Abstract
Background Prolonged use of anticholinergic and sedative medicines is correlated with worsening cognition and physical function decline. Deprescribing is a proposed intervention that can help to minimise polypharmacy whilst potentially improving several health outcomes in older people. Objective This study aimed to examine the feasibility of implementing a deprescribing intervention that utilises a patient-centred pharmacist-led intervention model; in order to address major deprescribing challenges such as general practitioner time constraints and lack of accessible deprescribing guidelines and processes. Setting Three residential care facilities. Methods The intervention involved a New Zealand registered pharmacist utilising peer-reviewed deprescribing guidelines to recommend targeted deprescribing of anticholinergic and sedative medicines to GPs. Main outcome measure The change in the participants’ Drug Burden Index (DBI) total and DBI ‘as required’ (PRN) was assessed 3 and 6 months after implementing the deprescribing intervention. Results Seventy percent of potential participants were recruited for the study (n = 46), and 72% of deprescribing recommendations suggested by the pharmacist were implemented by General Pratitioners (p = 0.01; Fisher’s exact test). Ninety-six percent of the residents agreed to the deprescribing recommendations, emphasising the importance of patient centred approach. Deprescribing resulted in a significant reduction in participants’ DBI scores by 0.34, number of falls and adverse drug reactions, 6 months post deprescribing. Moreover, participants reported lower depression scores and scored lower frailty scores 6 months after deprescribing. However, cognition did not improve; nor did participants’ reported quality of life. Conclusion This patient-centred deprescribing approach, demonstrated a high uptake of deprescribing recommendations and success rate. After 6 months, significant benefits were noted across a range of important health measures including mood, frailty, falls and reduced adverse reactions. This further supports deprescribing as a possible imperative to improve health outcomes in older adults.

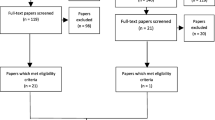
Adapted from the process outlined in reference 114 [8]

Similar content being viewed by others
References
Jansen J, Naganathan V, Carter SM, McLachlan AJ, Nickel B, Irwig L, et al. Too much medicine in older people? Deprescribing through shared decision making. BMJ (Clin Res Ed). 2016;353:i2893.
Beer C, Loh PK, Peng YG, Potter K, Millar A. A pilot randomized controlled trial of deprescribing. Ther Adv Drug Saf. 2011;2(2):37–43.
Ouellet G, Ouellet JA, Tinetti ME. Principle of rational prescribing and deprescribing in older adults with multiple chronic conditions. Ther Adv Drug Saf. 2018;9:2042098618791371.
Reeve E, Gnjidic D, Long J, Hilmer S. A systematic review of the emerging definition of ‘deprescribing’ with network analysis: implications for future research and clinical practice. Br J Clin Pharmacol. 2015;80(6):1254–68.
Garfinkel D, Mangin D. Feasibility study of a systematic approach for discontinuation of multiple medications in older adults: addressing polypharmacy. Arch Intern Med. 2010;170(18):1648–54.
Campbell AJ, Robertson MC, Gardner MM, Norton RN, Buchner DM. Psychotropic medication withdrawal and a home-based exercise program to prevent falls: a randomized, controlled trial. J Am Geriatr Soc. 1999;47(7):850–3.
Cumming RG, Le Couteur DG. Benzodiazepines and risk of hip fractures in older people: a review of the evidence. CNS Drugs. 2003;17(11):825–37.
Reeve E, Wiese MD. Benefits of deprescribing on patients’ adherence to medications. Int J Clin Pharm. 2014;36(1):26–9.
Garfinkel D, Zur-Gil S, Ben-Israel J. The war against polypharmacy: a new cost-effective geriatric-palliative approach for improving drug therapy in disabled elderly people. Isr Med Assoc J. 2007;9(6):430–4.
Ailabouni NJ, Nishtala PS, Mangin D, Tordoff JM. Challenges and enablers of deprescribing: a general practitioner perspective. PLoS ONE. 2016;11(4):e0151066.
Keehan SP, Stone DA, Poisal JA, Cuckler GA, Sisko AM, Smith SD, et al. National health expenditure projections, 2016–25: price increases, aging push sector to 20 percent of economy. Health Affairs (Millwood). 2017;36(3):553–63.
Howdon D, Rice N. Health care expenditures, age, proximity to death and morbidity: implications for an ageing population. J Health Econ. 2018;57:60–74.
Ni Chroinin D, Ni Chroinin C, Beveridge A. Factors influencing deprescribing habits among geriatricians. Age Ageing. 2015;44:704–8.
Page AT, Clifford RM, Potter K, Schwartz D, Etherton-Beer CD. The feasibility and effect of deprescribing in older adults on mortality and health: a systematic review and meta-analysis. Br J Clin Pharmacol. 2016;82(3):583–623.
Nishtala PS, Hilmer SN, McLachlan AJ, Hannan PJ, Chen TF. Impact of residential medication management reviews on drug burden index in aged-care homes: a retrospective analysis. Drugs Aging. 2009;26(8):677–86.
Koyama A, Steinman M, Ensrud K, Hillier TA, Yaffe K. Long-term cognitive and functional effects of potentially inappropriate medications in older women. J Gerontol A Biol Sci Med Sci. 2014;69(4):423–9.
Salahudeen MS, Duffull SB, Nishtala PS. Anticholinergic burden quantified by anticholinergic risk scales and adverse outcomes in older people: a systematic review. BMC Geriatr. 2015;15:31.
