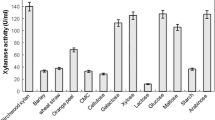
Abstract
The optimization of cultural variables resulted in a marked enhancement in the secretion of cellulase-free and alkali-thermostable xylanase (EC 3.2.1.8) by an extreme thermophile Geobacillus thermoleovorans. The enzyme secretion was enhanced when the medium was supplemented with xylan (0.15%) and Tween-80 (0.1% v/v). In wheat bran-tryptone medium, the peak in enzyme production was attained within 42 h in a fermenter as compared to 72 h in shake flasks. Optimization of the culture conditions resulted in a 7.72-fold enhancement in enzyme production. The cellulase-free xylanase was optimally active at pH 8.5 and 80°C, and it was found to be useful in the pre-bleaching process of paper pulps.








Similar content being viewed by others
References
Adamsen AK, Lindhagen J, Ahring BK (1995) Optimization of extracellular xylanase production by Dictyoglomus sp. B1 in continuous culture. Appl Microbiol Biotechnol 44:327–332
Archana A, Satyanarayana T (1997) Xylanase production by thermophilic Bacillus licheniformis A99 in solid-state fermentation. Enzyme Microb Technol 21:12–17
Archana A, Satyanarayana T (1998) Cellulase-free xylanase production by thermophilic Bacillus licheniformis A99. Indian J Microbiol 38:135–139
Babu KR, Satyanarayana T (1993) Extracellular calcium-inhibited alpha-amylase of Bacillus coagulans B49. Enzyme Microb Technol 15:1066–1069
Bailey MJ, Buchert J, Viikari L (1993) Effect of pH on production of xylanase by Trichoderma reesei on xylan and cellulose-based media. Appl Microbiol Biotechnol 40:224–229
Bartholomew WH, Karrow EO, Sfat MR, Wilhelm RH (1950) Oxygen transfer in submerged fermentations of Streptomyces griseus. Indust Eng Chem 42:1801–1809
Bastawde KB (1992) Xylan structure, microbial xylanases, and their mode of action. World J Microbiol Biotechnol 8:353–368
Beg QK, Bhushan B, Kapoor M, Hoondal G (2000) Production and characterization of thermostable xylanase and pectinase from Streptomyces sp. QG-11-3. J Indust Microbiol Biotechnol 24:396–402
Biely P, Hirch J, la Grange DC, Van Zyl WH, Prior BA (2000) A chromogenic substrate for β-xylosidase-coupled assay of α-glucuronidase. Ann Biochem 286:289–294
Breccia JD, Sifieriz F, Baigori MD, Castro GR, Hatti-Kaul R (1998) Purification and characterization of a thermostable xylanase from Bacillus amyloliquefaciens. Enzyme Microb Technol 22:42–49
Castenholz RW (1979) In: Shilo M (ed) Strategies of microbial life in extreme environments. Science Research Report 13. Dahlem Konferenzen, Berlin, pp 373–392
Damiano VB, Bocchini DA, Gomes E, Da Silva R (2003) Application of crude xylanase from Bacillus licheniformis 77-2 to the bleaching of eucalyptus Kraft pulp. World J Microbiol Biotechnol 19:139–144
Demain ACR (1972) Influence of environment on the control of enzyme synthesis. J Appl Chem Biotechnol 22:245–259
Dhillon A, Gupta JK, Khanna S (2000) Enhanced production, purification and characterization of a novel cellulase-poor, thermostable alkalitolerant xylanase from Bacillus circulans AB16. Process Biochem 35:210–215
Esteban R, Villanueva JR, Villa TG (1982) β-D-xylanases of Bacillus circulans WL-12. Can J Microbiol 28:733–739
Gomes DJ, Gomes J, Steiner W (1994) Factors influencing the induction of endoxylanases by Thermoascus aurantiacus. J Biotechnol 33:87–94
Gruninger H, Fiechter A (1986) A novel highly thermostable D-xylanase. Enzyme Microb Technol 8:309–314
Gübitz GM, Haltrich D, Latal B, Steiner W (1997) Mode of depolymerization of hemicellulose by various mannanases and xylanases in relation to their ability to bleach softwood pulp. Appl Microbiol Biotechnol 47:658–662
Humphrey A (1998) Shake-flask to fermenter, what have we learnt? Biotechnol Prog 14:3–7
Illias M, Hoq MM (1998) Effect of agitation rate on growth and production of cellulase-free xylanase by Thermomyces lanuginosus MH4 in a bioreactor. World J Microbiol Biotechnol 14:765–767
Kamra P, Satyanarayana T (2004) Xylanase production by the thermophilic mold Humicola lanuginosa in solid-state fermentation. Appl Biochem Biotechnol 119:145–157
Kantelinen A, Rantanen T, Buchert J, Vikari L (1993) Enzymatic solubilization of fiber-bound and isolated birch xylans. J Biotechnol 28:219–228
Karlsson EN, Dahlberg L, Jorto N Gorton L, Holst H (1998) Enzymatic specificity and hydrolysis pattern of the catalytic domain of the xylanase (Xynl) from Rhodothermus marinus. J Biotechnol 60:23–35
Kaupinen T, Teschke K, Savela. A, Kogevinas M, Boffetta P (1997) International data base of exposure measurement in the pulp, paper and paper product industries. Int Arch Occup Environ Health 70:119–127
Kaur P, Satyanarayana T (2004) Production and saccharification by a thermostable and neutral glucoamylase of thermophilic mould Thermomucor indicae-seudaticae. World J Microbiol Biotechnol 20:419–425
Khasin A, Alchanati I, Shoham Y (1993) Purification and characterization of a thermostable xylanase from Bacillus stearothermophilus T-6. Appl Environ Microbiol 59:1725–1730
