Abstract
Objective
The available content of mercury (Hg) in farmland soil is directly related to the safety of agricultural products. Meanwhile, humans may accumulate high concentrations of Hg through the food chain, resulting in health damage. Regarding the remediation technologies of Hg-contaminated soil, research and development is mainly concentrated on the immobilisation of Hg in soil and efficient extraction by accumulators. Therefore, in this work, the highly Hg-tolerant strain Pseudomonas alkylphenolica KL28 was used to study the removal effect of Hg in a solution, immobilization effect of Hg in soil, and its effect on growth, Hg accumulation and photosynthetic characteristics of Brassica campestris L.
Results
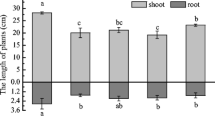
KL28 could effectively remove Hg2+ in the solution, with the removal ratio of 96.0% at 24 h. This strain could reduce decreases in shoots’ and roots’ dry weights by 31% and 16%, respectively, at a Hg concentration of 20 mg/L. The available Hg in the soil decreased to 4.7–9.4% in 8 days treated with KL28 bacterial solution at a dosage of 100 L/hm2. Meanwhile, with increases in Hg concentrations, Fv/Fm, Y(II), Y(I) and Y(NPQ) in the leaves of B. campestris showed a downward trend while Y(ND) and Y(NO) displayed an upward trend. Under the stress of 20 mg/L Hg2+, KL28 could reduce the Fv/Fm from 11.2 to 6.1%.
Conclusions
KL28 could effectively remove Hg in the solution, immobilize Hg in soil, promote growth, decrease Hg accumulation and affect photosynthetic characteristics of B. campestris, indicating its potential use in Hg contaminated soils.








Similar content being viewed by others
References
Abu-Dieyeh MH, Alduroobi HM, Al-Ghouti MA (2019) Potential of mercury-tolerant bacteria for bio-uptake of mercury leached from discarded fluorescent lamps. J Environ Manag 237(1):217–227
Antal TK, Graevskaia EE, Matorin DN, Voronova EN, Pogosian SI, Krendeleva TE, Rubin AB (2004) Effect of mercuric chloride and methylmercury on photosynthetic activity of the diatom Thalassiosira weissflogii by a fluorescent method. Biofizika 49(1):72–78
Asztalos E, Sipka G, Kis M, Trotta M, Maroti P (2012) The reaction center is the sensitive target of the mercury(II) ion in intact cells of photosynthetic bacteria. Photosynth Res 112(2):129–140
Baldi F, Gallo M, Battistel D, Barbaro E, Gambaro A, Daniele S (2016) A broad mercury resistant strain of Pseudomonas putida secretes pyoverdine under limited iron conditions and high mercury concentrations. Biometals 29(6):1097–1106
Bertrand M, Poirier I (2005) Photosynthetic organisms and excess of metals. Photosynthetica 43(3):345–353
Clijsters H, Assche FV (1985) Inhibition of photosynthesis by heavy metals. Photosynth Res 7(1):31–40
De Filippis LF, Rüdiger Hampp, Ziegler H (1981) The effects of sublethal concentrations of zinc, cadmium and mercury on Euglena. Arch Microbiol 128(4):407–411
Deng C, Zhang D, Pan X, Chang F, Wang S (2013) Toxic effects of mercury on PSI and PSII activities, membrane potential and transthylakoid proton gradient in Microsorium pteropus. J Photochem Photobiol B 127:1–7
Fitzgerald WF, Engstrom DR, Mason RP, Nater EA (1998) The case for atmospheric mercury contamination in remote areas. Environ Sci Technol 32(1):1–7
García-Sánchez M, Száková J (2016) Chapter 12—biological remediation of mercury-polluted environments. Plant metal interaction, Elsevier Inc., Amsterdam, pp 311–334.
Han HS, Supanjani Lee KD (2006) Effect of co-inoculation with phosphate and potassium solubilizing bacteria on mineral uptake and growth of pepper and cucumber. Plant Soil Environ 52(3):130–136
Jeong JJ, Kim JH, Kim CK, Hwang I, Lee K (2003) 3- and 4-alkylphenol degradation pathway in Pseudomonas sp. strain KL28: genetic organization of the lap gene cluster and substrate specificities of phenol hydroxylase and catechol 2,3-dioxygenase. Microbiology 149(11): 3265–3277.
