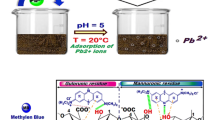
Abstract
The alginate extraction residue (RES) from the Brazilian Sargassum filipendula was successfully employed as biosorbent in this binary equilibrium study, revealing a greater affinity and selectivity for Cr(III) than for Zn(II). Experimental results also revealed that the process is of endothermic nature and well adjusted by Langmuir–Freundlich binary model. The X-ray photoelectron spectroscopy (XPS) analysis revealed that coordination with hydroxyl groups of RES prevailed in Cr removal, followed by carboxyl-metal complexation. As far as Zn(II) is concerned, ion exchange with carboxylate groups of RES was the largest contributor. Nevertheless, scanning electron microscopy coupled with Fourier transform infrared spectroscopy indicated the participation of sulfate functions in a minor degree.





Similar content being viewed by others
References
Apiratikul R, Pavasant P (2006) Sorption isotherm model for binary component sorption of copper, cadmium, and lead ions using dried green macroalga, Caulerpa lentillifera. Chem Eng J 119:135–145. https://doi.org/10.1016/j.cej.2006.02.010
Asami K, Hashimoto K (1977) The X-ray photo-electron spectra ofseveral oxides of iron and chromium. Corros Sci 17:559–570. https://doi.org/10.1016/S0010-938X(77)80002-4
Bertagnolli C, da Silva MGC, Guibal E (2014a) Chromium biosorption using the residue of alginate extraction from Sargassum filipendula. Chem Eng J 237:362–371. https://doi.org/10.1016/j.cej.2013.10.024
Bertagnolli C, Uhart A, Dupin JC, da Silva MGC, Guibal E, Desbrieres J (2014b) Biosorption of chromium by alginate extraction products from Sargassum filipendula: investigation of adsorption mechanisms using X-ray photoelectron spectroscopy analysis. Bioresour Technol 164:264–269. https://doi.org/10.1016/j.biortech.2014.04.103
Bhatnagar A, Vilar VJP, Ferreira C, Botelho CMS, Boaventura RAR (2012) Optimization of nickel biosorption by chemically modified brown macroalgae (Pelvetia canaliculata). Chem Eng J 193–194:256–266. https://doi.org/10.1016/j.cej.2012.04.037
Biesinger MC, Lau LWM, Gerson AR, Smart RSC (2010) Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Sc, Ti, V, cu and Zn. Appl Surf Sci 257:887–898. https://doi.org/10.1016/j.apsusc.2010.07.086
Cardoso SL, Costa CSD, Nishikawa E, da Silva MGC, Vieira MGA (2017) Biosorption of toxic metals using the alginate extraction residue from the brown algae Sargassum filipendula as a natural ion-exchanger. J Clean Prod 165:491–499. https://doi.org/10.1016/j.jclepro.2017.07.114
Cardoso SL, Costa CSD, da Silva MGC, Vieira MGA (2018) Dealginated seaweed waste for Zn(II) continuous removal from aqueous solution on fixed-bed column. J Chem Technol Biotechnol 93:1183–1189. https://doi.org/10.1002/jctb.5479
Chen JP, Yang L (2005) Chemical modification of Sargassum sp. for prevention of organic leaching and enhancement of uptake during metal biosorption. Ind Eng Chem Res 44:9931–9942. https://doi.org/10.1021/ie050678t
Chen JP, Yang L (2006) Study of a heavy metal biosorption onto raw and chemically modified Sargassum sp. via spectroscopic and modeling analysis. Langmuir 22:8906–8914
Chen JP, Hong L, Wu S, Wang L (2002) Elucidation of interactions between metal ions and Ca alginate-based ion-exchange resin by spectroscopic analysis and modeling simulation. Langmuir 18:9413–9421. https://doi.org/10.1021/la026060v
Chong KH, Volesky B (1995) Description of two-metal biosorption equilibria by Langmuir-type models. Biotechnol Bioeng 47:451–460. https://doi.org/10.1002/bit.260470406
Costa CSD, Cardoso SL, Nishikawa E et al (2016) Characterization of the residue from double alginate extraction from Sargassum filipendula seaweed. Chem Eng Trans 52:133–138. https://doi.org/10.3303/CET1652023
Davis TA, Volesky B, Mucci A (2003) A review of the biochemistry of heavy metal biosorption by brown algae. Water Res 37:4311–4330. https://doi.org/10.1016/S0043-1354(03)00293-8
De Andrade JR, da Silva MGC, Gimenes ML, Vieira MGA (2018) Bioadsorption of trivalent and hexavalent chromium from aqueous solutions by sericin-alginate particles produced from Bombyx mori cocoons. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-018-2651-5
