Abstract
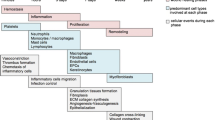
Skin grafts are commonly utilized and proven effective methods of open wound coverage. Revascularization through neoangiogenesis is a pivotal mechanism for skin graft integration and durability. Extracorporeal shock-wave treatment (ESWT) has been demonstrated to accelerate wound repair; however, its mechanism-of-action is unclear. We investigated the role of ESWT in early revascularization of full-thickness skin isografts in a murine model. Cohorts of mice were euthanized and skin grafts were harvested 6 h, 2, 4, and 7 days post grafting ± ESWT. Various aspects of graft neovascularization were measured including gross morphology, quantitative microscopy (vessel number, density), immunohistochemistry (CD31), cDNA SuperArrays for 84 angiogenesis-specific genes, and custom-designed ‘Wound Repair’ TaqMan® Low Density Array (TLDA) cards to assess expression of 188 wound repair genes. We demonstrate that a single administration of ESWT immediately following skin grafting significantly enhances recipient graft revascularization (increased vessel number, size, and density). An augmented early pro-angiogenic and suppressed delayed pro-inflammatory response to ESWT was accompanied by significantly increased expression of both skin graft CD31 and angiogenesis pathway-specific genes, including ELR-CXC chemokines (CXCL1, CXCL2, CXCL5), CC chemokines (CCL2, CCL3, CCL4), cytokines (IL-1β, IL-6, G-CSF, VEGF-A), matrix metalloproteinases (MMP3, MMP9, MMP13), hypoxia-inducible factors (HIF-1α), and vascular remodeling kinase (Mst1), as early as 6 h and up to 7 days post grafting and treatment. These findings suggest that early pro-angiogenic and anti-inflammatory effects of ESWT promote tissue revascularization and wound healing by augmenting angiogenesis and dampening inflammation.




Similar content being viewed by others
Abbreviations
- ESWT:
-
Extracorporeal shock wave treatment
References
Singer AJ, Clark RA (1999) Cutaneous wound healing. N Engl J Med 341:738
Ceradini DJ, Kulkarni AR, Callaghan MJ, Tepper OM, Bastidas N, Kleinman ME, Capla JM, Galiano RD, Levine JP, Gurtner GC (2004) Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Nat Med 10:858
Carmeliet P (2000) Mechanisms of angiogenesis and arteriogenesis. Nat Med 6:389
Burbridge MF, Coge F, Galizzi JP, Boutin JA, West DC, Tucker GC (2002) The role of the matrix metalloproteinases during in vitro vessel formation. Angiogenesis 5:215
Bauer SM, Bauer RJ, Velazquez OC (2005) Angiogenesis, vasculogenesis, and induction of healing in chronic wounds. Vasc Endovascular Surg 39:293
Rafii S, Avecilla S, Shmelkov S, Shido K, Tejada R, Moore MA, Heissig B, Hattori K (2003) Angiogenic factors reconstitute hematopoiesis by recruiting stem cells from bone marrow microenvironment. Ann N Y Acad Sci 996:49
Distler JH, Hirth A, Kurowska-Stolarska M, Gay RE, Gay S, Distler O (2003) Angiogenic and angiostatic factors in the molecular control of angiogenesis. Q J Nucl Med 47:149
Capla JM, Ceradini DJ, Tepper OM, Callaghan MJ, Bhatt KA, Galiano RD, Levine JP, Gurtner GC (2006) Skin graft vascularization involves precisely regulated regression and replacement of endothelial cells through both angiogenesis and vasculogenesis. Plast Reconstr Surg 117:836
Trengove NJ, Stacey MC, MacAuley S, Bennett N, Gibson J, Burslem F, Murphy G, Schultz G (1999) Analysis of the acute and chronic wound environments: the role of proteases and their inhibitors. Wound Repair Regen 7:442
Yager DR, Nwomeh BC (1999) The proteolytic environment of chronic wounds. Wound Repair Regen 7:433
Mast BA, Schultz GS (1996) Interactions of cytokines, growth factors, and proteases in acute and chronic wounds. Wound Repair Regen 4:411
Menke NB, Ward KR, Witten TM, Bonchev DG, Diegelmann RF (2007) Impaired wound healing. Clin Dermatol 25:19
Greenhalgh DG (1996) The healing of burn wounds. Dermatol Nurs 8:13
Steed DL (2006) Clinical evaluation of recombinant human platelet-derived growth factor for the treatment of lower extremity ulcers. Plast Reconstr Surg 117:143S
Wieman TJ (1998) Clinical efficacy of becaplermin (rhPDGF-BB) gel. Becaplermin gel studies group. Am J Surg 176:74S
Ingber DE (2006) Cellular mechanotransduction: putting all the pieces together again. FASEB J 20:811
