Abstract
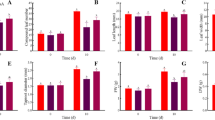
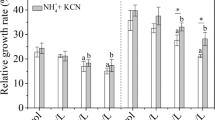
Pakchoi plants were grown in 32 mM NO3− nutrient solution with or without 2.5 mM γ-aminobutyric acid (GABA) to investigate metabolite changes, gene and protein expression levels, and the activities of key enzymes related to nitrate metabolism in the leaves over a period of 0–12 days. High-nitrogen treatment enhanced plant growth and the NO3−, NO2−, NH4+, Gln, and Glu contents in the leaves; promoted the gene and protein expression of nitrate reductase (NR) and glutamate decarboxylase (GAD); and increased the activities of NR, nitrite reductase (NiR), glutamine synthetase (GS), glutamate synthase (GOGAT), and GAD. The endogenous GABA concentration in the leaves was enhanced in parallel with the increase in GAD activity. The GABA-treated leaves displayed the greatest increases in the gene and protein expression levels of NR and GAD and in the activities of NR, NiR, GS, GOGAT, and GAD. In addition, accelerated rates of nitrate reduction and assimilation were detected, and these changes occurred concurrently with the observed increases in gene or protein expression and enzyme activity. As a result, the concentrations of NH4+, Gln, Glu, and endogenous GABA were significantly elevated, and the NO3− and NO2− contents were significantly decreased, in GABA-treated leaves compared with plants exposed to nitrogen-rich conditions. Our results reveal a potential positive that GABA may act as a nitrogen source to improve the plant growth and the most prominent effect of decreasing nitrate contents by accelerating NO3− reduction and assimilation. Exogenous GABA plays an important role in reducing the NO3− content of leaves, and thereby improves the ability to harvest leafy vegetables containing higher levels of endogenous GABA.











Similar content being viewed by others
References
Barbosa JM, Locy RD, Barger TW, Singh NK, Cherry JH (2000) GABA increases the rate of nitrate uptake and utilization in Arabidopsis roots. Nato Science 83:53–63
Barbosa JM, Singh NK, Cherry JH, Locy RD (2010) Nitrate uptake and utilization is modulated by exogenous γ-aminobutyric acid in Arabidopsis thaliana seedlings. Plant Physiol Biochem 48:443–450
Beuve N, Rispail N, Laine P, Cliquet JB, Ourry A, Le deunff E (2004) Putative role of gamma-aminobutyric acid (GABA) as a long-distance signal in up-regulation of nitrate uptake in Brassica napus L. Plant Cell Environ 27:1035–1046
Britto DT, Kronzucker HJ (2002) NH4 + toxicity in higher plants: a critical review. J Plant Physiol 159:567–584
Bryan NS, Alexander DD, Coughlin JR, Milkowski AL, Boffetta P (2012) Ingested nitrate and nitrite and stomach cancer risk: an updated review. Food Chem Toxicol l50:3646–3665
Cavaiuolo M, Ferrante A (2014) Nitrates and glucosinolates as strong determinants of the nutritional quality in rocket leafy salads. Nutrients 6:1519–1538
Chen B, Wang Z, Li S, Wang G, Song H, Wang X (2004) Effects of nitrate supply on plant growth, nitrate accumulation, metabolic nitrate concentration and nitrate reductase activity in three leafy vegetables. Plant Sci 167:635–643
Colonna E, Rouphael Y, Barbieri G, De Pascale S (2016) Nutritional quality of ten leafy vegetables harvested at two light intensities. Food Chem 199:702–710
Fait A, Fromm H, Walter D, Galili G, Fernie AR (2008) Highway or byway: the metabolic role of the GABA shunt in plants. Trends Plant Sci 13:14–19
Fan L, Wu X, Tian Z, Jia K, Pan Y, Li J, Gao H (2015) Comparative proteomic analysis of gamma-aminobutyric acid responses in hypoxia-treated and untreated melon roots. Phytochemistry 116:28–37
