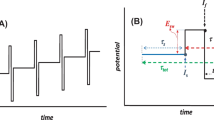
Abstract
Square-wave voltammetry (SWV) is applied to simulate two-step diffusional electrode mechanism, in which the electrochemically active species generated in the second redox step are involved in a reversible follow-up chemical reaction. The model provides insight into relevant mechanistic and kinetic aspects of this complex mechanism. Alongside the scenario of separated SW voltammetric peaks for at least 150 mV (in absolute value), an attention is given to the case when both electron-transfer steps take place at the same formal potential. The last scenario enables development of a strategy to differentiate this complex mechanism relevant for multi-electron redox systems, revealing a profound effect of the follow-up chemical reaction on the voltammetric characteristics of the electron-transfer steps. The presented analysis might help for a rational benchmarking of hydrophilic redox systems that proceed as consecutive multi-electron transformations.








Similar content being viewed by others
References
Saveant JM, Costent C (2019) in Elements of molecular and biomolecular electrochemistry: an electrochemical approach to electron-transfer chemistry. 2nd edition, John Willey&Sons.
Armstrong FA (2015) Electrifying metalloenzymes in: Metalloproteins: Theory, calculations and experiments (A. E. Cho, W. A. Goddar III, eds), CRC Press, Taylor & Francis Group, London, New York USA.
Bucher ES, Wightman RM (2015) Electrochemical analysis of neurotransmitters. Annu Rev Anal Chem 8:239–261
Compton RG, Banks CE (2018) Understanding voltammetry, 2nd edn. World Scientific
Mirceski V, Komorsky-Lovric S, Lovric M (2007) Square-wave voltammetry, theory and application (F. Scholz, ed.) Springer, Berlin, Germany
Lovric M (2010) Square-wave voltammetry in electroanalytical methods (Scholz F, ed) Springer, Berlin, Germany, 2nd edition
Leger C, Elliott SJ, Hoke KR, Jeuken LJC, Jones AK, Armstrong FA (2003) Enzyme electrokinetics: using protein-film voltammetry to investigate redox enzymes and their mechanism. Biochem 42:8653–8662
Armstrong FA (2020) Voltammetry of proteins in: Encyclopaedia of electrochemistry (Bard AJ, Stratmann M, Wilson GS, eds), Wiley VCH, Weinheim
Jenner LP, Butt JN (2018) Electrochemistry of surface-confined enzymes: inspiration, insight and opportunity for sustainable biotechnology. Curr Opin Eletrochem 8:81–88
Armstrong FA, Heering HA, Hirst J (1997) Reactions of complex metalloproteins studied by protein-film voltammetry. Chem Soc Rev 26:169–179
Fourmond V, Wiedner ES, Shaw WJ, Leger C (2019) Understanding and design of bidirectional and reversible catalysts of multielectron, multistep reactions. J Am Chem Soc 141:11269–11285
Gulaboski R, Mirceski V, Bogeski I, Hoth M (2012) Protein-film voltammetry-electrochemical enzymatic spectroscopy: a review on recent progress. J Solid State Electrochem 16:315–2328
Mirceski V, Gulaboski R, Lovric M, Bogeski I, Kappl R, Hoth M (2013) Square-wave voltammetry: a review on recent progress. Electroanalysis 25:2411–2422
Gulaboski R, Mirceski V (2020) Application of voltammetry in biomedicine–recent achievements in enzymatic voltammetry. Maced J Chem Chem Eng 39:153–166
Janeva M, Kokoskarova P, Gulaboski R (2020) Multistep surface electrode mechanism coupled with preceding chemical reaction — theoretical analysis in square-wave voltammetry. Anal & Bioanal Electrochem 12:766–779
Gulaboski R (2009) Surface ECE mechanism in protein-film voltammetry—a theoretical study under conditions of square-wave voltammetry. J Solid State Electrochem 13:1015–1024
Janeva M, Kokoskarova P, Maksimova V, Gulaboski R (2019) Square-wave voltammetry of two-step surface redox mechanisms coupled with chemical reactions - a theoretical overview. Electroanalysis 31:2488–2506
