Abstract
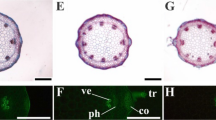
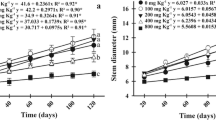
Copper (Cu) plays crucial biochemical and physiological functions in plants. However, at high concentrations, Cu can become extremely toxic, inducing several physiological changes and symptoms of toxicity. The aim was to analyze the anatomical and physiological changes of the bromeliad, Billbergia zebrina, in response to Cu excess under in vitro conditions. B. zebrina plants, previously established in vitro, were transferred to culture media containing 0, 2, 20, or 200 µM Cu. Growth traits and enzyme activity of superoxide dismutase and ascorbate peroxidase were measured over a period of 80 days. To verify the biological activity of roots grown under different Cu concentrations, a biospeckle laser was used at 20 and 80 days. Biomass accumulation, Cu content and anatomical analyses were performed at 80 days. Plants did not show any visible signs of disturbance and all plants survived the experimental treatments. Plants grown with 200 µM Cu showed anatomical changes, such as increased stomatal index and thicker exodermal cell walls of the roots, which may aid in heavy metal tolerance. High biological activity was observed only at 20 days. This biological activity was linked to increased thickness of the cell wall at the exodermis. The Cu content evidenced that the anatomical changes were effective against the transport of excess Cu into the plants. Although Cu exposure affected leaf and root anatomy as well as growth, exposure only induced minimal changes in the antioxidant system. B. zebrina tolerates high amounts of Cu and could potentially be used as a bioindicator species.












Similar content being viewed by others
References
Abbruzzese G, Beritognolo I, Muleo R, Piazzai M, Sabatti M, Mugnozza GS, Kuzminsky E (2009) Leaf morphological plasticity and stomatal conductance in three Populus alba L. genotypes subjected to salt stress. Environ Exp Bot 66:381–388. doi:10.1016/j.envexpbot.2009.04.008
Alves E, Perina FJ (2012) Apostila do curso introdutório à microscopia eletrônica de varredura e microanálise de raios-X. UFLA/FAEPE, Lavras, p 63
Alves ES, Moura BB, Domingos M (2008) Structural analysis of Tillandsia usneoides L. exposed to air pollutants in São Paulo city—Brazil. Water Air Soil Pollut 189:61–68. doi:10.1007/s11270-007-9555-1
Amado-Filho GM, Andrade LR, Farina M, Malm O (2002) Hg localisation in Tillandsia usneoides L. (Bromeliaceae), an atmospheric biomonitor. Atmos Environ 36:881–887. doi:10.1016/S1352-2310(01)00496-4
Bermudez GMA, Pignata ML (2011) Antioxidant response of three Tillandsia species transplanted to urban, agricultural, and industrial areas. Arch Environ Contam Toxicol 61:401–413. doi:10.1007/s00244-010-9642-y
Braga RA, Nobre CMB, Costa AG, Sáfadi T, Costa FM (2011) Evaluation of activity through dynamic laser speckle using the absolute value of the differences. Opt Commun 284:646–650. doi:10.1016/j.optcom.2010.09.064
Brundrett MC, Enstone DE, Peterson CA (1988) A berberine-aniline blue fluorescent staining procedure for suberin, lignin, and callose in plant tissue. Protoplasma 146:133–142. doi:10.1007/BF01405922
Bukatsch F (1972) Benerkemgem zeir doppelfarbeing astrablausafranina. Microkosmos 61:255–256
Burkhead JL, Reynolds KAG, Abdel-Ghany SE, Cohu CM, Pilon M (2009) Copper homeostasis. New Phytol 182:799–816. doi:10.1111/j.1469-8137.2009.02846.x
Cheng H, Liu Y, Tam NFY, Wang X, Li SY, Chen GZ, Ye ZH (2010) The role of radial oxygen loss and root anatomy on zinc uptake and tolerance in mangrove seedlings. Environ Pollut 158:1189–1196. doi:10.1016/j.envpol.2010.01.025
Cheng H, Tam NFY, Wang Y, Li S, Chen G, Ye Z (2012) Effects of copper on growth, radial oxygen loss and root permeability of seedlings of the mangroves Bruguiera gymnorrhiza and Rhizophora stylosa. Plant Soil 359:255–266. doi:10.1007/s11104-012-1171-1
