Abstract
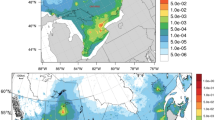
Due to the heavy and expanding agricultural use of neurotoxic pesticides suspected to affect dopaminergic neurons, it is imperative to closely examine the role of pesticides in the development of Parkinson’s disease (PD). We focus our investigation on pesticide use in California’s heavily agricultural central valley by utilizing a unique pesticide use reporting system. From 2001 to 2007, we enrolled 362 incident PD cases and 341 controls living in the Central Valley of California. Employing our geographic information system model, we estimated ambient exposures to the pesticides ziram, maneb, and paraquat at work places and residences from 1974 to 1999. At workplaces, combined exposure to ziram, maneb, and paraquat increased risk of PD three-fold (OR: 3.09; 95% CI: 1.69, 5.64) and combined exposure to ziram and paraquat, excluding maneb exposure, was associated with a 80% increase in risk (OR:1.82; 95% CI: 1.03, 3.21). Risk estimates for ambient workplace exposure were greater than for exposures at residences and were especially high for younger onset PD patients and when exposed in both locations. Our study is the first to implicate ziram in PD etiology. Combined ambient exposure to ziram and paraquat as well as combined ambient exposure to maneb and paraquat at both workplaces and residences increased PD risk substantially. Those exposed to ziram, maneb, and paraquat together experienced the greatest increase in PD risk. Our results suggest that pesticides affecting different mechanisms that contribute to dopaminergic neuron death may act together to increase the risk of PD considerably.
Similar content being viewed by others
Abbreviations
- CA DPR:
-
California department of pesticide regulation
- CI:
-
Confidence interval
- GIS:
-
Geographic information system
- HIPAA:
-
Health insurance portability and accountability act
- OR:
-
Odds ratio
- PD:
-
Parkinson’s disease
- PLSS:
-
Public land survey system
- PUR:
-
Pesticide use report
- UCLA:
-
University of California Los Angeles
- UPS:
-
Ubiquitin proteasome system
References
Twelves D, Perkins KS, Counsell C. Systematic review of incidence studies of Parkinson’s disease. Mov Disord. 2003;18(1):19–31.
Shimizu K, Matsubara K, Ohtaki K, Fujimaru S, Saito O, Shiono H. Paraquat induces long-lasting dopamine overflow through the excitotoxic pathway in the striatum of freely moving rats. Brain Res. 2003;976(2):243–52.
Purisai MG, McCormack AL, Cumine S, Li J, Isla MZ, Di Monte DA. Microglial activation as a priming event leading to paraquat-induced dopaminergic cell degeneration. Neurobiol Dis. 2007;25(2):392–400.
McCormack AL, Thiruchelvam M, Manning-Bog AB, et al. Environmental risk factors and Parkinson’s disease: selective degeneration of nigral dopaminergic neurons caused by the herbicide paraquat. Neurobiol Dis. 2002;10(2):119–27.
Ossowska K, Smialowska M, Kuter K, et al. Degeneration of dopaminergic mesocortical neurons and activation of compensatory processes induced by a long-term paraquat administration in rats: implications for Parkinson’s disease. Neuroscience. 2006;141(4):2155–65.
Zhou Y, Shie FS, Piccardo P, Montine TJ, Zhang J. Proteasomal inhibition induced by manganese ethylene-bis-dithiocarbamate: relevance to Parkinson’s disease. Neuroscience. 2004;128(2):281–91.
Zhang J, Fitsanakis VA, Gu G, et al. Manganese ethylene-bis-dithiocarbamate and selective dopaminergic neurodegeneration in rat: a link through mitochondrial dysfunction. J Neurochem. 2003;84(2):336–46.
Chou AP, Maidment N, Klintenberg R, et al. Ziram causes dopaminergic cell damage by inhibiting E1 ligase of the proteasome. J Biol Chem. 2008;283(50):34696–703.
Cory-Slechta DA, Thiruchelvam M, Barlow BK, Richfield EK. Developmental pesticide models of the Parkinson disease phenotype. Environ Health Perspect. 2005;113(9):1263–70.
Barlow BK, Thiruchelvam MJ, Bennice L, Cory-Slechta DA, Ballatori N, Richfield EK. Increased synaptosomal dopamine content and brain concentration of paraquat produced by selective dithiocarbamates. J Neurochem. 2003;85(4):1075–86.
Thiruchelvam M, Prokopenko O, Cory-Slechta DA, Buckley B, Mirochnitchenko O. Overexpression of superoxide dismutase or glutathione peroxidase protects against the paraquat + maneb-induced Parkinson disease phenotype. J Biol Chem. 2005;280(23):22530–9.
WHO. Public health impact of pesticide used in agriculture. Geneva: World Health Organization; 1990.
Ecobichon DJ. Pesticide use in developing countries. Toxicology. 2001;1–3(160):27–33.
Ward MH, Lubin J, Giglierano J, et al. Proximity to crops and residential exposure to agricultural herbicides in iowa. Environ Health Perspect. 2006;114(6):893–7.
Priyadarshi A, Khuder SA, Schaub EA, Priyadarshi SS. Environmental risk factors and Parkinson’s disease: a metaanalysis. Environ Res. 2001;86(2):122–7.
Petrovitch H, Ross GW, Abbott RD, et al. Plantation work and risk of Parkinson disease in a population-based longitudinal study. Arch Neurol. 2002;59(11):1787–92.