Kouladjian L, Gnjidic D, Chen TF, Mangoni AA, Hilmer SN. Drug Burden Index in older adults: theoretical and practical issues. Clin Interv Aging. 2014;9:1503–15.
Hilmer SN, Mager DE, Simonsick EM, Cao Y, Ling SM, Windham BG, et al. A drug burden index to define the functional burden of medications in older people. Arch Intern Med. 2007;167(8):781–7.
Ailabouni NJ, Mangin D, Nishtala PS. Deprescribing anticholinergic and sedative medicines: protocol for a Feasibility Trial (DEFEAT-polypharmacy) in residential aged care facilities. BMJ Open. 2017;7(4):e013800.
CONSORT: Transparent, Recording, of, trails. The CONSORT flow diagram 2016. http://www.consort-statement.org/consort-statement/flow-diagram. Accessed Oct 2016.
Gnjidic D, Le Couteur DG, Abernethy DR, Hilmer SN. A pilot randomized clinical trial utilizing the drug burden index to reduce exposure to anticholinergic and sedative medications in older people. Ann Pharmacother. 2010;44(11):1725–32.
Hilmer SN, Mager DE, Simonsick EM, Ling SM, Windham BG, Harris TB, et al. Drug burden index score and functional decline in older people. Am J Med. 2009;122(12):1142–9 (e1–2).
Crotty M, Rowett D, Spurling L, Giles LC, Phillips PA. Does the addition of a pharmacist transition coordinator improve evidence-based medication management and health outcomes in older adults moving from the hospital to a long-term care facility? Results of a randomized, controlled trial. Am J Geriatr Pharmacother. 2004;2(4):257–64.
InterRAI Long Term Care Facilities. The InterRAI organisation: Mission and Vision 2016. http://www.interrai.org/index.php?id=2. Accessed April 2017.
Hirdes JP, Ljunggren G, Morris JN, Frijters DH, Finne Soveri H, Gray L, et al. Reliability of the interRAI suite of assessment instruments: a 12-country study of an integrated health information system. BMC Health Serv Res. 2008;8:277.
Potter K, Flicker L, Page A, Etherton-Beer C. Deprescribing in frail older people: a randomised controlled trial. PLoS ONE. 2016;11(3):e0149984.
Edmonton Frailty Scale. https://www.nscphealth.co.uk/edmontonscale-pdf. Accessed May 2017.
Sheikh JI, Yesavage JA, Brooks JO 3rd, Friedman L, Gratzinger P, Hill RD, et al. Proposed factor structure of the Geriatric Depression Scale. Int Psychogeriatr. 1991;3(1):23–8.
Lingjærde O, Ahlfors UG, Bech P, Dencker SJ, Elgen K. The UKU side effect rating scale. A new comprehensive rating scale for psychotropic drugs and a cross-sectional study of side effects in neuroleptic-treated patients. Acta Psychatr Scand. 1987;76(334):1–100.
The EuroQol Group. EuroQol-a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199–208.
Team R Core. R: a language and environment for statistical computing 2016. http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.470.5851&rep=rep1&type=pdf. Accessed Sep 2017.
Gnjidic D, Hilmer SN, Blyth FM, Naganathan V, Waite L, Seibel MJ, et al. Polypharmacy cutoff and outcomes: five or more medicines were used to identify community-dwelling older men at risk of different adverse outcomes. J Clin Epidemiol. 2012;65(9):989–95.
Kalogianis MJ, Wimmer BC, Turner JP, Tan EC, Emery T, Robson L, et al. Are residents of aged care facilities willing to have their medications deprescribed? Res Soc Adm Pharm. 2016;12(5):784–8.
Cao YJ, Mager DE, Simonsick EM, Hilmer SN, Ling SM, Windham BG, et al. Physical and cognitive performance and burden of anticholinergics, sedatives, and ACE inhibitors in older women. Clin Pharmacol Ther. 2008;83(3):422–9.
Gnjidic D, Cumming RG, Le Couteur DG, Handelsman DJ, Naganathan V, Abernethy DR, et al. Drug Burden Index and physical function in older Australian men. Br J Clin Pharmacol. 2009;68(1):97–105.
Reeve E, Andrews JM, Wiese MD, Hendrix I, Roberts MS, Shakib S. Feasibility of a patient-centered deprescribing process to reduce inappropriate use of proton pump inhibitors. Ann Pharmacother. 2015;49(1):29–38.
Reeve E, Wiese MD, Hendrix I, Roberts MS, Shakib S. People’s attitudes, beliefs, and experiences regarding polypharmacy and willingness to Deprescribe. J Am Geriatr Soc. 2013;61(9):1508–14.
Schuling J, Gebben H, Veehof LJ, Haaijer-Ruskamp FM. Deprescribing medication in very elderly patients with multimorbidity: the view of Dutch GPs. A qualitative study. BMC Family Pract. 2012;13:56.
Acknowledgements
The authors appreciate the co-operation of the residential care provider who agreed to take part in the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Lotteries Health Research (LHR). No other funding was received for this study.
Conflicts of interest
The author(s) declare no competing interests and are responsible for this report’s content.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ailabouni, N., Mangin, D. & Nishtala, P.S. DEFEAT-polypharmacy: deprescribing anticholinergic and sedative medicines feasibility trial in residential aged care facilities. Int J Clin Pharm 41, 167–178 (2019). https://doi.org/10.1007/s11096-019-00784-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-019-00784-9