Gulaboski R, Mihajlov L (2011) Catalytic mechanism in successive two-step protein-film voltammetry — theoretical study in square-wave voltammetry. Biophys Chem 155:1–9
Laborda E, Henstdridge MC, Molina A, Martinez-Ortiz F, Compton RG (2011) A comparison of Marcus-Hush vs. Butler-Volmer electrode kinetics using potential pulse voltammetric techniques. J Electroanal Chem 660:169–177
Ryan MD (1978) The effect of slow two-electron transfers and disproportionation on cyclic voltammograms. J Electrochem Soc 125:547–555
Olmstead ML, Hamilton RG, Nicholson RS (1969) Theory of cyclic voltammetry for a dimerization reaction initiated electrochemically. Anal Chem 41:60–267
Lovric M, Komorsky-Lovric S (2011) Theory of square-wave voltammetry of two-step electrode reaction using an inverse scan direction. Int J Electrochem. https://doi.org/10.4061/2011/538341
Quan M, Sanchez D, Wasylkiw MF, Smith DK (2007) Voltammetry of quinones in unbuffered aqueous solution: reassessing the roles of proton transfer and hydrogen bonding in the aqueous electrochemistry of quinones. J Am Chem Soc 129:12847–12856
Evans DH, Hu K (1996) Inverted potentials in two-electron processes in organic electrochemistry. J Chem Soc Farad Trans 92:3983–3990
Macias-Ruvalcaba NA, Evans DH (2006) Studies of potential inversion in the electrochemical reduction of 11,11,12,12-tetracyano-9,10-anthraquinodimethane and 2,3,5,6-tetramethyl-7,7,8,8-tetracyano-1,4-benzoquinodimethane. J Phys Chem B 110:5155–5160
Amatore C, Gazard S, Maisonhaute E, Pebay C, Schöllhor B, Syssa-Magalé JL, Wadhawan J (2007) Ferrocenyloligo(phenylene‐vinylene) thiols for the construction of self‐assembled monolayers. EurJ Inor Chem 4035–4042 https://doi.org/10.1002/ejic.200700266
Sies H, Parker L (2004) Quinones and quinone enzymes, In: Methods in enzymology. Academic Press, London UK.
Mirceski V, Laborda E, Guziejewski D, Compton RG (2013) New approach to electrode kinetic measurements in square-wave voltammetry: amplitude based quasireversible maximum. Anal Chem 85:5586–5594
Guziejewski D (2020) Electrode mechanisms with coupled chemical reactions-amplitude effect in square-wave voltammetry. J Electroanal Chem 870:114186.
Molina A, Serna C, Li Q, Laborda E, Batchelor-McAuley C, Compton RG (2012) Analytical solutions for the study of multi-electron-transfer processes by staircase, cyclic, and differential voltammetries at disc microelectrodes. J Phys Chem C116:11470–11479
Lopez-Tenes M, Gonzalez J, Molina A (2014) Two-electron transfer reactions in electrochemistry for solution-soluble and surface-confined molecules: a common approach. J Phys Chem C 118:12312–12324
Batchelor-McAuley C, Compton RG (2012) Voltammetry of multi-electron electrode processes of organic species. J Electroanal Chem 669:73–81
Gulaboski R, Mirceski V, Lovric M (2021) Critical aspects in exploring time analysis for the voltammetric estimation of kinetic parameters of surface electrode mechanisms coupled with chemical reaction. Maced J Chem Chem Eng 40:1–9
Mirceski V, Guziejewski D, Lisickov K (2013) Electrode kinetic measurements with square-wave voltammetry at a constant scan rate. Electrochim Acta 114:667–673
Acknowledgements
Rubin Gulaboski thanks the Alexander von Humboldt Foundation (Germany) for the support. Valentin Mirceski acknowledges with gratitude the support through the NATO Grant No. SPS G5550.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
We dedicate this work on the occasion of 75th birthday of professor Gyorgy Inzelt
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gulaboski, R., Mirceski, V. Square-wave voltammetry of two-step diffusional electrode mechanism coupled with a reversible follow-up chemical reaction. J Solid State Electrochem 25, 2893–2901 (2021). https://doi.org/10.1007/s10008-021-05027-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-021-05027-4