Ascherio A, Chen H, Weisskopf MG, et al. Pesticide exposure and risk for Parkinson’s disease. Ann Neurol. 2006;60(2):197–203.
Costello S, Cockburn M, Bronstein J, Zhang X, Ritz B. Parkinson’s disease and residential exposure to maneb and paraquat from agricultural applications in the central valley of California. Am J Epidemiol. 2009;169(8):919–26.
Kamel F, Tanner C, Umbach D, et al. Pesticide exposure and self-reported Parkinson’s disease in the agricultural health study. Am J Epidemiol. 2007;165(4):364–74.
Elbaz A, Clavel J, Rathouz PJ, Moisan F, Galanaud JP, Delemotte B, Alperovitch A, Tzourio C. Professional exposure to pesticides and Parkinson’s disease. Ann Neurol. 2009;66(4)494–504.
Liou HH, Tsai MC, Chen CJ, et al. Environmental risk factors and Parkinson’s disease: a case-control study in Taiwan. Neurology. 1997;48(6):1583–8.
Dhillon AS, Tarbutton GL, Levin JL, et al. Pesticide/environmental exposures and Parkinson’s disease in East Texas. J Agromed. 2008;13(1):37–48.
Seidler A, Hellenbrand W, Robra BP, et al. Possible environmental, occupational, and other etiologic factors for Parkinson’s disease: a case-control study in Germany. Neurology. 1996;46(5):1275–84.
Hancock DB, Martin ER, Mayhew GM, et al. Pesticide exposure and risk of Parkinson’s disease: a family-based case-control study. BMC Neurol. 2008;8:6.
Tanner CM, Goldman SM. Epidemiology of Parkinson’s disease. Neurol Clin. 1996;14(2):317–35.
Goldberg DW, Zhang, X, Marusek JC, Wilson JP, Ritz B, Cockburn MG. Development of an automated pesticide exposure analyst for California’s central valley. Proceedings of the urban and regional information systems association GIS in public health conference. 2007;136–56 http://www.dwgold.com/conferences/Proceedings/urisaHealth2007.pdf.
Kang GA, Bronstein JM, Masterman DL, Redelings M, Crum JA, Ritz B. Clinical characteristics in early Parkinson’s disease in a central California population-based study. Mov Disord. 2005;20(9):1133–42.
Rull RP, Ritz B. Historical pesticide exposure in California using pesticide use reports and land-use surveys: an assessment of misclassification error and bias. Environ Health Perspect. 2003;111(12):1582–9.
Chester G, Ward RJ. Occupational exposure and drift hazard during aerial application of paraquat to cotton. Arch Environ Contam Toxicol. 1984;13(5):551–63.
McElroy JA, Remington PL, Trentham-Dietz A, Robert SA, Newcomb PA. Geocoding addresses from a large population-based study: lessons learned. Epidemiology. 2003;14(4):399–407.
MacCollom GB. Drift comparisons between aerial and ground orchard application. J Econ Entomol. 1986;79:459–64.
Ritz B, Costello S. Geographic model and biomarker-derived measures of pesticide exposure and Parkinson’s disease. Ann N Y Acad Sci. 2006;1076:378–87.
CDWR. Land use survey (2010). Available from: http://www.water.ca.gov/landwateruse/lusrvymain.cfm. Cited 5 May 2010.
Chan JY, Chan SH, Dai KY, Cheng HL, Chou JL, Chang AY. Cholinergic-receptor-independent dysfunction of mitochondrial respiratory chain enzymes, reduced mitochondrial transmembrane potential and ATP depletion underlie necrotic cell death induced by the organophosphate poison mevinphos. Neuropharmacology. 2006;51(7–8):1109–19.
Hatcher JM, Richardson JR, Guillot TS, et al. Dieldrin exposure induces oxidative damage in the mouse nigrostriatal dopamine system. Exp Neurol. 2007;204(2):619–30.
Baldi I, Cantagrel A, Lebailly P, et al. Association between Parkinson’s disease and exposure to pesticides in southwestern France. Neuroepidemiology. 2003;22(5):305–10.
Dick FD, De Palma G, Ahmadi A, et al. Environmental risk factors for Parkinson’s disease and parkinsonism: the Geoparkinson study. Occup Environ Med. 2007;64(10):666–72.
Firestone JA, Smith-Weller T, Franklin G, Swanson P, Longstreth WT Jr, Checkoway H. Pesticides and risk of Parkinson disease: a population-based case-control study. Arch Neurol. 2005;62(1):91–5.
Frigerio R, Sanft KR, Grossardt BR, et al. Chemical exposures and Parkinson’s disease: a population-based case-control study. Mov Disord. 2006;21(10):1688–92.
Nuti A, Ceravolo R, Dell’Agnello G, et al. Environmental factors and Parkinson’s disease: a case-control study in the Tuscany region of Italy. Parkinsonism Relat Disord. 2004;10(8):481–5.
Acknowledgments
This work was supported by National Institute of Environmental Health Science [grant numbers ES10544, U54ES12078, 5P30 ES07048], National Institute of Neurological Disorders and Stroke [grant number NS 038367], and Department of Defense Prostate Cancer Research Program [grant number 051037]; in addition, initial pilot funding was provided by the American Parkinson’s Disease Association.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, A., Costello, S., Cockburn, M. et al. Parkinson’s disease risk from ambient exposure to pesticides. Eur J Epidemiol 26, 547–555 (2011). https://doi.org/10.1007/s10654-011-9574-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-011-9574-5