Abstract
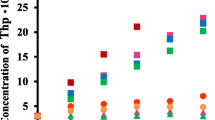
The interaction of two benzocycloheptanes namely, pizotifen (Pizo) and ketotifen (Keto), with cyclodextrins (CDs: α-, β-, γ-, and HP-β-CDs) has been investigated by several techniques including phase solubility, X-ray powder diffractometry, 1H-nuclear magnetic resonance and molecular mechanical modeling. The effects of CD type, pH, ionic strength and temperature on complex stability were also explored. The complex formation constant (K 11) values for the Pizo/CD system follows the decreasing order β-CD > γ-CD > HP-β-CD > α-CD. However, for the Keto/CD system it follows the decreasing order γ-CD > β-CD > HP-β-CD > α-CD. The tendency of Pizo and Keto to complex with β-CD is driven to the extent of 70% by the hydrophobic effect. Complex formation of Keto and Pizo was substantially driven by entropy (>100 J⋅mol−1⋅K−1) but slightly retarded by enthalpy (3–8 kJ⋅mol−1). 1H-NMR and MM+ studies indicate multimodal inclusion of the methylpiperadine, thiophene and phenyl moieties into the β-CD cavity.
Similar content being viewed by others
References
Cramer, F., Saenger, W., Spatz, H.-Ch.: Inclusion compounds. XIX. The formation of inclusion compounds of α-cyclodextrin in aqueous solutions: thermodynamic and kinetics. J. Am. Chem. Soc. 89, 14–20 (1967)
Liu, F.-Y., Kildsig, D.O., Mitra, A.K.: Beta-cyclodextrin/steroid complexation: effect of steroid structure on association equilibria. Pharm. Res. 7, 869–873 (1990)
Mura, P., Bettinetti, G.P., Manderioli, A., Faucci, M.T., Bramanti, G., Sorrenti, M.: Interaction of ketoprofen and ibuprofen with β-cyclodextrins in solution and in the solid state. Int. J. Pharm. 166, 189–203 (1998)
Tong, W.-Q., Lach, J.L., Chin, T.-F., Guillory, J.K.: Structural effects on the binding of amine drugs with the diphenylmethyl functionality to cyclodextrins. I. A microcalorimetric study. Pharm. Res. 8, 951–957 (1991)
Tong, W.-Q., Lach, J.L., Chin, T.-F., Guillory, J.K.: Structural effects on the binding of amine drugs with the diphenylmethyl functionality to cyclodextrins. II. A molecular modeling study. Pharm. Res. 8, 1307–1312 (1991)
Müller, B.W., Albers, E.: Complexation of dihydropyridine derivatives with cyclodextrins and 2-hydroxypropyl-β-cyclodextrin in solution. Int. J. Pharm. 79, 273–288 (1992)
Murphy, R.S., Barros, T.C., Barnes, J., Mayer, B., Marconi, G., Bohne, C.: Complexation of fluorenone and xanthone to cyclodextrins: comparison of theoretical and experimental studies. J. Phys. Chem. A 103, 137–146 (1999)
El-Barghouthi, M.I., Masoud, N.A., Al-Kafawein, J.K., Zughul, M.B., Badwan, A.A.: Host-guest interactions of risperidone with natural and modified cyclodextrins: phase solubility, thermodynamics and molecular modeling studies. J. Incl. Phenom. Macrocycl. Chem. 53, 15–22 (2005)
Al Omari, M.M., Zughul, M.B., Davies, J.E.D., Badwan, A.A.: Cisapride/β-cyclodextrin complexation: stability constants, thermodynamics, and guest-host interactions probed by 1H-NMR and molecular modeling studies. J. Incl. Phenom. Macrocycl. Chem. 57, 511–517 (2007)
Al Omari, M.M., Zughul, M.B., Davies, J.E.D., Badwan, A.A.: Effect of buffer species on the complexation of basic drug terfenadine with β-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 58, 227–235 (2007)
Omar, L., El-Barghouthi, M.I., Masoud, N.A., Abdoh, A.A., Al Omari, M.M., Zughul, M.B., Badwan, A.A.: Inclusion complexation of loratadine with natural and modified cyclodextrins: phase solubility and thermodynamic studies. J. Solution Chem. 36, 605–616 (2007)
Al Omari, M.M., Zughul, M.B., Davies, J.E.D., Badwan, A.A.: Fexofenadine/cyclodextrin inclusion complexation: phase solubility, thermodynamic, physicochemical and computational analysis. Drug Dev. Ind. Pharm. 33, 1205–1215 (2007)
Marzona, M., Pessione, E., Di Martino, S., Giunta, C.: Benzothiophene and dibenzothiophene as the sole sulfur source in Acinetobacter: Growth kinetics and oxidation products. Fuel Process. Technol. 52, 199–205 (1997)
Takashima, Y., Sakamoto, K., Oizumi, Y., Yamaguchi, H., Kamitori, S., Harada, A.: Complexation formation of cyclodextrins with various thiophenes and their polymerization in water: preparation of poly-pseudo-rotaxanes containing poly (thiophene)s. J. Incl. Phenom. Macrocycl. Chem. 56, 45–53 (2006)
