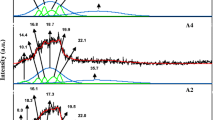
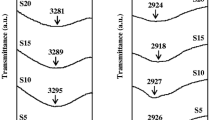
Abstract
A proton-conducting polymer electrolyte based on starch and ammonium nitrate (NH4NO3) has been prepared through solution casting method. Ionic conductivity for the system was conducted over a wide range of frequency between 50 Hz and 1 MHz and at temperatures between 303 K and 373 K. Impedance analysis shows that sample with 25 wt.% NH4NO3 has a smaller bulk resistance (R b) compared to that of the pure sample. The amount of NH4NO3 was found to influence the proton conduction; the highest obtainable room temperature conductivity was 2.83 × 10−5 S cm−1, while at 100 °C, the conductivity in found to be 2.09 × 10−4 S cm−1. The dielectric analysis demonstrates a non-Debye behavior. Transport parameters of the samples were calculated using the Rice and Roth model and thus shows that the increase in conductivity is due to the increase in the number of mobile ions.








Similar content being viewed by others
References
Riess I (2000) Polymeric mixed ionic electronic conductors. Solid State Ionics 136–137:1119–1130
Scoenenberger C, Le Nest JF, Gandini A (1995) Polymer electrolytes based on modified polysaccharides. 2. Polyether-modified cellulosics. Electrochim Acta 40:2281–2284
Morales PV, Le Nest JF, Gandini A (1998) Polymer electrolytes derived from chitosan/polyether networks. Electrochim Acta 43:1275–1279
Tambelli CE, Donoso JP, Regiani AM, Pawlicka A, Gandini A, LeNest JF (2001) Nuclear magnetic resonance and conductivity study of HEC/Polyether-based polymer electrolytes. Electrochim Acta 46:1665–1672
Lenz RW (1993) Biodegradable polymers. Adv Polym Sci 107:1–40
Avella M, Vlieger JDJ, Errico MA, Fischer S, Vacca P, Volpe MG (2005) Biodegradable starch/clay nanocomposite films for food packaging applications. Food Chem 93:467–474
Biswas A, Willet JL, Gordon SH, Finkenstadt VL, Cheng HN (2006) Complexation and blending of starch, poly(acrylic acid), and poly(N-vinyl pyrrolidone). Carbohydr Polym 65:397–403
Gomes ME, Ribeiro AS, Malafaya PB, Reis RL, Cunha AM (2001) A new approach based on injection molding to produce biodegradable starch-based polymeric scaffolds: morphology, mechanical and degradation behavior. Biomaterials 22:883–889
Kirby AR, Clark SA, Parker R, Smith AC (1993) The deformation and failure behavior of wheat starch plasticized with water and polyols. J Mater Sci 28:5937–5942
Ollett AL, Parker R, Smith AC (1991) Deformation and fracture behaviour of wheat starch plasticized with glucose and water. J Mater Sci 26:1351–1356
Sen A, Bhattacharya M (2000) Residual stresses and density gradient in injection molded starch/synthetic polymer blends. Polymer 41:9177–9190
Soestvan JJG, Borger DB (1997) Structure and properties of compression-molded thermoplastic starch materials from normal and high-amylose maize starches. J Appl Polym Sci 64:631–634
Ramya CS, Selvasekarapandian S, Savitha T, Hirankumar G, Baskaran R, Bhuvaneswari MS, Angelo PC (2006) Conductivity and thermal behavior of proton conducting polymer electrolyte based on poly(N-vinyl pyrrolidone). Eur Polym J 42:2672–2677
Ramesh S, Arof AK (2001) Ionic conductivity studies of plasticized poly(vinyl chloride) polymer electrolytes. Mater Sci Eng B 85:11–15
Baskaran R, Selvasekarapandian S, Kuwata N, Kawamura J, Hattori T (2004) AC impedance, DSC and FT-IR investigations on (x)PVAc–(1 − x) PVdF blends with LiClO4. Mater Chem Phys 98:55–61
Ratner MA, Nitzan A (1989) Conductivity in polymer ionics. Dynamic disorder and correlation. Faraday Discuss Chem Soc 88:19–42