Pietramaggiori G, Liu P, Scherer SS, Kaipainen A, Prsa MJ, Mayer H, Newalder J, Alperovich M, Mentzer SJ, Konerding MA, Huang S, Ingber DE, Orgill DP (2007) Tensile forces stimulate vascular remodeling and epidermal cell proliferation in living skin. Ann Surg 246:896
Argenta LC, Morykwas MJ, Marks MW, DeFranzo AJ, Molnar JA, David LR (2006) Vacuum-assisted closure: state of clinic art. Plast Reconstr Surg 117:127S
Saxena V, Hwang CW, Huang S, Eichbaum Q, Ingber D, Orgill DP (2004) Vacuum-assisted closure: microdeformations of wounds and cell proliferation. Plast Reconstr Surg 114:1086
Ennis WJ, Foremann P, Mozen N, Massey J, Conner-Kerr T, Meneses P (2005) Ultrasound therapy for recalcitrant diabetic foot ulcers: results of a randomized, double-blind, controlled, multicenter study. Ostomy Wound Manage 51:24
Ennis WJ, Lee C, Meneses P (2007) A biochemical approach to wound healing through the use of modalities. Clin Dermatol 25:63
Schaden W, Thiele R, Kolpl C, Pusch M, Nissan A, Attinger CE, Maniscalco-Theberge ME, Peoples GE, Elster EA, Stojadinovic A (2007) Shock wave therapy for acute and chronic soft tissue wounds: a feasibility study. J Surg Res 143:1
Davis TA, Stojadinovic A, Anam K, Amare M, Naik S, Peoples GE, Tadaki D, Elster EA (2008) Extracorporeal shock wave therapy suppresses the early proinflammatory immune response to a severe cutaneous burn injury. Int Wound J. doi:10.1111/j.1742-481X.2008.00540.x
Meirer R, Kamelger FS, Huemer GM, Wanner S, Piza-Katzer H (2005) Extracorporal shock wave may enhance skin flap survival in an animal model. Br J Plast Surg 58:53
Nishida T, Shimokawa H, Oi K, Tatewaki H, Uwatoku T, Abe K, Matsumoto Y, Kajihara N, Eto M, Matsuda T, Yasui H, Takeshita A, Sunagawa K (2004) Extracorporeal cardiac shock wave therapy markedly ameliorates ischemia-induced myocardial dysfunction in pigs in vivo. Circulation 110:3055
Wang CJ, Huang HY, Pai CH (2002) Shock wave-enhanced neovascularization at the tendon-bone junction: an experiment in dogs. J Foot Ankle Surg 41:16
Wang FS, Yang KD, Chen RF, Wang CJ, Sheen-Chen SM (2002) Extracorporeal shock wave promotes growth and differentiation of bone-marrow stromal cells towards osteoprogenitors associated with induction of TGF-beta1. J Bone Joint Surg Br 84:457
Anam K, Akpinar E, Craighead N, Black AT, Hale DA (2004) Targeted T-cell depletion or CD154 blockade generates mixed hemopoietic chimerism and donor-specific tolerance in mice treated with sirolimus and donor bone marrow. Transplantation 78:1290
Kuo YR, Wu WS, Hsieh YL, Wang FS, Wang CT, Chiang YC, Wang CJ (2007) Extracorporeal shock wave enhanced extended skin flap tissue survival via increase of topical blood perfusion and associated with suppression of tissue pro-inflammation. J Surg Res 143:385
Huemer GM, Meirer R, Gurunluoglu R, Kamelger FS, Dunst KM, Wanner S, Piza-Katzer H (2005) Comparison of the effectiveness of gene therapy with transforming growth factor-beta or extracorporal shock wave therapy to reduce ischemic necrosis in an epigastric skin flap model in rats. Wound Repair Regen 13:262
Isenberg JS, Pappan LK, Romeo MJ, Abu-Asab M, Tsokos M, Wink DA, Frazier WA, Roberts DD (2008) Blockade of thrombospondin-1-CD47 interactions prevents necrosis of full thickness skin grafts. Ann Surg 247:180
O’Ceallaigh S, Herrick SE, Bluff JE, McGrouther DA, Ferguson MW (2006) Quantification of total and perfused blood vessels in murine skin autografts using a fluorescent double-labeling technique. Plast Reconstr Surg 117:140
Belperio JA, Keane MP, Arenberg DA, Addison CL, Ehlert JE, Burdick MD, Strieter RM (2000) CXC chemokines in angiogenesis. J Leukoc Biol 68:1
Huemer GM, Shafighi M, Meirer R, Debagge P, Piza-Katzer H, Gurunluoglu R (2004) Adenovirus-mediated transforming growth factor-beta ameliorates ischemic necrosis of epigastric skin flaps in a rat model. J Surg Res 121:101
Jung H, Gurunluoglu R, Scharpf J, Siemionow M (2003) Adenovirus-mediated angiopoietin-1 gene therapy enhances skin flap survival. Microsurgery 23:374
Lubiatowski P, Goldman CK, Gurunluoglu R, Carnevale K, Siemionow M (2002) Enhancement of epigastric skin flap survival by adenovirus-mediated VEGF gene therapy. Plast Reconstr Surg 109:1986
Rinsch C, Quinodoz P, Pittet B, Alizadeh N, Baetens D, Montandon D, Aebischer P, Pepper MS (2001) Delivery of FGF-2 but not VEGF by encapsulated genetically engineered myoblasts improves survival and vascularization in a model of acute skin flap ischemia. Gene Ther 8:523
