Abstract
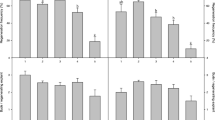
An efficient method for the generation of adventitious shoots from the cotyledons of Japanese pear and related species was developed. Cotyledons from seeds of the Japanese pear “Okusankichi” and the Asian pea pear “Hokushimamenashi” were used to determine the optimum concentrations of phytohormones in the medium. The rates of generation of adventitious shoots and the numbers of adventitious shoots per explant were highest when the media contained 5 μM 1-naphthaleneacetic acid combined with 10 or 25 μM 6-benzylaminopurine. These growth regulators were used to generate adventitious shoots from the cotyledons of 33 cultivars of Japanese, Chinese, Asian pea, and European pears. A high number of adventitious shoots per explant (1.3–2.3) and high rates of regeneration of adventitious shoots (60–76 %) were obtained from the cotyledons of Japanese pears “Imamuraaki” and “Agenosho Shinanashi.” Although pollination was not controlled, cotyledons from mother trees of old Japanese cultivars and Chinese pears tended to be more regenerable than those from other pear species. Since the rooting ability of the adventitious shoots was very low, micro-grafting was applied to obtain regenerated potted plants from adventitious shoots. Grafted regenerated plants were recovered at a rate of more than 60 %, regardless of cultivar. To our knowledge, this is the first report to evaluate shoot regeneration from cotyledons of major Pyrus species.


Similar content being viewed by others
References
Bell RL (1990) Pears (Pyrus). In: Moore JN, Ballington JR Jr (eds) Genetic resources of temperate fruit and nut crops I. International Society for Horticultural Science, Wageningen, pp 655–697
Bell RL, Quamme HA, Layne REC, Skirvin RM (1996) Pears. In: Janick J, Moore JN (eds) Fruit breeding, vol I: tree and tropical fruits. Wiley, London, pp 441–514
Bilyeu KD, Cole JL, Laskey JG, Riekhof WR, Esparza TJ, Kramer MD, Morris RO (2001) Molecular and biochemical characterization of a cytokinin oxidase from maize. Plant Physiol 125:378–386
Caboni E, Tonelli MG, Lauri P, D’Angeli S, Damiano C (1999) In vitro shoot regeneration from leaves of wild pear. Plant Cell Tissue Organ Cult 59:1–7
Casanova E, Valdés AE, Fernández B, Moysset L, Trillas MI (2004) Levels and immunolocalization of endogenous cytokinins in thidiazuron-induced shoot organogenesis in carnation. J Plant Physiol 161:95–104
Chevreau E, Skirvin RM, Abu-Qaoud HA, Korban SS, Sullivan JG (1989) Adventitious shoot regeneration from leaf tissue of three pear (Pyrus sp.) cultivars in vitro. Plant Cell Rep 7:688–691
Gao M, Murayama H, Matsuda N, Isuzugawa K, Dandekar AM, Nakano H (2002) Development of Agrobacterium-mediated transformation of pear (Pyrus communis L.) with cotyledon explants and production of transgenic pears using ACC oxidase cDNA. Plant Biotechnol 19:319–327
Hennayake CK, Dissanayake K, Matsuda N, Takasaki T, Nakanishi T (2003) An efficient and reproducible in vitro plant regeneration from leaf discs in pear cultivars (Pyrus spp.). Plant Biotechnol 20:283–289
Kaneyoshi J, Wabiko H, Kobayashi S, Tsuchiya T (2001) Agrobacterium tumefaciens AKE10-mediated transformation of an Asian pea pear, Pyrus betulaefolia Bunge: host specificity of bacterial strains. Plant Cell Rep 20:622–628
Lane WD, Iketani H, Hayashi T (1998) Shoot regeneration from cultured leaves of Japanese pear (Pyrus pyrifolia). Plant Cell Tissue Organ Cult 54:9–14
Leblay C, Chevreau E, Raboin LM (1991) Adventitious shoot regeneration from in vitro leaves of several pear cultivars (Pyrus communis L.). Plant Cell Tissue Organ Cult 25:99–105
Matsuda N, Gao M, Isuzugawa K, Takashina T, Nishimura K (2005) Development of an Agrobacterium-mediated transformation method for pear (Pyrus communis L.) with leaf-section and axillary shoot-meristem explants. Plant Cell Rep 24:45–51
Mourgues F, Chevreau E, Lambert C, de Bondt A (1996) Efficient Agrobacterium-mediated transformation and recovery of transgenic plants from pear (Pyrus communis L.). Plant Cell Rep 16:245–249
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Pérez-Clemente RM, Pérez-Sanjuán A, García-Férriz L, Beltrán JP, Cañas LA (2004) Transgenic peach plants (Prunus persica L.) produced by genetic transformation of embryo sections using the green fluorescent protein (GFP) as an in vivo marker. Mol Breed 14:419–427
Acknowledgments
We thank Dr. H. Iketani for valuable discussion and suggestions, Dr. M. Yamada for statistical analysis, and Mses. T. Akashi, M. Kimura, M. Oyama, and M. Kimura for their technical help. This work was supported in part by a Grant-in-Aid from the National Agriculture and Food Research Organization (NARO), Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Jude Grosser
Rights and permissions
About this article
Cite this article
Nakajima, I., Ito, A., Moriya, S. et al. Adventitious shoot regeneration in cotyledons from Japanese pear and related species. In Vitro Cell.Dev.Biol.-Plant 48, 396–402 (2012). https://doi.org/10.1007/s11627-012-9451-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-012-9451-2