Abstract
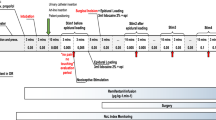
The objective of this work is to compare the performances of two electroencephalogram based indices for detecting loss of consciousness and loss of response to nociceptive stimulation. Specifically, their behaviour after drug induction and during recovery of consciousness was pointed out. Data was recorded from 140 patients scheduled for general anaesthesia with a combination of propofol and remifentanil. The qCON 2000 monitor (Quantium Medical, Barcelona, Spain) was used to calculate the qCON and qNOX. Loss of response to verbal command and loss of eye-lash reflex were assessed during the transition from awake to anesthetized, defining the state of loss of consciousness. Movement as a response to laryngeal mask (LMA) insertion was interpreted as the response to the nociceptive stimuli. The patients were classified as movers or non-movers. The values of qCON and qNOX were statistically compared. Their fall times and rise times defined at the start and at the end of the surgery were calculated and compared. The results showed that the qCON was able to predict loss of consciousness such as loss of verbal command and eyelash reflex better than qNOX, while the qNOX has a better predictive value for response to noxious stimulation such as LMA insertion. From the analysis of the fall and rise times, it was found that the qNOX fall time (median: 217 s) was significantly longer (p value <0.05) than the qCON fall time (median: 150 s). At the end of the surgery, the qNOX started to increase in median at 45 s before the first annotation related to response to stimuli or recovery of consciousness, while the qCON at 88 s after the first annotation related to response to stimuli or recovery of consciousness (p value <0.05). The indices qCON and qNOX showed different performances in the detection of loss of consciousness and loss of response to stimuli during induction and recovery of consciousness. Furthermore, the qCON showed faster decrease during induction. This behaviour is associated with the hypothesis that the loss of response to stimuli (analgesic effect) might be reached after the loss of consciousness (hypnotic effect). On the contrary, the qNOX showed a faster increase at the end of the surgery, associated with the hypothesis that a higher probability of response to stimuli might be reached before the recovery of consciousness.


Similar content being viewed by others
References
Lennmarken C, Sandin R. Neuromonitoring for awareness during surgery. Lancet. 2004;363:1747–8.
Vakkuri A, Yli-Hankala A, Sandin R, Mustola S, Høymork S, Nyblom S, Talja P, Sampson T, van Gils M, Viertiö-Oja H. Spectral entropy monitoring is associated with reduced propofol use and faster emergence in propofol–nitrous oxide–alfentanil anesthesia. Anesthesiology. 2005;103:274–9.
Recart A, White PF, Wang A, Gasanova I, Byerly S, Jones SB. Effect of auditory evoked potential index monitoring on anesthetic drug requirements and recovery profile after laparoscopic surgery: a clinical utility study. Anesthesiology. 2003;99:813–8.
Monk TG, Saini V, Weldon BC, Sigl JC. Anesthetic management and one-year mortality after noncardiac surgery. Anesth Analg. 2005;100:4–10.
Bruhn J, Myles PS, Sneyd R, Struys MM. Depth of anaesthesia monitoring: what’s available, what’s validated and what’s next? Br J Anaesth. 2006;97:85–94.
Mashour GA, Shanks A, Tremper KK, Kheterpal S, Turner CR, Ramachandran SK, Picton P, Schueller C, Morris M, Vandervest JC, Lin N, Avidan MS. Prevention of intraoperative awareness with explicit recall in an unselected surgical population: a randomized comparative effectiveness trial. Anesthesiology. 2012;117(4):717–25.
Migeon A, Desgranges FP, Chassard D, Blaise BJ, De Queiroz M, Stewart A, Cejka JC, Combet S, Rhondali O. Pupillary reflex dilatation and analgesia nociception index monitoring to assess the effectiveness of regional anesthesia in children anesthetised with sevoflurane. Paediatr Anaesth. 2013;23(12):1160–5.
Jensen EW, Lindholm P, Henneberg SW. Autoregressive modeling with exogenous input of middle-latency auditory-evoked potentials to measure rapid changes in depth of anesthesia. Methods Inf Med. 1996;35(3):256–60.
Henneberg SW, Rosenborg D, Jensen EW, Ahn P, Burgdorff B, Thomsen LL. Peroperative depth of anaesthesia may influence postoperative opioid requirements. Acta Anaesthesiol Scand. 2005;49:2293–6.
Rehberg B, Ryll C, Hadzidiakos D, Dincklage FW, Baars JH. Variability comparison of the composite auditory evoked potential index and the bispectral index during propofol-fentanyl anesthesia. Technol Comput Simul. 2008;107(1):117–24.