Aicher A, Heeschen C, Sasaki K, Urbich C, Zeiher AM, Dimmeler S (2006) Low-energy shock wave for enhancing recruitment of endothelial progenitor cells: a new modality to increase efficacy of cell therapy in chronic hind limb ischemia. Circulation 114:2823
Bach S, Bilgrav K, Gottrup F, Jorgensen TE (1991) The effect of electrical current on healing skin incision. An experimental study. Eur J Surg 157:171
Emsen IM (2007) The effect of ultrasound on flap survival: an experimental study in rats. Burns 33:369
Im MJ, Lee WP, Hoopes JE (1990) Effect of electrical stimulation on survival of skin flaps in pigs. Phys Ther 70:37
Kjartansson J, Lundeberg T, Korlof B (1988) Transcutaneous electrical nerve stimulation (TENS) in ischemic tissue. Plast Reconstr Surg 81:813
Politis MJ, Zanakis MF, Miller JE (1989) Enhanced survival of full-thickness skin grafts following the application of DC electrical fields. Plast Reconstr Surg 84:267
Wang CJ, Wang FS, Yang KD, Weng LH, Hsu CC, Huang CS, Yang LC (2003) Shock wave therapy induces neovascularization at the tendon-bone junction. A study in rabbits. J Orthop Res 21:984
Young SR, Dyson M (1990) The effect of therapeutic ultrasound on angiogenesis. Ultrasound Med Biol 16:261
Zhao M, Bai H, Wang E, Forrester JV, McCaig CD (2004) Electrical stimulation directly induces pre-angiogenic responses in vascular endothelial cells by signaling through VEGF receptors. J Cell Sci 117:397
Hill JM, Zalos G, Halcox JP, Schenke WH, Waclawiw MA, Quyyumi AA, Finkel T (2003) Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N Engl J Med 348:593
Jackson JR, Seed MP, Kircher CH, Willoughby DA, Winkler JD (1997) The codependence of angiogenesis and chronic inflammation. FASEB J 11:457
Hart J (2002) Inflammation. 1: Its role in the healing of acute wounds. J Wound Care 11:205
Cohen T, Nahari D, Cerem LW, Neufeld G, Levi BZ (1996) Interleukin 6 induces the expression of vascular endothelial growth factor. J Biol Chem 271:736
Jung YD, Liu W, Reinmuth N, Ahmad SA, Fan F, Gallick GE, Ellis LM (2001) Vascular endothelial growth factor is upregulated by interleukin-1 beta in human vascular smooth muscle cells via the P38 mitogen-activated protein kinase pathway. Angiogenesis 4:155
Mori R, Kondo T, Ohshima T, Ishida Y, Mukaida N (2002) Accelerated wound healing in tumor necrosis factor receptor p55-deficient mice with reduced leukocyte infiltration. FASEB J 16:963
Ishida Y, Kondo T, Kimura A, Matsushima K, Mukaida N (2006) Absence of IL-1 receptor antagonist impaired wound healing along with aberrant NF-kappaB activation and a reciprocal suppression of TGF-beta signal pathway. J Immunol 176:5598
Uwatoku T, Ito K, Abe K, Oi K, Hizume T, Sunagawa K, Shimokawa H (2007) Extracorporeal cardiac shock wave therapy improves left ventricular remodeling after acute myocardial infarction in pigs. Coron Artery Dis 18:397
Acknowledgments
This work was supported by ONR work unit 602236N.42237.W160.A0806 and by the Combat Wound Initiative Program, a Congressionally funded program of the Henry M. Jackson Foundation for the Advancement of Military Medicine.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors are employees of the U.S. Government. This work was prepared as part of their official duties. Title 17 U.S.C. §105 provides that ‘Copyright protection under this title is not available for any work of the U.S. Government’. Title 17 U.S.C. §101 defined a U.S. Government work as a work prepared by a military service member or employees of the U.S. Government as part of that person’s official duties. The opinions or assertions contained in this paper are the private views of the authors and are not to be construed as reflecting the views, policy or positions of the Department of the Navy or Army, Department of Defense nor the U.S. Government. The experiments reported herein were conducted in compliance with the Animal welfare Act and in accordance with the principles set forth in the current edition of the Guide for Care and Use of Laboratory Animals, Institute for Laboratory Animal Resources, National Research Council, National Academy Press, 1996.
Rights and permissions
About this article
Cite this article
Stojadinovic, A., Elster, E.A., Anam, K. et al. Angiogenic response to extracorporeal shock wave treatment in murine skin isografts. Angiogenesis 11, 369–380 (2008). https://doi.org/10.1007/s10456-008-9120-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-008-9120-6