Abstract
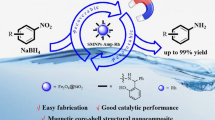
A new chemical approach for the fabrication of Fe3O4 embedded ZnO magnetic semicondutctor composite is reported. The method consists in increasing the pH of the synthesis solution by the thermal decomposition of urea instead of using common alkaline agents, such as NaOH and NH4OH. The material (Fe3O4@ZnO) was used as a platform for the fabrication of highly dispersed gold nanoparticles (~5 nm). The catalytic efficiency of the material, Fe3O4@ZnO@Au, was tested in the photodegradation of Rhodamine-B solutions, and prominent catalytic efficiency, stability, and recycling were achieved. A single portion of the catalyst could be used up to five times without significant loss of activity and its photodegradation efficiency was considered high even after the 12th cycle (56%). Catalyst separation after each batch could be easily achieved because of the intrinsic magnetic property of the material. Leaching monitoring of free Zn species during the fabrication of the catalyst suggests that the use of urea decreased substantially the formation of non-magnetic-semiconducting species and provided a higher mass yield of the magnetic composite compared to an analogous protocol using NaOH. The catalyst was also characterized by detailed structural and chemical analyses, such as transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), and vibration sample magnetometer (VSM).









Similar content being viewed by others
References
Fageria P, Gangopadhyay S, Pande S (2014) Rsc Adv 4(48):24962–24972
Nagaraja R, Kottam N, Girija CR, Nagabhushana BM (2012) Powder Technol 215:91–97
Singh S, Barick KC, Bahadur D (2013) J Mater Chem A 1:3325–3333
Zhang K, Zhou W, Zhang X, Qu Y, Wang L, Hu W, Tian G (2016) RSC Adv 6:50506–50512
Chamjangali MA, Boroumand S (2013) J Braz Chem Soc 24(8):1329–1338
Hernández S, Hidalgo D, Sacco A, Chiodoni A, Lamberti A, Cauda V, Saracco G (2015) Phys Chem Chem Phys 17(12):7775–7786
Height MJ, Pratsinis SE, Mekasuwandumrong O, Praserthdam P (2006) Appl Catal B-Environ 3:305–312
Alshammari A, Bagabas A, Assulami M (2014) Photodegradation of rhodamine B over semiconductor supported gold nanoparticles: The effect of semiconductor support identity. Arab J Chem. https://doi.org/10.1016/j.arabjc.2014.11.013
Hu X, Xu Q, Ge C, Su N, Zhang J, Huang H, Cheng J (2016) Nanotechnology 28(4):045604
Collard X, El Hajj M, Su BL, Aprile C (2014) Micro Mesopor Mat 184:90–96
Wang F, Liang L, Shi L, Liu M, Sun J (2014) Dalton T 43:16441–16449
Yang L, Wu H, Jia J, Ma B, Li J (2017) Micro Mesopor Mat 253:151–159
Jacinto MJ, Kiyohara PK, Masunaga SH, Jardim RF, Rossi LM (2008) Appl Catal A-Gen 338(1):52–57
Nguyen VC, Nguyen NLG, Pho QH (2015) ADV Nat Sci-Nanosc 6(3):035001
Park J, An K, Hwang Y, Park JG, Noh HJ, Kim JY, Park JH, Hwang NM, Hyeon T (2004) Nat Mater 3:891–895
Kandula S, Jeevanandam P (2015) RSC Adv 5(7):5295–5306
Farrokhi M, Hosseini SC, Yang JK, Shirzad-Siboni M (2014) Water Air Soil Pollut 225(9):2113
Teng Z, Li J, Yan F, Zhao R, Yang W (2009) J Mater Chem 19:1811–1815
Worawong A, Jutarosaga T, Onreabroy W (2014) Adv Mat Res 979:208–211
Devaraj N K, Ong B H (2011) In AIP Conference Proceedings 1328(1):288-290
Lu J, Wang H, Peng D, Chen T, Dong S, Chang Y (2016) Phys E 78:41–48
Peng C, Liu Y (2013) Appl Phys A 111:1151–1157
Yamashita T, Hayes P (2008) Appl Surf Sci 254:2441–2449
Hosseini-Sarvari M, Khanivar A, Moeini F (2015) J Mater Sci 50(8):3065–3074
Olteanu NL, Rogozea EA, Popescu SA, Petcu AR, Lazăr CA, Meghea A, Mihaly M (2016) J Mol Catal A-Chem 414:148–159
Pauporte T, Rathouský J (2007) J Phys Chem C 111:7639–7644
Acknowledgements
The authors are grateful to Fundação de Amparo a Pesquisa do Estado de Mato Grosso (FAPEMAT) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support, and indebted to LNNano-Brazil, LME-DEMA-UFSCAR and LMC-UnB for XPS, TEM and BET analyses, respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Highlights
-
A novel mesoporous ZnO-modified magnetite as gold catalyst support has been synthesized.
-
The amount of free Zn species in the solution was notably lower compared to traditional protocols.
-
Enhanced photocatalytic performance was achieved towards the photodegradation of Rhodamine-B.
-
The intrinsic magnetic property allowed an easy and straightforward catalyst separation.
Rights and permissions
About this article
Cite this article
da Silva, R.A., Jacinto, M.J., Silva, V.C. et al. Urea-assisted fabrication of Fe3O4@ZnO@Au composites for the catalytic photodegradation of Rhodamine-B. J Sol-Gel Sci Technol 86, 94–103 (2018). https://doi.org/10.1007/s10971-018-4607-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-018-4607-0