Deroubaix G, Marcus P (1992) X-ray photoelectron spectroscopy analysis of copper and zinc oxides and sulphides. Surf Interface Anal 18:39–46. https://doi.org/10.1002/sia.740180107
Dupont L, Guillon E (2003) Removal of hexavalent chromium with a lignocellulosic substrate extracted from wheat bran. Environ Sci Technol 37:4235–4241. https://doi.org/10.1021/es0342345
Fagundes-klen MR, Ferri P, Martins TD et al (2007) Equilibrium study of the binary mixture of cadmium–zinc ions biosorption by the Sargassum filipendula species using adsorption isotherms models and neural network. Biochem Eng J 34:136–146. https://doi.org/10.1016/j.bej.2006.11.023
Figueira MM, Volesky B, Mathieu HJ (1999) Instrumental analysis study of iron species biosorption by Sargassum biomass. Environ Sci Technol 33:1840–1846. https://doi.org/10.1021/es981111p
Fourest E, Volesky B (1996) Contribution of sulfonate groups and alginate to heavy metal biosorption by the dry biomass of Sargassum fluitans. Environ Sci Technol 30:277–282. https://doi.org/10.1021/es950315s
Freitas GR, Vieira MGA, Silva MGC (2017) Kinetic adsorption of copper ions by the residue of alginate extraction from the seaweed Sargassum filipendula. Chem Eng Trans 57:655–660. https://doi.org/10.3303/CET1757110
Gazzoli D, Occhiuzzi M, Cimino A, Minelli G, Valigi M (1992) Chromium oxidation states and XPS analysis of the chromia/zirconia system. Surf Interface Anal 18:315–322
Greunz T, Duchaczek H, Sagl R, Duchoslav J, Steinberger R, Strauß B, Stifter D (2017) Quantification of the toxic hexavalent chromium content in an organic matrix by X-ray photoelectron spectroscopy (XPS) and ultra-low-angle microtomy (ULAM). Appl Surf Sci 396:665–671. https://doi.org/10.1016/j.apsusc.2016.11.004
Gustafsson JP (2012) Download Visual MINTEQ version 3.0. In: KTH R Inst Technol https://vminteq.lwr.kth.se/download/. Accessed 3 Feb 2018
Lima LKS, Pelosi BT, Silva MGC, Vieira MGA (2013) Lead and chromium biosorption by Pistia stratiotes biomass. Chem Eng Trans 32:1045–1050. https://doi.org/10.3303/CET1332175
Lima LKS, Silva MGC, Vieira MGA (2016) Study of binary and single biosorption by the floating aquatic macrophyte Salvinia natans. Brazilian J Chem Eng 33:649–660. https://doi.org/10.1590/0104-6632.20160333s20150483
Liu CC, Wang MK, Chiou CS, Li YS, Yang CY, Lin YA (2009) Biosorption of chromium, copper and zinc by wine-processing waste sludge: single and multi-component system study. J Hazard Mater 171:386–392. https://doi.org/10.1016/j.jhazmat.2009.06.012
Luna AS, Costa ALH, da Costa ACA, Henriques CA (2010) Competitive biosorption of cadmium(II) and zinc(II) ions from binary systems by Sargassum filipendula. Bioresour Technol 101:5104–5111. https://doi.org/10.1016/j.biortech.2010.01.138
Malamis S, Katsou E (2013) A review on zinc and nickel adsorption on natural and modified zeolite, bentonite and vermiculite: examination of process parameters, kinetics and isotherms. J Hazard Mater 252–253:428–461. https://doi.org/10.1016/j.jhazmat.2013.03.024
Mall ID, Srivastava VC, Agarwal NK, Mishra IM (2005) Adsorptive removal of malachite green dye from aqueous solution by bagasse fly ash and activated carbon-kinetic study and equilibrium isotherm analyses. Colloids Surfaces A Physicochem Eng Asp 264:17–28. https://doi.org/10.1016/j.colsurfa.2005.03.027
McCabe WL, Smith JC, Harriott P (1993) Unit Operations of Chemical Engineering 1130
McHugh DJ (1987) Production, properties and uses of alginates. In: Production and utilization of products from commercial seaweeds. FAO Fisheries Technical Paper, Rome
Moino BP, Costa CSD, da Silva MGC, Vieira MGA (2017) Removal of nickel ions on residue of alginate extraction from Sargassum filipendula seaweed in packed bed. Can J Chem Eng 95:2120–2128. https://doi.org/10.1002/cjce.22859
Moulder JF, Stickle WF, Sobol PE, Bomben KD (1992) Handbook of X-ray photoelectron spectroscopy: a reference book of standard spectra for identification and interpretation of XPS data. Perkin-Elmer Coorporation, Physical Electronics Division, Eden Prairie
Murphy V, Tofail SAM, Hughes H, McLoughlin P (2009) A novel study of hexavalent chromium detoxification by selected seaweed species using SEM-EDX and XPS analysis. Chem Eng J 148:425–433. https://doi.org/10.1016/j.cej.2008.09.029