Gao H, Jia Y, Guo S, Lv G, Wang T, Juan L (2011) Exogenous calcium affects nitrogen metabolism in root-zone hypoxia-stressed muskmelon roots and enhances short-term hypoxia tolerance. J Plant Physiol 168:1217–1225
Gibon Y, Blaesing OE, Hannemann J, Carillo P, Höhne M, Hendriks JH, Palacios N, Cross J, Selbig J, Stitt M (2004) Robot-based platform to measure multiple enzyme activities in Arabidopsis using a set of cycling assays: comparison of changes of enzyme activities and transcript levels during diurnal cycles and in prolonged darkness. Plant Cell 16:3304–3325
Han YR, Wang XF, Yang FJ, Wei M, Shi QH, Li QM, Cui XM (2015) Effects of NO3 − stress on photosynthetic characteristics and nitrogen metabolism of strawberry seedlings. Chin J Appl Ecol 26:2314–2320
Hellmann H, Funck D, Rentsch D, Frommer WB (2000) Hypersensitivity of an Arabidopsis sugar signaling mutant toward exogenous proline application. Plant Physiol 123:779–789
Hu X, Xu Z, Xu W, Li J, Zhao N, Zhou Y (2015) Application of γ-aminobutyric acid demonstrates a protective role of polyamine and GABA metabolism in muskmelon seedlings under Ca(NO3)2 stress. Plant Physiol Biochem 92:1–10
Hyun TK, Eom SH, Jeun YC, Han SH, Kim J (2013) Identification of glutamate decarboxylases as a γ-aminobutyric acid (GABA) biosynthetic enzyme in soybean. Ind Crop Prod 49:864–870
Kusano M, Tabuchi M, Fukushima A, Funayama K, Diaz C, Kobayashi M, Hayashi N, Tsuchiya YN, Takahashi H, Kamata A, Yamaya T, Saito K (2011) Metabolomics data reveal a crucial role of cytosolic glutamine synthetase 1; 1 in coordinating metabolic balance in rice. Plant J 66:456–466
Lea PJ, Miflin BJ (2003) Glutamate synthase and the synthesis of glutamate in plants. Plant Physiol Biochem 41:555–564
Li JR, Tian Z, Wu XL, Zhang Y, Gong BB, Gao HB (2017) Cloning of GAD gene in Pakchoi(Brassica campestris ssp. chinensis) and induced expression analysis treated with exogenous GABA under higer nitrogen level. J Agric Biotechnol 25:1217–1227
Li JR, Tian Z, Wu XL, Gong BB, Gao HB (2016) Regulation of γ-aminobutyric acid on growth and nitrate metabolism of Pak-choi treated with high nitrogen application. Acta Horticulturae Sinica 43:2182–2192
Lin CC, Kao CH (1996) Disturbed ammonium assimilation is associated with growth inhibition of roots in rice seedlings caused by NaCl. Plant Growth Regul 18:233–238
Liu TT, Tseng YW, Yang TS (2016) Functionalities of conjugated compounds of γ-aminobutyric acid with salicylaldehyde or cinnamaldehyde. Food Chem 190:1102–1108
Liu X, Wang L, Li Z, Huang D (2014) Nitrate/Gly ratios in nutrition influenced the growth and amino acid composition in spinach (Spinaciaoleracea L.). J Plant Nutr 37:765–776
Lu Y, Luo F, Yang M, Li X, Lian X (2011) Suppression of glutamate synthase genes significantly affects carbon and nitrogen metabolism in rice (Oryza sativa L.). Sci China Life Sci 54:651–663
Luo JK, Sun SB, Jia LJ, Chen W, Shen QR (2006) The mechanism of nitrate accumulation in Pakchoi [Brassica campestris L. ssp. Chinensis (L.)]. Plant Soil 282:291–300
Ma X, Zhu C, Yang N, Gan L, Xia K (2016) γ-Aminobutyric acid addition alleviates ammonium toxicity by limiting ammonium accumulation in rice (Oryza sativa) seedlings. Physiol Plant 158:389–401
Masclaux-Daubresse C, Daniel-Vedele F, Dechorgnat J, Chardon F, Gaufichon L, Suzuki A (2010) Nitrogen uptake, assimilation and remobilization in plants: challenges for sustainable and productive agriculture. Ann Bot 105:1141–1157
Muller B, Touraine B (1992) Inhibition of NO3 − uptake by various phloem-translocated amino acids in soybean seedlings. J Exp Bot 43:617–623
Näsholm T, Kielland K, Ganeteg U (2009) Uptake of organic nitrogen by plants. New Phytol 182:31–48