Jensen EW, Litvan H, Revuelta M, Rodriguez BE, Caminal P, Martinez P, Vereecke H, Struys MM. Cerebral state index during propofol anesthesia: a comparison with the bispectral index and the A-line ARX index. Anesthesiology. 2006;105:28–36.
Boselli E, Daniela-Ionescu M, Bégou G, Bouvet L, Dabouz R, Magnin C, Allaouchiche B. Prospective observational study of the non-invasive assessment of immediate postoperative pain using the analgesia/nociception index (ANI). Br J Anaesth. 2013;111(3):453–9.
Storm H. Changes in skin conductance as a tool to monitor nociceptive stimulation and pain. Curr Opin Anaesthesiol. 2008;21(6):796–804.
Schneider G, Jordan D, Schwarz G, Bischoff P, Kalkman CJ, Kuppe H, Rundshagen I, Omerovic A, Kreuzer M, Stockmanns G, Kochs EF. Monitoring depth of anesthesia utilizing a combination of electroencephalographic and standard measures. Anesthesiology. 2014;120(4):819–28.
Ben-Israel N, Kliger M, Zuckerman G, Katz Y, Edry R. Monitoring the nociception level: a multi-parameter approach. J Clin Monit Comput. 2013;27(6):659–68.
Barvais L, Engelman E, Eba JM, Coussaert E, Cantraine F, Kenny GN. Effect site concentrations of remifentanil and pupil response to noxious stimulation. Br J Anaesth. 2003;91(3):347–52.
Rollins MD, Feiner JR, Lee JM, Shah S, Larson M. Pupillary effects of high-dose opioid quantified with infrared pupillometry. Anesthesiology. 2014;121(5):1037–44.
Gruenewald M, Ilies C, Herz J, Schoenherr T, Fudickar A, Höcker J, Bein B. Influence of nociceptive stimulation on analgesia nociception index (ANI) during propofol–remifentanil anaesthesia. Br J Anaesth. 2013;110:1024–30.
Luginbuhl M, Schumacher PM, Vuilleumier P, Vereecke H, Heyse B, Bouillon TW, Struys M. Noxious stimulation response index: a novel anesthetic state index based on hypnotic-opioid interaction. Anesthesiology. 2010;112(4):872–80.
Bouillon TW, Bruhn J, Radulescu L, Andresen C, Shafer TJ, Cohane C, Shafer SL. Pharmacodynamic interaction between propofol and remifentanil regarding hypnosis, tolerance of laryngoscopy, bispectral index, and electroencephalographic approximate entropy. Anesthesiology. 2004;100:1353–72.
Vereecke HEM, Hannivoort L N, Proost J H, Eleveld D J, Struys MMRF, Luginbühl M (2014) Predictive performance of the noxious stimulation response index as a measure of anesthetic potency during sevoflurane, propofol and remifentanil anesthesia. International Society for Anaesthetic Pharmacology (ISAP) Annual Meeting 2014. http://f1000research.com/posters/1097048.
von Dincklage F, Correll C, Schneider MH, Rehberg B, Baars JH. Utility of nociceptive flexion reflex threshold, bispectral index, composite variability index and noxious stimulation response index as measures for nociception during general anaesthesia. Anaesthesia. 2012;67(8):899–905.
Jensen EW, Valencia JF, López A, Anglada T, Agustí M, Ramos Y, Gambus P. Monitoring hypnotic effect and nociception with two EEG-derived indices, qCON and qNOX, during general anaesthesia. Acta Anaesthesiol Scand. 2014;58(8):933–41.
Schnider TW, Minto CF, Shafer SL, Gambus PL, Andresen C, Goodale DB, Youngs EJ. The influence of age on propofol pharmacodynamics. Anesthesiology. 1999;90:1502–16.
Schnider TW, Minto CF, Gambus PL, Andresen C, Goodale DB, Shafer SL, Youngs EJ. The influence of method of administration and covariates on the pharmacokinetics of propofol in adult volunteers. Anesthesiology. 1998;88:1170–82.
Minto CF, Schnider TW, Egan TD, Youngs E, Lemmens HJ, Gambus PL, Billard V, Hoke JF, Moore KH, Hermann DJ, Muir KT, Mandema JW, Shafer SL. Influence of age and gender on the pharmacokinetics and pharmacodynamics of remifentanil: I model development. Anesthesiology. 1997;86:10–23.