Jiang H, Qiu B (2011) Inhibition of photosynthesis by UV-B exposure and its repair in the bloom-forming cyanobacterium Microcystis aeruginosa. J Appl Phycol 23(4):691–696
Khadivinia E, Shara H, Hadi F, Zahiri HS, Modiri S, Tohidi A, Mousavi A, Salmanian AH, Noghabi KA (2014) Cadmium biosorption by a glyphosate-degrading bacterium, a novel biosorbent isolated from pesticide-contaminated agricultural soils. J Ind Eng Chem 20:4304–4310
Kojima Y, Hiyama T, Sakurai H (1987) Effects of Mercurials on iron-sulfur centers of photosystem I of Anacystis nidulans. Progress photosynthesis research, Springer, Netherlands, pp 57–60
Kramer DM, Johnson G, Kiirats O, Edwards GE (2004) New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth Res 79(2):209–218
Laacouri A, Nater EA, Kolka RK (2013) Distribution and uptake dynamics of mercury in leaves of common deciduous tree species in Minnesota, USA. Environ Sci Technol 47(18):10462–10470
Lee K, Lim JL, Kim KS, Huang SL, Veeranagouda Y, Rehm BHA (2014) An alginate-like exopolysaccharide biosynthesis gene cluster involved in biofilm aerial structure formation by Pseudomonas alkylphenolia. Appl Microbiol Biotechnol 98:4137–4148
Lefebvre DD, Kelly D, Budd K (2007) Biotransformation of Hg(II) by cyanobacteria. Appl Environ Microbiol 73(1):243–249
Li XJ, Li DB, Yan ZN, Ao YS (2018) Biosorption and bioaccumulation characteristics of cadmium by plant growth-promoting rhizobacteria. RSC Adv 8(54):30902–30911
Lim JY, Lee K, Hwang I (2014) Complete genome sequence of the mushroom-like aerial structure-forming Pseudomonas alkylphenolia, a platform bacterium for mass production of poly-β-d-mannuronates. J Biotechnol 192:20–21
Lu CM, Chau CW, Zhang JH (2000) Acute toxicity of excess mercury on the photosynthetic performance of cyanobacterium, S. platensis—assessment by chlorophyll fluorescence analysis. Chemosphere 41(1): 191–196.
Mason RP, Fitzgerald WF, Morel FMM (1994) The biogeochemical cycling of elemental mercury: anthropogenic influences. Geochim Cosmochim Acta 58(15):3191–3198
Patra M, Sharma A (2000) Mercury toxicity in plants. Bot Rev 66(3):379–422
Pfündel E, Klughammer C, Schreiber U (2008) Monitoring the effects of reduced PS II antenna size on quantum yields of photosystems I and II using the Dual-PAM-100 measuring system. PAM Appl Notes 1:21–24
Sinha A, Khare SK (2012) Mercury bioremediation by mercury accumulating Enterobacter sp. cells and its alginate immobilized application. Biodegradation 23(1): 25–34.
Tyagi AK, Malik A (2010) In situ SEM, TEM and AFM studies of the antimicrobial activity of lemon grass oil in liquid and vapour phase against Candida albicans. Micron 41:797–805
Veeranagouda Y, Lee K, Cho AR, Cho K, Anderson EM, Lam JS (2011) Ssg, a putative glycosyltransferase, functions in lipo- and exopolysaccharide biosynthesis and cell surface-related properties in Pseudomonas alkylphenolia. FEMS Microbiol Lett 315:38–45
Wagner-Dōbler I (2013) Bioremediation of mercury: current research and industrial applications. Caister Academic Press, Norfolk, pp 1–147
Yu ZS, Li J, Li YB, Wang Q, Zhai XP, Wu GF, Liu P, Li XK (2014) A mer operon confers mercury reduction in a Staphylococcus epidermidis strain isolated from Lanzhou reach of the Yellow River. Inter Biodeterior Biodegrad 90:57–63
Zhou YT, Aamir M, Liu K, Yang FX, Liu WP (2018) Status of mercury accumulation in agricultural soil across China: spatial distribution, temporal trend, influencing factor and risk assessment. Environ Pollut 240:116–124
Acknowledgements
This work was supported by Agriculture Committee of Shanghai (2014/5–2).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shi, D., Li, D., Zhang, Y. et al. Effects of Pseudomonas alkylphenolica KL28 on immobilization of Hg in soil and accumulation of Hg in cultivated plant. Biotechnol Lett 41, 1343–1354 (2019). https://doi.org/10.1007/s10529-019-02736-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02736-9