Abstract
Purpose
The use of adjuvant treatment has not been sufficiently investigated in duodenal adenocarcinoma. This study evaluated the effect of postoperative radiotherapy (PORT) on survival outcomes in this rare malignancy.
Methods
We identified patients who were diagnosed between 2004 and 2013 in the Surveillance, Epidemiology, and End Results database. Overall survival (OS) and disease-specific survival (DSS) were analyzed before and after propensity score matching.
Results
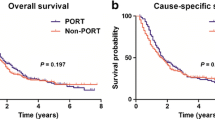
Among the 701 eligible patients, 116 (17%) underwent PORT. There were no significant differences in OS and DSS according to receipt of PORT in the unmatched population (P = 0.982 and 0.496, respectively), whereas the propensity-matched analysis showed improved OS and DSS with PORT (P = 0.053 and 0.019, respectively). No receipt of PORT was an independent poor prognostic factor in multivariate analysis of both OS (P = 0.022) and DSS (P = 0.005). The potential survival benefits of PORT were observed in subgroups of T4 stage, larger tumor size, higher lymph node ratio, and total/radical resection.
Conclusions
We provide useful insights into the therapeutic role of PORT in adenocarcinoma of the duodenum. Adjuvant strategy with PORT needs to be considered in locally advanced tumors.


Similar content being viewed by others
References
Surveillance, Epidemiology and End Results Program (2017) Cancer of the small intestine: cancer stat facts. https://seer.cancer.gov/statfacts/html/smint.html. Accessed 9 Oct 2017
Jabbour SK, Mulvihill D (2014) Defining the role of adjuvant therapy: ampullary and duodenal adenocarcinoma. Semin Radiat Oncol 24:85–93
Cloyd JM, George E, Visser BC (2016) Duodenal adenocarcinoma: advances in diagnosis and surgical management. World J Gastrointest Surg 8:212–221
Onkendi EO, Boostrom SY, Sarr MG et al (2012) 15-year experience with surgical treatment of duodenal carcinoma: a comparison of periampullary and extra-ampullary duodenal carcinomas. J Gastrointest Surg 16:682–691
Singhal N, Singhal D (2007) Adjuvant chemotherapy for small intestine adenocarcinoma. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD005202.pub2
Poultsides GA, Huang LC, Cameron JL et al (2012) Duodenal adenocarcinoma: clinicopathologic analysis and implications for treatment. Ann Surg Oncol 19:1928–1935
Kelsey CR, Nelson JW, Willett CG et al (2007) Duodenal adenocarcinoma: patterns of failure after resection and the role of chemoradiotherapy. Int J Radiat Oncol Biol Phys 69:1436–1441
Bakaeen FG, Murr MM, Sarr MG et al (2000) What prognostic factors are important in duodenal adenocarcinoma? Arch Surg 135:635–641
Kim K, Chie EK, Jang JY et al (2012) Role of adjuvant chemoradiotherapy for duodenal cancer: a single center experience. Am J Clin Oncol 35:533–536
Surveillance, Epidemiology and End Results Program (2017) About the SEER Program. http://seer.cancer.gov/about. Accessed 9 Oct 2017
World Health Organization (2013) International classification of diseases for oncology, 3rd edn. World Health Organization, Geneva
Rosenbaum PR, Rubin DB (1983) The central role of the propensity score in observational studies for causal effects. Biometrika 70:41–55
Rosenbaum PR, Rubin DB (1985) Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat 39:33–38
Austin PC (2011) An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res 46:399–424
Swartz MJ, Hughes MA, Frassica DA et al (2007) Adjuvant concurrent chemoradiation for node-positive adenocarcinoma of the duodenum. Arch Surg 142:285–288
Ecker BL, McMillan MT, Datta J et al (2016) Efficacy of adjuvant chemotherapy for small bowel adenocarcinoma: a propensity score-matched analysis. Cancer 122:693–701
Ecker BL, McMillan MT, Datta J et al (2017) Adjuvant chemotherapy versus chemoradiotherapy in the management of patients with surgically resected duodenal adenocarcinoma: a propensity score-matched analysis of a nationwide clinical oncology database. Cancer 123:967–976
Glynn RJ, Schneeweiss S, Sturmer T (2006) Indications for propensity scores and review of their use in pharmacoepidemiology. Basic Clin Pharmacol Toxicol 98:253–259
Mantripragada KC, Hamid F, Shafqat H et al (2017) Adjuvant therapy for resected gallbladder cancer: analysis of the national cancer data base. J Natl Cancer Inst 109:1–8
Mitin T, Enestvedt CK, Jemal A et al (2017) Limited use of adjuvant therapy in patients with resected gallbladder cancer despite a strong association with survival. J Natl Cancer Inst 109:djw324
Kaklamanos IG, Bathe OF, Franceschi D et al (2000) Extent of resection in the management of duodenal adenocarcinoma. Am J Surg 179:37–41
Overman MJ, Hu CY, Kopetz S et al (2012) A population-based comparison of adenocarcinoma of the large and small intestine: insights into a rare disease. Ann Surg Oncol 19:1439–1445
Wilhelm A, Muller SA, Steffen T et al (2016) Patients with adenocarcinoma of the small intestine with 9 or more regional lymph nodes retrieved have a higher rate of positive lymph nodes and improved survival. J Gastrointest Surg 20:401–410
Overman MJ, Hu CY, Wolff RA et al (2010) Prognostic value of lymph node evaluation in small bowel adenocarcinoma: analysis of the surveillance, epidemiology, and end results database. Cancer 116:5374–5382
Nicholl MB, Ahuja V, Conway WC et al (2010) Small bowel adenocarcinoma: understaged and undertreated? Ann Surg Oncol 17:2728–2732
Yovino S, Poppe M, Jabbour S et al (2011) Intensity-modulated radiation therapy significantly improves acute gastrointestinal toxicity in pancreatic and ampullary cancers. Int J Radiat Oncol Biol Phys 79:158–162
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
About this article
Cite this article
Lim, Y.J., Kim, K. Effect of postoperative radiotherapy on survival in duodenal adenocarcinoma: a propensity score-adjusted analysis of Surveillance, Epidemiology, and End Results database. Int J Clin Oncol 23, 473–481 (2018). https://doi.org/10.1007/s10147-017-1226-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-017-1226-7