Minto CF, Schnider TW, Shafer SL. Pharmacokinetics and pharmacodynamics of remifentanil: II Model application. Anesthesiology. 1997;86:24–33.
Altman DG. Practical statistics for medical research. London: Chapman & Hall; 1991.
Smith WD, Dutton RC, Smith NT. Measuring the performance of anesthetic depth indicators. Anesthesiology. 1996;84:38–51.
Irwin MG, Hui TWC, Milne SE, Kenny GNC. Propofol effective concentration 50 and its relationship to bispectral index. Anaesthesia. 2002;57(3):242–8.
Gepts E. Pharmacokinetic concepts for TCI anaesthesia. Anaesthesia. 1998;53:4–12.
Stuart PC, Stott SM, Millar A, Kenny GN, Russell D. Propofol with and without nitrous oxide. Br J Anaesth. 2000;85:666.
Wakeling HG, Zimmerman JB, Howell S, Glass PSA. Targeting effect compartment or central compartment concentration of propofol. Anesthesiology. 1999;90:92–7.
Borrat X, Trocóniz IF, Valencia JF, Rivadulla S, Sendino O, Llach J, Castells A. Modeling the Influence of the A118G polymorphism in the OPRM1 gene and of noxious stimulation on the synergistic relation between propofol and remifentanil sedation and analgesia in endoscopic procedures. J Am Soc Anesthesiol. 2013;118(6):1395–407.
Glass PS, Hardman D, Kamiyama Y, Quill TJ, Marton G, Donn KH, Hermann D. Preliminary pharmacokinetics and pharmacodynamics of an ultra-short-acting opioid: remifentanil (GI87084B). Anesth Analg. 1993;77:1031–40.
Egan TD, Lemmens HJ, Fiset P, Hermann DJ, Muir KT, Stanski DR, Shafer SL. The pharmacokinetics of the new short-acting opioid remifentanil (GI87084B) in healthy adult male volunteers. Anesthesiology. 1993;79:881–92.
Glass PS, Gan TJ, Howell S. A review of the pharmacokinetics and pharmacodynamics of remifentanil. Anesth Analg. 1999;89(4S):7.
Kissin I. Depth of anaesthesia and bispectral index monitoring. Anesth Analg. 2000;90:1114–7.
Tams C, Johnson K. Prediction variability of combined pharmacokinetic pharmacodynamic models: a simulation study of propofol in combination with remifentanil and fentanyl. J Anesth Clin Res. 2014;5:393.
Kapila A, Glass P, Jacobs J, Muir K, Hermann D, Shiraishi M, Smith R. Measured context-sensitive half times of remifentanil and alfentanil. Anesthesiology. 1995;83:968–75.
Zbinden AM, Maggiorini M, Petersen-Felix S, Lauber R, Thomson DA, Minder CE. Anesthetic depth defined using multiple noxious stimuli during isoflurane/oxygen anesthesia. I. Motor reactions. Anesthesiology. 1994;80:253–60.
Katoh T, Ikeda K. The effects of fentanyl on sevoflurane requirements for loss of consciousness and skin incision. J Am Soc Anesthesiol. 1998;88(1):18–24.
Gambús PL, Jensen EW, Jospin M, Borrat X, Martínez Pallí G, Fernández-Candil J, Valencia JF, Barba X, Caminal P, Trocóniz IF. Modeling the effect of propofol and remifentanil combinations for sedation-analgesia in endoscopic procedures using an adaptive neuro fuzzy inference system (ANFIS). Anesth Analg. 2011;112(2):331–9.
Acknowledgments
The qNOX was based on an idea from the Department of Anesthesia Hospital CLINIC de Barcelona (Spain) funded by grant PS09/01209 of the Fondo de Investigaciones Sanitarias (FIS), Health Department, Government of Spain and has been developed in collaboration with Quantium Medical.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Umberto Melia, Erik Weber Jensen, Joan Fontanet, Eva Gabarron and Patricia Pineda are employees of Quantium Medical. Quantium Medical is the commercial developer of both qCON and qNOX indices.
Ethical Statements
The study was performed after IRB approval (Committee on Ethics in Research, Hospital CLINIC de Barcelona no 2013/8356). All the patients has written informed consent.
Rights and permissions
About this article
Cite this article
Melia, U., Gabarron, E., Agustí, M. et al. Comparison of the qCON and qNOX indices for the assessment of unconsciousness level and noxious stimulation response during surgery. J Clin Monit Comput 31, 1273–1281 (2017). https://doi.org/10.1007/s10877-016-9948-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-016-9948-z