Abstract
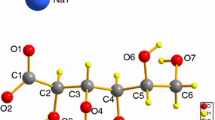
An important coordination compound potassium d-gluconate K[d-C6H11O7](s) has been synthesized by liquid phase method. The chemical component and crystal structure of the important compound are characterized by chemical analysis, elemental analysis, and X-ray crystallography. Single-crystal X-ray analysis reveals that the compound exhibits the chelate property of d-gluconate anions to K+ cations, a six-membered chelate ring is formed by the coordination of K+ with O2 of carboxylate and O4 of hydroxyl in a d-gluconate, and one cation is coordinated to six d-gluconate anions. The lattice potential energy and ionic volume of the anion [d-C6H11O7]− are obtained from crystallographic data. In accordance with Hess’ law, a reasonable thermochemical cycle is designed according to the practical synthesis reaction of the compound and the standard molar enthalpy of formation of the compound is calculated to be −(1754.17 ± 0.19) kJ mol−1 as an important physical quantity in chemical thermodynamics by an isoperibol solution–reaction calorimeter. Molar enthalpies of dissolution of the compound at various molalities are measured at T = 298.15 K in the double-distilled water. According to Pitzer’s electrolyte solution theory, molar enthalpy of dissolution of the title compound at infinite dilution is calculated to be \(\Delta_{\text{s}} H_{\text{m}}^{\infty }\) = (27.92 ± 0.21) kJ mol−1. In terms of the above value, the standard molar enthalpy of formation of the anion [d-C6H11O7]− in the aqueous solution is determined to be = −(1473.87 ± 0.28) kJ mol−1. The values of relative apparent molar enthalpies (Φ L) and relative partial molar enthalpies of the solvent (\(\bar{L}_{1}\)) and the compound (\(\bar{L}_{2}\)) at different concentrations m/(mol kg−1) are derived from the experimental values of the enthalpies of dissolution of the compound.





Similar content being viewed by others
References
Znad H, Markos J, Bales V. Production of gluconic acid from glucose by Aspergillus niger: growth and non-growth conditions. Process Biochem. 2004;39:1341–5.
Amin MA, Abd El Rehim SS, El-Lithy AS. Corrosion, passivation and breakdown of passivity of Al and Al–Cu alloys in gluconic acid solutions. Electrochim Acta. 2010;55:5996–6003.
Giroux S, Rubini P, Henry B, Aury S. Complexes of praseodymium(III) with d-gluconic acid. Polyhedron. 2000;19:1567–74.
Taylor DM, Williams DR. Trace element medicine and chelation therapy. Cambridge: The Royal Society of Chemistry; 1995. p. 77–97.
Di YY, Zhang YH, Kong YX, Zhou CS. Crystal structure and thermochemical properties of potassium pyruvate C3H3O3K(s). J Therm Anal Calorim. 2017;127:1523–32.
Di YY, Zhang YH, Liu YP, Kong YX, Zhou CS. Crystal structure and thermodynamic properties of sodium d-gluconate Na[d-C6H11O7](s). J Therm Anal Calorim. 2017;127:1835–43.
Venkata Krishnan R, Jogeswararao G, Ananthasivan K. The standard molar enthalpies of formation of RE6UO12 (RE = La, Nd) by acid solution calorimetry. J Therm Anal Calorim. 2015;121:1375–82.
Kusto AV, Antonova OA, Smirnova NL. Thermodynamics of solution of l-tryptophan in water. J Therm Anal Calorim. 2017. doi:10.1007/s10973-017-6172-0.
Wszelaka-Rylik M. Thermodynamics of β-cyclodextrin–ephedrine inclusion complex formation and covering of nanometric calcite with these substances. J Therm Anal Calorim. 2017;127:1825–34.
Usacheva TR, Pham Thi L, Kuzmina KI, et al. Thermodynamics of complex formation between Cu(II) and glycyl–glycyl–glycine in water–ethanol and water–dimethylsulfoxide solvents. J Therm Anal Calorim. 2017. doi:10.1007/s10973-017-6207-6.
Li CH, Jiang Y, Jiang JH, Li X, Xiao SX, Tao LM, Yao FH, Zhang H, Xia XM, Yao LH, Zhou H, Xiang YH, Tian Y, Li QG. Standard molar enthalpy of formation of [(C12H8N2)2Bi(O2NO)3] and its biological activity on Schizosaccharomyces pombe. J Therm Anal Calorim. 2017;128:1743–51.
Wang J-F, Wang X-L. Synthesis, characterization and determination of standard molar enthalpies of formation of two zinc borates with higher ratio of ZnO/B2O3 by solution calorimetry. J Therm Anal Calorim. 2017;129:1145–9.
Rychly R, Pekarek V. The use of potassium chloride and tris-(hydroxymethyl) aminomethane as standard substances for solution calorimetry. J Chem Thermodyn. 1977;9:391–6.
Littleton CD. A structure determination of the gluconate ion. Acta Cryst. 1953;6:775–81.
Lis T. Structure of sodium d-gluconate, Na[C6H11O7]. Acta Cryst. 1984;C40:376–8.
Lis T. Structure of lead(II) d-gluconate, Pb[C6H11O7]2. Acta Cryst. 1984;C40:374–6.
Jenkins HDB, Tudela D, Glasser L. Lattice potential energy estimation for complex ionic salts from density measurements. Inorg Chem. 2002;41:2364–7.
Glasser L, Jenkins HDB. Internally consistent ion volumes and their application in volume-based thermodynamics. Inorg Chem. 2008;47(14):6195–202.
Yao YB, Xie T, Gao YM. Handbook of physical chemistry. ShangHai: Shanghai Science and Technique Publishing House; 1985.
Chase MW. NIST-JANAF thermochemical tables, fourth edition. J Phys Chem Ref Data Monogr. 1998;9:1–1951.
Pitzer KS, editor. Chapter 3: ion interaction approach: theory and data correlation. In: Activity coefficients in electrolyte solutions, 2nd ed. Boca Raton: CRC Press; 1991. p. 75–153.
Silvester LF, Pitzer KS. Thermodynamics of electrolytes. High-temperature properties, including enthalpy and heat capacity, with application to sodium chloride. J Phys Chem. 1977;81(19):1822–8.
Acknowledgements
This work is financially supported by the National Natural Science Foundations of China under the contract NSFC No. 21273100.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di, YY., Zhang, YH., Liu, YP. et al. Crystal structure and chemical thermodynamic properties of potassium d-gluconate K(d-C6H11O7)(s). J Therm Anal Calorim 131, 1373–1383 (2018). https://doi.org/10.1007/s10973-017-6657-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-017-6657-x