Nazoa P, Vidmar JJ, Tranbarger TJ, Mouline K, Damiani I, Tillard P, Zhuo D, Glass AD, Touraine B (2003) Regulation of the nitrate transporter gene AtNRT2.1 in Arabidopsis thaliana: responses to nitrate, amino acids and developmental stage. Plant Mol Biol 52:689–703
Oliveira HC, Freschi L, Sodek L (2013) Nitrogen metabolism and translocation in soybean plants subjected to root oxygen deficiency. Plant Physiol Biochem 66:141–149
Persson J, Gardeström P, Näsholm T (2006) Uptake, metabolism and distribution of organic and inorganic nitrogen sources by Pinus sylvestris. J Exp Bot 57:2651–2659
Petersen A, Stoltze S (1999) Nitrate and nitrite in vegetables on the Danish market: content and intake. Food Addit Contam 16:291–299
Salvatierra A, Pimentel P, Almada R, Hinrichsen P (2016) Exogenous GABA application transiently improves the tolerance to root hypoxia on a sensitive genotype of Prunus rootstock. Environ Exp Bot 125:52–66
Scheible WR, Lauerer M, Schulze ED, Caboche M, Stitt M (1997) Accumulation of nitrate in the shoot acts as a signal to regulate shoot-root allocation in tobacco. Plant J 11:671–691
Sun F, Hou X, Li Y, Yang X (2008) Molecular cloning and characterization of nitrate reductase gene from non-heading Chinese cabbage. Sci Hortic 119:1–10
Takahashi M, Sasaki Y, Ida S, Morikawa H (2001) Nitrite reductase gene enrichment improves assimilation of NO2 in Arabidopsis. Plant Physiol 126:731–741
Takayama M, Koike S, Kusano M, Matsukura C, Saito K, Ariizumi T, Ezura H (2015) Tomato glutamate decarboxylase genes SlGAD2 and SlGAD3 play key roles in regulating γ-aminobutyric acid levels in tomato (Solanum lycopersicum). Plant Cell Physiol 56:1533–1545
Thornton B, Robinson D (2005) Uptake and assimilation of nitrogen from solutions containing multiple N sources. Plant Cell Environ 28:813–821
Wang C, Fan L, Gao H, Wu X, Li J, Lv G, Gong B (2014a) Polyamine biosynthesis and degradation are modulated by exogenous gamma-aminobutyric acid in root-zone hypoxia-stressed melon roots. Plant Physiol Biochem 82:17–26
Wang H, Wu L, Wang M, Zhu Y, Tao Q, Zhang F (2007) Effects of amino acids replacing nitrate on growth, nitrate accumulation, and macroelement concentrations in Pakchoi (Brassica chinensis L.). Pedosphere 17:595–600
Wang HF, Tsai YS, Lin ML, Ou AS (2006) Comparison of bioactive components in GABA tea and green tea produced in Taiwan. Food Chem 96:648–653
Wang X, Yu W, Zhou Q, Han R, Huang D (2014b) Metabolic response of pakchoi leaves to amino acid nitrogen. J Integr Agric 13:778–788
Williams L, Miller A (2001) Transporters responsible for the uptake and partitioning of nitrogenous solutes. Annu Rev Plant Physiol Plant Mol Biol 52:659–688
Xu G, Fan X, Miller AJ (2012) Plant nitrogen assimilation and use efficiency. Ann Rev Plant Biol 63:153–182
Acknowledgements
This research was performed by the Collaborative Innovation Center of vegetable Industry in Hebei, which is financially supported by the Nature Science Foundation of Hebei (No. C2014204074), the Key Program of Science and Technology of the Education of Department of Hebei (No. ZH2012048), and the Scientific Research Foundation for Returned Overseas Scholars of Hebei (No. 130601224).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Tian, Z., Wu, X. et al. Gamma-Aminobutyric Acid (GABA) Modulates Nitrate Concentrations and Metabolism in the Leaves of Pakchoi (Brassica campestris ssp. chinensis Makino) Treated with a Nitrogen-Rich Solution. Plant Mol Biol Rep 36, 530–542 (2018). https://doi.org/10.1007/s11105-018-1092-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-018-1092-0