Cutter EG (1969) Plant anatomy: experiment and interpretation. Part I. Cells and tissues. Addison, Wesley, London
Delmail D, Labrousse P, Hourdin P, Larcher L, Moesch C, Botineau M (2011) Differential responses of Myriophyllum alterniflorum DC (Haloragaceae) organs to copper: physiological and developmental approaches. Hydrobiologia 664:95–105. doi:10.1007/s10750-010-0589-9
Deng H, Ye ZH, Wong MH (2009) Lead, zinc and iron (Fe2+) tolerance in wetland plants and relation to root anatomy and spatial pattern of ROL. Environ Exp Bot 65:353–362. doi:10.1016/j.envexpbot.2008.10.005
Eapen S, D’Souza SF (2005) Prospects of genetic engineering of plants for phytoremediation of toxic metals. Biotechnol Adv 23:97–114. doi:10.1016/j.biotechadv.2004.10.001
Elias C, Fernandes EAN, França EJ, Bacchi MA, Tagliaferro FS (2008) Native bromeliads as biomonitors of airborne chemical elements in a Brazilian Restinga forest. J Radioanal Nucl Chem 278:423–427. doi:10.1007/s10967-008-0813-5
Enstone DE, Peterson CA, Ma F (2003) Root endodermis and exodermis: structure, function, and responses to the environment. J Plant Growth Regul 21:335–351. doi:10.1007/s00344-003-0002-2
Figueiredo AMG, Nogueira CA, Saiki M, Milian FM, Domingos M (2007) Assessment of atmospheric metallic pollution in the metropolitan region of São Paulo, Brazil, employing Tillandsia usneoides L. as biomonitor. Environ Pollut 145:279–292. doi:10.1016/j.envpol.2006.03.010
Giampaoli P, Tresmondi F, Lima GPP, Kanashiro S, Alves ES, Domingos E, Tavares AR (2012) Analysis of tolerance to copper and zinc in Aechmea blanchetiana grown in vitro. Biol Plant 56:83–88
Giannopolitis CN, Ries SK (1977) Superoxide dismutases. Plant Physiol 59:309–314. doi:10.1104/pp.59.2.309
Gomes MP, Sa TCLL, Marques M, Martins GA, Carneiro MMLC, Soares AM (2012) Cd-tolerance markers of Pfaffia glomerata (Spreng.) Pedersen plants: anatomical and physiological features. Braz J Plant Physiol 24:293–304. doi:10.1590/S1677-04202012000400008
Gong HJ, Zhu XY, Chen KM, Wang S, Zhang C (2005) Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Sci 169:313–321. doi:10.1016/j.plantsci.2005.02.023
Gupta P, Bhatnaga AK (2015) Spatial distribution of arsenic in different leaf tissues and its effect on structure and development of stomata and trichomes in mung bean, Vigna radiata (L.) Wilczek. Environ Exp Bot 109:12–22. doi:10.1016/j.envexpbot.2014.08.001
Himelblau E, Amasino RM (2000) Delivering copper within plant cells. Curr Opin Plant Biol 3:205–210. doi:10.1016/S1369-5266(00)80066-7
Johansen DA (1940) Plant microtechnique, 2a edn. Mc Graw-Hill, New York, p 523
Kabata-Pendias A (2010) Trace elements in soils and plants, 4th edn. CRC Press, London, p 520
Khatun S, Ali MB, Hahn EJ, Paek KY (2008) Copper toxicity in Withania somnifera: growth and antioxidant enzymes responses of in vitro grown plants. Environ Exp Bot 64:279–285. doi:10.1016/j.envexpbot.2008.02.004
Kováčik J, Klejdus B, Štork F, Hedbavny J (2012) Physiological responses of Tillandsia albida (Bromeliaceae) to long-term foliar metal application. J Hazard Mat 239–240:175–182. doi:10.1016/j.jhazmat.2012.08.062
Li T, Xiong ZT (2004) A novel response of wild type duckweed (Lemna paucicostata Hegelm.) to heavy metals. Environ Toxicol 19:95–102. doi:10.1002/tox.20000
Llorens N, Arola L, Blade C, Mas A (2000) Effects of copper exposure upon nitrogen metabolism in tissue cultured Vitis vinifera. Plant Sci 160:159–163. doi:10.1016/S0168-9452(00)00379-4
Lüttge U (2010) Ability of crassulacean acid metabolism plants to overcome interacting stresses in tropical environments. AoB Plants 10:plq005. doi:10.1093/aobpla/plq005
Lux A, Martinka M, Vaculík M, White PJ (2011) Root responses to cadmium in the rhizosphere: a review. J Exp Bot 62:21–37. doi:10.1093/jxb/erq281