Jaipal Reddy M, Sreekanth T, Subba Rao UV (1999) Study of the plasticizer effect on A (PEO+NaYF4) polymer electrolyte and its use in an electrochemical cell. Solid State Ionics 126:55–63
Subba Reddy Ch V, Sharma AK, Narasimha Rao VVR (2003) Conductivity and discharge characteristics of polyblend (PVP + PVA + KIO3) electrolyte. J Power Sources 114:338–345
Baskaran R, Selvasekarapandian S, Hirankumar G, Bhuvaneswari GS (2004) Dielectric and conductivity relaxations in PVAc based polymer electrolytes. Ionics 10:129–134
Kopitzke RW, Linkous CA, Anderson HR, Nelson GL (2000) Conductivity and water uptake of aromatic-based proton exchange membrane electrolytes. J Electrochem Soc 147(5):1677–1681
Mellander BE, Albinsson I (1996) In: Chowdari BVR, Dissanayake MAKL, Carrem MA (eds) Solid state ionics: new developments. World Scientific, Singapore, pp 97–115
Govindaraj G, Baskaran N, Shahi KA, Monoravi P (1996) Preparation, conductivity, complex permittivity and electric modulus in AgI––Ag2O––SeO3––MoO3 glasses. Solid State Ionics 76:47–55
Qian X, Gu N, Cheng Z, Yang X, Wang EA, Dong S (2001) Impedance study of (PEO) 10LiClO4–Al2O3 composite polymer electrolyte with blocking electrodes. Electrochim Acta 46:1829–1836
Shafee EE (1996) Dielectric and conductivity relaxation in sodium carboxymethyl cellulose and its acid form. Carbohydr Polym 31:93–98
Muralidharan P, Venkateswarlu M, Satyanarayana N (2005) Acid catalyst concentration effect on structure and ion relaxation studies of Li2O–P2O5–B2O3–SiO2 glasses synthesized by sol–gel process. J Non-Cryst Solids 351:583–594
Baskaran R, Selvasekarapandian S, Kuwata N, Kawamura J, Hattori T (2006) ac impedance, DSC and FT-IR investigations on (x)PVAc–(1 − x) PVdF blends with LiClO4. Mater Chem Phys 98:55–61
Prabakar K, Narayandass SK, Mangalaraj D (2003) Dielectric and electric modulus properties of vacuum evaporated Cd0.8Zn0.2Te thin films. Mater Sci Eng B 98:225–231
Anantha PS, Hariharan K (2005) Structure and ionic transport studies of sodium borophosphate glassy system. Mater Chem Phys 89:428–437
Rice MJ, Roth WL (1972) Ionic transport in super ionic conductors: a theoretical model. J Solid State Chem 4(2):294–310
Vijayakumar M, Selvasekarapandian S, Jayanthi D, Kavitha R (2002) Effect of annealing on Li2O–TiO2–V2O5 electrolyte materials. Solid State Ionics 154–155:773–777
Majid SR, Arof AK (2005) Proton-conducting polymer electrolyte films based on chitosan acetate complexed with NH4NO3 salt. Physica B 355:78–82
Wu HCH, Sarko A (1978) The double-helical molecular structure of crystalline B-amylose. Carbohydr Res 61:7–25
Wu HCH, Sarko A (1978) The double-helical molecular structure of crystalline a-amylose. Carbohydr Res 61:27–40
Bruice PY (2006) Essential organic chemistry. Pearson Education International
Acknowledgments
A.S.A. Khiar would like to thank the Applied Material Science Lab of the Physics Department, University of Malaya for providing the impedance spectroscopy that facilitates this work. Special thanks also are attributed to Mr. Ariffin Muda for his invaluable help in carrying out some of the experiments. This work was financed by Universiti Sains Islam Malaysia project no. PPPP(G)/2007.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khiar, A.S.A., Arof, A.K. Conductivity studies of starch-based polymer electrolytes. Ionics 16, 123–129 (2010). https://doi.org/10.1007/s11581-009-0356-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-009-0356-y