Abstract
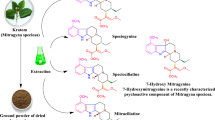
The leaves of Mitragyna speciosa, a tropical plant known as “kratom,” have been traditionally used as a substitute for opium in Thailand and Malaysia. Mitragynine, a major constituent of M. speciosa, has an opioid agonistic activity, and its derivative 7-hydroxymitragynine (7-OH-mitragynine) (a minor constituent) is much more potent than mitragynine or morphine. Recently, many products containing this plant have been distributed as “incense” on the drug market in Japan for their expected narcotic effects. Despite their potency and their wide distribution for abuse, there are no reports on the quantitative analysis of mitragynine and 7-OH-mitragynine in the raw materials or in the commercial products of kratom. In this study, a method for simultaneous analysis of mitragynine, 7-OH-mitragynine, and other indole alkaloids (speciogynine, speciociliatine, and paynantheine), present in the raw materials and commercial products of kratom, was developed using liquid chromatography-electrospray ionization mass spectrometry (LC-ESI-MS). By this method, mitragynine, 7-OH-mitragynine, and the other alkaloids were detected in 11 of the 13 products. The content of mitragynine in the products ranged from 1% to 6%, and that of 7-OH-mitragynine from 0.01% to 0.04%. Because 7-OH-mitragynine is much more potent than morphine, M. speciosa abuse is a matter of major concern. The present analytical method is considered useful for the screening of M. speciosa products in the drug market.
Similar content being viewed by others
References
Kikura-Hanajiri R, Hayashi M, Saisho K, Goda Y (2005). Simultaneous determination of 19 hallucinogenic tryptamines/β-calbolines and phenethylamines using GC-MS and LC-ESIMS. J Chromatogr B 825:29–37
Kikura-Hanajiri R, Kawamura M, Uchiyama N, Ogata J, Kamakura H, Saisho K, Goda Y (2008). Analytical data of designated substances (Shitei-Yakubutsu) controlled by the Pharmaceutical Affairs Law in Japan, part I: GC-MS and LC-MS. Yakugaku Zasshi 128:971–979
Uchiyama N, Kawamura M, Kamakura H, Kikura-Hanajiri R, Goda Y (2008). Analytical data of designated substances (Shitei-Yakubutsu) controlled by the Pharmaceutical Affairs Law in Japan, part II: color test and TLC. Yakugaku Zasshi 128:981–987
Uchiyama N, Kikura-Hanajiri R, Kawahara N, Goda Y (2008). Analysis of designer drugs detected in the products purchased in fiscal year 2006. Yakugaku Zasshi 128:1499–1505
Min JZ, Shimizu Y, Toyoóka T, Inagaki S, Kikura-Hanajiri R, Goda Y (2008). Simultaneous determination of 11 designated hallucinogenic phenethylamines by ultra-fast liquid chromatography with fluorescence detection. J Chromatogr B 873:187–194
Maruyama T, Kamakura H, Kikura-Hanajiri R, Goda Y (2008). Authentication and ultra performance liquid chromatography (UPLC)/MS analysis of magic mint, Salvia divinorum and its related plants. Yakugaku Zasshi 128:179–183
Kawamura M, Kikura-Hanajiri R, Goda Y (2008). Survey of current trends in the abuse of psychotropic plants using LCMS. Jpn J Food Chem 15:73–78
Takayama H, Aimi N, Sakai S (2000). Chemical studies on the analgesic indole alkaloids from the traditional medicine (Mitragyna speciosa) used for opium substitute. Yakugaku Zasshi 120:959–967
Takayama H (2004). Chemistry and pharmacology of analgesic indole alkaloids from the rubiaceous plant, Mitragyna speciosa. Chem Pharm Bull 52:916–928
Ponglux D, Wongseripipatana S, Takayama H, Kikuchi M, Kurihara M, Kitajima M, Aimi N, Sakai S (1994). A new indole alkaloid, 7 alpha-hydroxy-7H-mitragynine, from Mitragyna speciosa in Thailand. Planta Med 60:580–581
Matsumoto K, Mizowaki M, Suchitra T, Takayama H, Sakai S, Aimi N, Watanabe H (1996). Antinociceptive action of mitragynine in mice: evidence for the involvement of supraspinal opioid receptors. Life Sci 59:1149–1155
Thongpradichote S, Matsumoto K, Tohda M, Takayama H, Aimi N, Sakai S, Watanabe H (1998). Identification of opioid receptor subtypes in antinociceptive actions of supraspinally administered mitragynine in mice. Life Sci 62:1371–1378
Matsumoto K, Horie S, Ishikawa H, Takayama H, Aimi N, Ponglux D, Watanabe K (2004). Antinociceptive effect of 7-hydroxymitragynine in mice: discovery of an orally active opioid analgesic from the Thai medicinal herb Mitragyna speciosa. Life Sci 74:2143–2155
Horie S, Koyama F, Takayama H, Ishikawa H, Aimi N, Ponglux D, Matsumoto K, Murayama T (2005). Indole alkaloids of a Thai medicinal herb, Mitragyna speciosa, that has opioid agonistic effect in guinea-pig ileum. Planta Med 71:231–236
Matsumoto K, Horie S, Takayama H, Ishikawa H, Aimi N, Ponglux D, Murayama T, Watanabe K (2005). Antinociception, tolerance and withdrawal symptoms induced by 7-hydroxymitragynine, an alkaloid from the Thai medicinal herb Mitragyna speciosa. Life Sci 78:2–7
Matsumoto K, Hatori Y, Murayama T, Tashima K, Wongseripipatana S, Misawa K, Kitajima M, Takayama H, Horie S (2006). Involvement of mu-opioid receptors in antinociception and inhibition of gastrointestinal transit induced by 7-hydroxymitragynine, isolated from Thai herbal medicine Mitragyna speciosa. Eur J Pharmacol 549:63–70
Maruyama T, Kawamura M, Kikura-Hanajiri R, Takayama H, Goda Y (2009). The botanical origin of Kratom (Mitragyna speciosa; Rubiaceae) available as abused drugs in the Japanese markets. J Nat Med. doi:10.1007/s11418-009-0325-9
Kitajima M, Nakayama T, Kogure N, Wongseripipatana S, Takayama H (2007). New heteroyohimbine-type oxindole alkaloid from the leaves of Thai Mitragyna hirsute. J Nat Med 61:192–195
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kikura-Hanajiri, R., Kawamura, M., Maruyama, T. et al. Simultaneous analysis of mitragynine, 7-hydroxymitragynine, and other alkaloids in the psychotropic plant “kratom” (Mitragyna speciosa) by LC-ESI-MS. Forensic Toxicol 27, 67–74 (2009). https://doi.org/10.1007/s11419-009-0070-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11419-009-0070-5