Uekama, K., Narisawa, S., Hirayama, F., Otagiri, M.: Improvement of dissolution and absorption characteristics of benzodiazepines by cyclodextrin complexation. Int. J. Pharm. 16, 327–338 (1983)
Loftsson, T., Guðmundsdóttir, H., Sigurjónsdóttir, J.F., Sigurðsson, H.H., Sigfússon, S.D., Másson, M., Stefánsson, E.: Cyclodextrin solubilization of benzodiazepines: formation of midazolam nasal spray. Int. J. Pharm. 212, 29–40 (2001)
Shapiro, Y.E., Gorbatyuk, V.Y., Yakubovskaya, L.N., Andronati, K.S., Andronati, S.A.: Spatial structure of β-cyclodextrin calthrates with 1,4-benzodiazepine derivatives. J. Struct. Chem. 37, 747–758 (1996)
Kobetić, R., Jursic, B.S., Bonnette, S., Tsai, J.S.-C., Salvatore, S.J.: Study of the lorazepam: cyclodextrin inclusion complexes using electrospray ionization mass spectrometry. Tetrahedron Lett. 42, 6077–7082 (2001)
Al Omari, M.M., Zughul, M.B., Davies, J.E.D., Badwan, A.A.: Sildenafil/cyclodextrin complexation: stability constants, thermodynamics, and guest-host interactions probed by 1H-NMR and molecular modeling studies. J. Pharm. Biomed. Anal. 41, 857–865 (2006)
Higuchi, T., Connors, K.A.: Phase solubility techniques. Adv. Anal. Chem. Instrum. 4, 117–212 (1965)
Zughul, M.B., Badwan, A.A.: SL2 type phase solubility diagrams, complex formation and chemical speciation of soluble species. J. Incl. Phenom. Macrocycl. Chem. 31, 243–264 (1998)
Al Omari, M.M., Zughul, M.B., Davies, J.E.D., Badwan, A.A.: Effect of buffer species on the inclusion complexation of acidic drug celecoxib with cyclodextrin in solution. J. Incl. Phenom. Macrocycl. Chem. 55, 247–254 (2006)
Taraszewska, J., Migut, K., Kozbiat, M.: Complexation of flutamide by native and modified cyclodextrins. J. Phys. Org. Chem. 16, 121–126 (2003)
Nalluri, B.N., Chowdary, K.R., Murthy, K.R., Hayman, A.R., Becket, G.: Physicochemical characterization and dissolution properties of nimesulide-cyclodextrin binary systems. AAPS Pharm. Sci. Tech. 4(1), Article 2 (2003). http://www.aapspharmscitech.org
Ribeiro, L., Veiga, F.: Complexation of vinpocetine with cyclodextrins in the presence or absence of polymers, binary and ternary complexes preparation and characterization. J. Incl. Phenom. Macrocycl. Chem. 44, 251–256 (2002)
Saichek, R.E., Reddy, K.R.: Electrokinetically enhanced remediation of hydrophobic organic compounds in soils: a review. Crit. Rev. Environ. Sci. Technol. 35, 115–192 (2005)
Ventura, C.A., Giannone, I., Paolino, D., Pistará, V., Corsaro, A., Puglisi, G.: Preparation of celecoxib-dimethyl-β-cyclodextrin inclusion complex: characterization and in vitro permeation study. Eur. J. Med. Chem. 40, 624–631 (2005)
Connors, K.A.: The stability of cyclodextrin complexes in solution. Chem. Rev. 97, 1325–1358 (1997)
Fernandes, C.M., Carvalho, R.A., da Costa, S.P., Veiga, F.J.B.: Multimodal molecular encapsulation of nicardipine hydrochloride by β-cyclodextrin, hydroxypropyl-β-cyclodextrin and triacetyl-β-cyclodextrin in solution. Structural studies by 1H NMR and ROESY experiments. Eur. J. Pharm. Sci. 18, 285–296 (2003)
Aki, H., Niiya, T., Iwase, Y., Kawasaki, Y., Kumai, K., Kimura, T.: Multimodal inclusion complexes of ampicillin with β-cyclodextrin in aqueous solution. Thermochim. Acta 416, 87–92 (2004)
Uccello-Barretta, G., Balzano, F., Sicoli, G., Fríglola, C., Aldana, I., Monge, A., Paolino, D., Guccione, S.: Combining NMR and molecular modeling in a drug delivery context: investigation of the multi-mode inclusion of a new NPY-5 antagonist bromobenzenesulfonamide into β-cyclodextrin. Biorgan. Med. Chem. 12, 447–458 (2004)
Liu, Y., Chen, G.-S., Chen, Y., Lin, J.: Inclusion complexes of azadirachtin with native and methylated cyclodextrins: solubilization and binding ability. Biorgan. Med. Chem. 13, 4037–4042 (2005)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al Omari, M.M., El-Barghouthi, M.I., Zughul, M.B. et al. Comparative Study of the Inclusion Complexation of Pizotifen and Ketotifen with Native and Modified Cyclodextrins. J Solution Chem 37, 249–264 (2008). https://doi.org/10.1007/s10953-007-9234-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-007-9234-2