Kulkarni N, Rao M (1996) Application of xylanases from alkaliphilic thermophilic Bacillus sp. NCIM 59: biobleaching of baggasse pulp. J Biotechnol 13:151–172
Kulkarni N, Shendye A, Rao M (1999) Molecular and Biological aspects of xylanases. FEMS Microbiol Rev 23:411–456
Levin L, Forschiassin F (1998) Influence of growth conditions on the production of xylanolytic enzymes by Trametes trogii. World J Microbiol Biotechnol 14:443–446
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin-phenol reagent. J Biol Chem 193:265–275
Mamo G, Gessesse A (2000) Immobilization and alkaliphilic Bacillus sp. cells for xylanase production using batch and continuous culture. Appl Biochem Biotechnol 87:95–101
Mandels M, Hontz L, Nystrom J (1974) Enzymatic hydrolysis of waste cellulose. Biotechnol Bioeng 16:1471–1493
Marques S, Alves L, Ribeiros GE, Collaco MTA (1998) Characterization of a thermotolerant and alkalitolerant xylanase from a Bacillus sp. Appl Biochem Biotechnol 73:159–171
Mathrani IM, Ahring BK (1992) Thermophilic and alkaliphilic xylanase from several Dictyoglomus isolates. Appl Microbiol Biotechnol 38:23–27
Meyrath J, Suchanek G (1972) Inoculation techniques–effects due to quality and quantity of inoculum. In: Norris Jr, Ribbons DW (eds) Methods in microbiology, vol 7B. Academic Press, London, pp 159–209
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugars. Anal Chem 31:426–428
Moon SH, Parulekar SJ (1991) A parametric study of protease production in batch and fed batch cultures of Bacillus firmus. Biotechnol Bioeng 37:467–483
Narang S, Satyanarayana T (2001) Thermostable α-amylase production by an extreme thermophile Bacillus thermoleovorans. Lett Appl Microbiol 32:31–35
Okazaki W, Akiba T, Horikoshi K, Akahoshi R (1985) Purification and characterization of xylanases from alkaliphilic thermophilic Bacillus spp. Agric Biol Chem 49:2033–2039
Prema P (2005) Xylanases. In: Pandey A, Webb C, Soccol CR, Larroche C (eds) Enzyme technology. Asiatech Publ, New Delhi, pp 333–346
Priest FG (1977) Extracellular enzyme synthesis in the genus Bacillus. Bacteriol Rev 41:711–753
Saraswat V, Bisaria VS (1997) Biosynthesis of xylanolytic and xylan-debranching enzymes in Melanocarpus albomyces 11S 68. J Ferment Bioeng 83:352–357
Sen S, Satyanarayana T (1993) Optimization of alkaline protease production by thermophilic Bacillus licheniformis S-40. Indian J Microbiol 33:43–47
Shaku M, Koike S, Udaka S (1980) Cultural conditions for protein production by Bacillus brevis No. 47. Agric Biol Chem 44:99–103
Sunna A, Prowe SG, Stoffregen T, Antranikian G (1997) Characterization of the xylanases from the newly isolated thermophilic xylan degrading Bacillus thermoleovorans strain K-3d and Bacillus flavothermus strain LB3A. FEMS Microbiol Lett 148:209–216
TAPPI (1992) Kappa number of pulp. T-236 CM-85. In: TAPPI Test Methods (1992–93). Technical Association of the Pulp and Paper Industry, Atlanta
Techapun C, Poosaran N, Watanabe M, Sasaki K (2003) Thermostable and alkaline-tolerant microbial cellulase-free xylanases produced from agricultural wastes and the properties required for use in pulp bleaching bioprocesses: a review. Process Biochem 38:1327–1340
Torres AL, Roncero MB, Colom JF, Pastor FIJ, Blanco A, Vidal T (2000) Effect of a novel enzyme on fibre morphology during ECF bleaching of oxygen delignified Eucalyptus kraft pulps. Bioresource Technology 74:135–140
Tuohy MG, Buckley RJ, Griffin TO, Connelly IC, Shanley NA, Filho EXF (1989) Enzyme production by solid-state cultures of aerobic fungi on lignocellulosic substrates. In: Coughlan MP (ed) Enzyme systems for lignocellulose degradation. Elsevier, London, pp 293–312
Uma Maheswar Rao JL, Satyanarayana T (2003) Enhanced secretion and low temperature stabilization of a hyperthermostable and Ca2+-independent α-amylase of Geobacillus thermoleovorans by surfactants. Lett Appl Microbiol 36:191–196
Viikari L, Ranua M, Kantelinen A, Sunduist J, Linko M (1986) Bleaching with enzymes. In: Proc 3rd Int Conf Biotechnology in the Pulp and Paper Industry. STFI, Stockholm, Sweden, pp 67–69
Vyas P, Chauthaiwale V, Phadatare S, Deshpande V, Srinivasan MC (1990) Studies on the alkaliphilic Streptomyces with extracellular xylanolytic activity. Biotechnol Lett 12:225–228
Yin YL, Baidoo SK, Jin LZ, Liu YG, Schulze H, Simmins PH (2001) The effect of different carbohydrate and protease supplementation on apparent (ideal and overall) digestibility of nutrients of five hulless barley varieties in young pigs. Livest Prod Sci 71:109–120
Acknowledgements
AS and SK are grateful to the University Grants Commission (UGC), Government of India for providing financial assistance during the course of this investigation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, A., Adhikari, S. & Satyanarayana, T. Alkali-thermostable and cellulase-free xylanase production by an extreme thermophile Geobacillus thermoleovorans . World J Microbiol Biotechnol 23, 483–490 (2007). https://doi.org/10.1007/s11274-006-9250-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-006-9250-1