Abstract
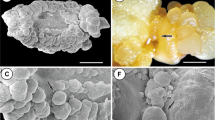
A liquid paraffin overlay (LPO) was used for storage of rapidly multiplying somatic embryos of Dendrocalamus hamiltonii under slow-growth conditions. Slow growth was associated with changes in sugar metabolism. In rapidly growing embryogenic tissues, a sharp decline in starch and non-reducing sugars indicated rapid utilization of starch. In contrast, under slow-growth conditions in somatic embryos stored under LPO, a gradual decline in starch indicated its slower utilization. As a result, growth of somatic embryos under LPO was suppressed and subculturing was not required. Following retrieval from growth under LPO after 30, 90, 180, 270, and 365 d of storage, the somatic embryos showed high recovery and germination (79.78%, 77.49%, 71.22%, 67.13%, and 59.99%, respectively) and were able to proliferate following transfer to Murashige and Skoog’s (Physiol. Plant. 15:473–497, 1962) medium containing 1 mgl−1 BAP and 2% sucrose. The study provides useful information on in vitro storage of embryogenic tissue of D. hamiltonii.


Similar content being viewed by others
References
Adams C. A.; Rinnie R. W.; Fjerstad M. C. Starch deposition and carbohydrates activity in developing and germinating soybean seeds. Ann. Bot. 45: 577–582; 1980.
Augereau J. M.; Courtois D.; Petiard V. Long term storage of callus at low temperatures or under mineral oil layer. Plant Cell Rep. 5: 372–376; 1986.
Barclay A. M.; Carwford R. M. M. The effect of anaerobiosis on carbohydrate levels in storage tissues of wetland plants. Ann. Bot. 51: 255–259; 1983.
Bhattacharya A.; Saini U.; Sharma P.; Nagar P. K.; Ahuja P. S. Osmotin-regulated reserve accumulation and germination in genetically transformed tea somatic embryos: a step towards regulation of stress tolerance and seed recalcitrance. Seed Sci. Res. 16: 203–211; 2006.
Caplin S. M. Mineral oil overlay for conservation of plant tissue cultures. Am. J. Bot. 46: 324–329; 1959.
Cervelli R.; Seneranta T. Economic aspects of somatic embryogenesis. In: Aitken J. M.; Kozai T.; Smith M. A. (eds) Automation and environmental control in plant tissue culture. Kluwer, Dordrecht, The Netherlands, pp 29–64; 1995.
Chambers S. M.; Heuch J. H. R.; Pirrie A. Micropropagation and in vitro flowering of bamboo Dendrocalamus hamiltonii Munro. Plant Cell Tiss. Org. Cult. 27: 45–48; 1991.
Dubois M.; Gilles K. A.; Hamilton J. K.; Rebers P. A.; Smith F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 28: 350–356; 1956.
Godbole S.; Sood A.; Sharma M.; Nagar P. K.; Ahuja P. S. Starch deposition and amylase accumulation during somatic embryogenesis in bamboo (Dendrocalamus hamiltonii). J. Plant Physiol. 161: 245–248; 2004.
Godbole S.; Sood A.; Thakur R.; Sharma M.; Ahuja P. S. Somatic embryogenesis and its conversion into plantlets in a multipurpose bamboo Dendrocalamus hamiltonii Nees et Arn. Ex. Munro. Curr. Sci. 83: 885–889; 2002.
Harding K. The methylation status of DNA derived from patato plants recovered from slow growth. Plant Cell Tiss. Org. Cult. 37: 31–38; 1994.
Jang J. C.; Zhou P. L.; Sheen J. Hexokinase as a sugar sensor in higher plants. Plant Cell 9: 5–19; 1997.
Kushwaha R.; Pandey S.; Chanda S.; Bhattacharya A.; Ahuja P. S. GA3 induced changes in slow growing endangered Himalayan plant Podophyllum hexandrum and hastening of vegetative growth. Plant Growth Regul. 51: 207–215; 2007.
Miller G. L. Use of dinitrosalicylic acid reagent for the determination of reducing sugar. Anal. Chem. 31: 426–428; 1959.
Moriguchi T.; Kozaki I.; Matsuta N.; Yamaki S. Plant regeneration from grape callus under a combination of low temperature and silicone treatment. Plant Cell Tiss. Org. Cult. 15: 67–71; 1988.
Murashige T.; Skoog F. A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol. Plant. 15: 473–497; 1962.
Ogita S. Callus and cell suspension culture of bamboo plant, Phyllostachys nigra. Plant Biotehnol. 22: 119–125; 2005.
Rawyler A.; Arpagaus S.; Braendle R. Impact of oxygen stress and energy availability on membrane stability of plant cells. Ann. Bot. 90: 499–507; 2002.
Sharma YML (1980) Bamboo research in Asia Pacific region. In: Lessard G, Chouinard A (eds) Proceedings of a workshop on bamboo research in Asia, Singapore, International Development Research Centre, Ottawa, Canada, pp. 99–120
Sood A.; Ahuja P. S.; Sharma M.; Sharma O. P.; Godbole S. In vitro protocols and field performance of elites of an important bamboo Dendrocalamus hamiltonii Nees et Arn. Ex. Munro. Plant Cell Tiss. Org. Cult. 71: 55–63; 2002a.
Sood A.; Palni L. M. S.; Sharma M.; Chand G.; Sharma O. P. Micropropagation of Dendrocalamus hamiltonii Munro (Magar Bamboo) using explants taken from seed-raised and field-tested plants. J. Plant Biol. 29(2): 125–132; 2002b.
Sutton B. C. S.; Polonenko D. R. Commercialization of plant somatic embryogenesis. In: Jain S. M.; Gupta P. K.; Newton R. J. (eds) Somatic embryogenesis in woody plants, forestry sciences, vol. 4. Kulwer, Dordrecht, The Netherlands, pp 263–291; 1999.
Withers L. A. In vitro conservation. Biol. J. Linn. Soc. 43: 31–42; 1991.
Acknowledgments
The authors gratefully acknowledge the Council of Scientific and Industrial Research and the Director, Dr. P S Ahuja, for facilitating the entire work. The financial assistance provided by the Department of Biotechnology, Govt. of India is gratefully acknowledged by the authors. The authors also thank Dr. R D Singh for his help in statistical analyses of the results.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Finer
IHBT Publication No.: 0837
Rights and permissions
About this article
Cite this article
Kaur, D., Ogra, R.K., Bhattacharya, A. et al. Changes in sugar levels during slow growth of Dendrocalamus hamiltonii somatic embryos due to liquid paraffin overlay. In Vitro Cell.Dev.Biol.-Plant 48, 120–126 (2012). https://doi.org/10.1007/s11627-011-9392-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-011-9392-1