Mackie KA, Müller T, Kandeler E (2012) Remediation of copper in vineyards—a mini review. Environ Pollut 167:16–26. doi:10.1016/j.envpol.2012.03.023
Madejón P, Ramírez-Benítez JE, Corrales I, Barceló J, Poschenrieder C (2009) Copper-induced oxidative damage and enhanced antioxidant defenses in the root apex of maize cultivars differing in Cu tolerance. Environ Exp Bot 67:415–420. doi:10.1016/j.envexpbot.2009.08.006
Martins JPR, Schimildt ER, Alexandre RS, Castro EM, Nani TF, Pires MF, Pasqual M (2014) Direct organogenesis and leaf-anatomy modifications in vitro of Neoregelia concentrica (Vellozo) L.B. Smith (Bromeliaceae). Pak J Bot 46:2179–2187
Martins JPR, Pasqual M, Martins AD, Ribeira SF (2015a) Effects of salts and sucrose concentrations on in vitro propagation of Billbergia zebrina (Herbert) Lindley (Bromeliaceae). Aust J Crop Sci 9:85–91
Martins JPR, Verdoodt V, Pasqual M, De Proft M (2015b) Impacts of photoautotrophic and photomixotrophic conditions on in vitro propagated Billbergia zebrina (Bromeliaceae). Plant Cell Tissue Organ Cul 123:121–132. doi:10.1007/s11240-015-0820-5
McElrone AJ, Pockman WT, Matínez-Vilalta J, Jackson RB (2004) Variation in xylem structure and function in stems and roots of trees to 20 m depth. New Phytol 163:507–517. doi:10.1111/j.1469-8137.2004.01127.x
Mishra VK, Upadhyaya AR, Pandey SK, Tripathi BD (2008) Heavy metal pollution induced due to coal mining effluent on surrounding aquatic ecosystem and its management through naturally occurring aquatic macrophytes. Bioresour Technol 99:930–936. doi:10.1016/j.biortech.2007.03.010
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Nagajyoti PC, Lee KD, Sreekanth TVM (2010) Heavy metals, occurrence and toxicity for plants: a review. Environ Chem Lett 8:99–216. doi:10.1007/s10311-010-0297-8
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Neelu N, Kumar M, Tomar M, Bhatnagar AK (2000) Influence of cadmium on growth and development of Vicia faba Linn. Indian J Exp Biol 38:819–823
Nothdurft R, Yao G (2005) Imaging obscured subsurface inhomogeneity using laser speckle. Opt Express 13:10034–10039. doi:10.1364/OPEX.13.010034
Panou-Filotheou H, Bosabalidis AM (2004) Root structural aspects associated with copper toxicity in oregano (Origanum vulgare subsp. hirtum). Plant Sci 166:1497–1504. doi:10.1016/j.plantsci.2004.01.026
Panou-Filotheou H, Bosabalidis AM, Karataglis S (2001) Effects of copper toxicity on leaves of oregano (Origanum vulgare subsp. hirtum). Ann Bot 88:207–214. doi:10.1006/anbo.2001.1441
Pereira TAR, Silva LC, Azevedo AA, Francino DMT, Coser TS, Pereira JD (2013) Leaf morpho-anatomical variations in Billbergia elegans and Neoregelia mucugensis (Bromeliaceae) exposed to low and high solar radiation. Botany 91:327–334. doi:10.1139/cjb-2012-0276
Pilon M, Abdel-Ghany SE, Cohu CM, Gogolin KA, Ye H (2006) Copper cofactor delivery in plant cells. Curr Opin Plant Biol 9:256–263. doi:10.1016/j.pbi.2006.03.007
Poschenrieder C, Barceló J (1999) Water relations in heavy metal stressed plants. In: Prasad MNV, Hagemeyer J (eds) Heavy metal stress in plants. Springer, Berlin, pp 207–229. doi:10.1007/978-3-662-07745-0_10
Proença SL, Sajo MG (2008) Rhizome and root anatomy of 14 species of Bromeliaceae. Rodriguésia 59:113–128
Ribeiro KM, Braga RA Jr, Horgan GW, Ferreira DD, Sáfadi T (2014) Principal component analysis in the spectral analysis of the dynamic laser speckle patterns. J Eur Opt Soc Rapid Publ 9:14009. doi:10.2971/jeos.2014.14009
Rodríguez-Gamir J, Intrigliolo DS, Primo-Millo E, Forner-Giner MA (2010) Relationships between xylem anatomy, root hydraulic conductivity, leaf/root ratio and transpiration in citrus trees on different rootstocks. Physiol Plant 139:159–169. doi:10.1111/j.1399-3054.2010.01351.x
Scholz AK, Klepsch M, Karimi Z, Jansen S (2013) How to quantify conduits in wood? Front Plant Sci 4:1–11