Nishikawa E, da Silva MGC, Vieira MGA (2018) Cadmium biosorption by alginate extraction waste and process overview in life cycle assessment context. J Clean Prod 178:166–175. https://doi.org/10.1016/j.jclepro.2018.01.025
Pagnanelli F, Esposito A, Vegliò F (2002) Multi-metallic modelling for biosorption of binary systems. Water Res 36:4095–4105. https://doi.org/10.1016/S0043-1354(02)00112-4
Papageorgiou SK, Kouvelos EP, Favvas EP, Sapalidis AA, Romanos GE, Katsaros FK (2010) Metal-carboxylate interactions in metal-alginate complexes studied with FTIR spectroscopy. Carbohydr Res 345:469–473. https://doi.org/10.1016/j.carres.2009.12.010
Parab H, Joshi S, Shenoy N, Lali A, Sarma US, Sudersanan M (2006) Determination of kinetic and equilibrium parameters of the batch adsorption of Co(II), Cr(III) and Ni(II) onto coir pith. Process Biochem 41:609–615. https://doi.org/10.1016/j.procbio.2005.08.006
Pozdniakova TA, Mazur LP, Boaventura RAR, Vilar VJP (2016) Brown macro-algae as natural cation exchangers for the treatment of zinc containing wastewaters generated in the galvanizing process. J Clean Prod 119:38–49. https://doi.org/10.1016/j.jclepro.2016.02.003
Ruthven DM (1984) Ruthven _ principles of adsorption and adsorption process.Pdf. John Wiley & sons
Schiewer S, Volesky B (1996) Modeling multi-metal ion exchange in biosorption. Environ Sci Technol 30:2921–2927. https://doi.org/10.1021/es950800n
Seolatto AA, Martins TD, Bergamasco R, Tavares CRG, Cossich ES, Silva EA (2014) Biosorption study of Ni2+ and Cr3+ by Sargassum filipendula: kinetics and equilibrium. Brazilian J Chem Eng 31:211–227. https://doi.org/10.1590/S0104-66322014000100020
Sheng PX, Ting YP, Chen JP, Hong L (2004) Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: characterization of biosorptive capacity and investigation of mechanisms. J Colloid Interface Sci 275:131–141. https://doi.org/10.1016/j.jcis.2004.01.036
Silva EA, Cossich ES, Tavares CG, Cardozo Filho L, Guirardello R (2003) Biosorption of binary mixtures of Cr(III) and Cu(II) ions by Sargassum sp. Brazilian J Chem Eng 20:213–227. https://doi.org/10.1590/S0104-66322003000300002
Suzaki PYR, Munaro MT, Triques CC, Kleinübing SJ, Fagundes Klen MR, Bergamasco R, de Matos Jorge LM (2017a) Phenomenological mathematical modeling of heavy metal biosorption in fixed-bed columns. Chem Eng J 326:389–400. https://doi.org/10.1016/j.cej.2017.05.157
Suzaki PYR, Munaro MT, Triques CC, Kleinübing SJ, Klen MRF, de Matos Jorge LM, Bergamasco R (2017b) Biosorption of binary heavy metal systems: phenomenological mathematical modeling. Chem Eng J 313:364–373. https://doi.org/10.1016/j.cej.2016.12.082
Tesson B, Genet MJ, Fernandez V, Degand S, Rouxhet PG, Martin-Jézéquel V (2009) Surface chemical composition of diatoms. ChemBioChem 10:2011–2024. https://doi.org/10.1002/cbic.200800811
VanBriesen JM, Small M, Weber C, Wilson J (2010) Modelling chemical speciation: thermodynamics, kinetics and uncertainty. In: Hanrahan G (ed) Modelling of pollutants in complex environmental systems. ILM, St. Albans, pp 135–151
Yalçin S, Sezer S, Apak R (2012) Characterization and lead(II), cadmium(II), nickel(II) biosorption of dried marine brown macro algae Cystoseira barbata. Environ Sci Pollut Res 19:3118–3125. https://doi.org/10.1007/s11356-012-0807-2
Zeraatkar AK, Ahmadzadeh H, Talebi AF, Moheimani NR, McHenry MP (2016) Potential use of algae for heavy metal bioremediation, a critical review. J Environ Manag 181:817–831. https://doi.org/10.1016/j.jenvman.2016.06.059
Acknowledgements
The authors acknowledge the financial support received from CNPq (Proc. 140472/2015-0) and São Paulo Research Foundation (FAPESP) (Proc. 2014/04050-5).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Electronic supplementary material
ESM 1
(DOCX 527 kb)
Rights and permissions
About this article
Cite this article
Costa, C.S.D., Queiroz, B.G.M., Landers, R. et al. Equilibrium study of binary mixture biosorption of Cr(III) and Zn(II) by dealginated seaweed waste: investigation of adsorption mechanisms using X-ray photoelectron spectroscopy analysis. Environ Sci Pollut Res 26, 28470–28480 (2019). https://doi.org/10.1007/s11356-018-2880-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-2880-7