: the biotic community of the great lagoon and its food web model. Versita Ltd, London, 289p
Wolnomiejski N, Woźniczka A (2008) A drastic reduction in abundance of Dreissena polymorpha pall in the Skoszewska cove (Szczecin Lagoon, River Odra estuary): effects in the population and habitat. Ecological Questions 9(9):103–111
Woźniczka A, Wawrzyniak-Wydrowska B, Radziejewska T, Skrzypacz A (2016) The quagga mussel (Dreissena rostriformis bugensis Andrusov, 1897)–another Ponto-Caspian dreissenid bivalve in the southern Baltic catchment: the first record from the Szczecin Lagoon. Oceanologia 58(2):154–159
Zaiko A, Daunys D, Olenin S (2009) Habitat engineering by the invasive zebra mussel Dreissena polymorpha (Pallas) in a boreal coastal lagoon: impact on biodiversity. Helgol Mar Res 63:85–94
Acknowledgements
We like to thank Jesko Hirschfeld, Luca Meyers, Simon Paysen and Nardine Stybel for supporting the work or the graphical visualization or for providing data and information; Aurelija Samuilovienė for help with molecular laboratory analyses. The work was financially supported by BONUS BaltCoast project. BONUS BaltCoast has received funding from BONUS (Art 185) funded jointly from the European Union’s Seventh Programme for research, technological development and demonstration, and from Baltic Sea national funding institutions (BMBF 03F0717A). Model simulations were supported by project MOSSCO-Synthese (BMBF 03F0740B).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Schernewski, G., Friedland, R., Buer, AL. et al. Ecological-social-economic assessment of zebra-mussel cultivation scenarios for the Oder (Szczecin) Lagoon. J Coast Conserv 23, 913–929 (2019). https://doi.org/10.1007/s11852-018-0649-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11852-018-0649-2