Schreibera L, Frankea R, Lessire R (2005) Biochemical characterization of elongase activity in corn (Zea mays L.) roots. Phytochemistry 66:131–138. doi:10.1016/j.phytochem.2004.11.022
Segecin S, Scatena VL (2004) Morfoanatomia de rizomas e raízes de Tillandsia L. (Bromeliaceae) dos Campos Gerais, PR, Brasil. Acta Bot Bras 18:253–260. doi:10.1590/S0102-33062004000200005
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defence mechanism in plants under stressful conditions. J Bot 2012:1–26. doi:10.1155/2012/217037
Shi G, Cai Q (2009) Leaf plasticity in peanut (Arachis hypogaea L.) in response to heavy metal stress. Environ Exp Bot 67:112–117. doi:10.1016/j.envexpbot.2009.02.009
Silva IV, Scatena VL (2011) Anatomia de raízes de nove espécies de Bromeliaceae (Poales) da região amazônica do estado de Mato Grosso, Brasil. Acta Bot Bras 25:618–627
Son JA, Narayanankutty DP, Roh KS (2014) Influence of exogenous application of glutathione on rubisco and rubisco activase in heavy metal-stressed tobacco plant grown in vitro. Saudi J Biol Sci 21:89–97. doi:10.1016/j.sjbs.2013.06.002
Srighar BBM, Diehl SV, Han FX, Monts DL, Su Y (2005) Anatomical changes due to uptake and accumulation of Zn and Cd in Indian mustard (Brassica juncea). Environ Exp Bot 54:131–141. doi:10.1016/j.envexpbot.2004.06.011
Thounaojam TC, Panda P, Mazumdar P, Kumar D, Sharma GD, Sahoo L, Sanji P (2012) Excess copper induced oxidative stress and response of antioxidants in rice. Plant Physiol Biochem 53:33–39. doi:10.1016/j.plaphy.2012.01.006
Vaculík M, Konlechner C, Langer I, Adlassnig W, Puschenreiter M, Lux A, Hauser MT (2012) Root anatomy and element distribution vary between two Salix caprea isolates with different Cd accumulation capacities. Environ Pollut 163:117–126. doi:10.1016/j.envpol.2011.12.031
Van Assche F, Clijsters H (1990) Effects of metals on enzyme activity in plants. Plant Cell Environ 13:195–206. doi:10.1111/j.1365-3040.1990.tb01304.x
Wainwright SJ, Woolhouse HW (1977) Some physiological aspects of copper and zinc tolerance in Agrostis tenuis sibth: cell elongation and membrane damage. J Exp Bot 28:1029–1036. doi:10.1093/jxb/28.4.1029
Wang J, Zhang H, Allen RD (1999) Overexpression of an Arabidopsis peroxisomal ascorbate peroxidase gene in tobacco increases protection against oxidative stress. Plant Cell Physiol 40:725–732
Wannaz LED, Zygadlo JA, Pignata ML (2003) Air pollutants effect on monoterpenes composition and foliar chemical parameters in Schinus areira. Sci Total Environ 305:177–193. doi:10.1016/S0048-9697(02)00466-7
Zabalza A, Galvez L, Marino D, Royuela M, Arrese-Igor C, Gonzalez EM (2008) The application of ascorbate or its immediate precursor, galactono-1, 4-lactone, does not affect the response of nitrogen-fixing pea nodules to water stress. J Plant Physiol 165:805–812. doi:10.1016/j.jplph.2007.08.005
Zhang LP, Mehta SK, Liu ZP, Yang ZM (2008) Copper-induced proline synthesis is associated with nitric oxide generation in Chlamydomonas reinhardtii. Plant Cell Physiol 49:411–419. doi:10.1093/pcp/pcn017
Zhao FJ, Lombi E, Breedon T, McGrath SP (2000) Zinc hyperaccumulation and cellular distribution in Arabidopsis halleri. Plant Cell Environ 23:507–514. doi:10.1046/j.1365-3040.2000.00569.x
Acknowledgments
The authors would like to acknowledge the scholarship awarded by CAPES (Coordination for the Improvement of Higher Education Personnel). The authors are also grateful to Italo Antônio Fernandes, Raquel Bezerra Chiavegatto and Vânia Helena Techio for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martins, J.P.R., Martins, A.D., Pires, M.F. et al. Anatomical and physiological responses of Billbergia zebrina (Bromeliaceae) to copper excess in a controlled microenvironment. Plant Cell Tiss Organ Cult 126, 43–57 (2016). https://doi.org/10.1007/s11240-016-0975-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-0975-8