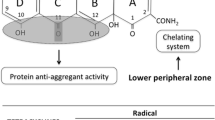
Abstract
The novel antibiotic-exploiting strategy in the treatment of Alzheimer’s (AD) and Parkinson’s (PD) disease has emerged as a potential breakthrough in the field. The research in animal AD/PD models provided evidence on the antiamyloidogenic, anti-inflammatory, antioxidant and antiapoptotic activity of tetracyclines, associated with cognitive improvement. The neuroprotective effects of minocycline and doxycycline in animals initiated investigation of their clinical efficacy in AD and PD patients which led to inconclusive results and additionally to insufficient safety data on a long-standing doxycycline and minocycline therapy in these patient populations. The safety issues should be considered in two levels; in AD/PD patients (particularly antibiotic-induced alteration of gut microbiota and its consequences), and as a world-wide threat of development of bacterial resistance to these antibiotics posed by a fact that AD and PD are widespread incurable diseases which require daily administered long-lasting antibiotic therapy. Recently proposed subantimicrobial doxycycline doses should be thoroughly explored for their effectiveness and long-term safety especially in AD/PD populations. Keeping in mind the antibacterial activity-related far-reaching undesirable effects both for the patients and globally, further work on repurposing these drugs for a long-standing therapy of AD/PD should consider the chemically modified tetracycline compounds tailored to lack antimicrobial but retain (or introduce) other activities effective against the AD/PD pathology. This strategy might reduce the risk of long-term therapy-related adverse effects (particularly gut-related ones) and development of bacterial resistance toward the tetracycline antibiotic agents but the therapeutic potential and desirable safety profile of such compounds in AD/PD patients need to be confirmed.

Similar content being viewed by others
References
Abramov AY, Canevari L, Duchen MR (2004) Calcium signals induced by amyloid β peptide and their consequences in neurons and astrocytes in culture. Biochim Biophys Acta 1742(1–3):81–87. https://doi.org/10.1016/j.bbamcr.2004.09.006
Agwuh KN, MacGowan A (2006) Pharmacokinetics and pharmacodynamics of the tetracyclines including glycylcyclines. J Antimicrob Chemother 58(2):256–265. https://doi.org/10.1093/jac/dkl224
Ahmad W, Ijaz B, Shabbiri K, Ahmed F, Rehman S (2017) Oxidative toxicity in diabetes and Alzheimer’s disease: mechanisms behind ROS/RNS generation. J Biomed Sci 24(1):76. https://doi.org/10.1186/s12929-017-0379-z
Airoldi C, Colombo L, Manzoni C et al (2011) Tetracycline prevents Aβ oligomer toxicity through an atypical supramolecular interaction. Org Biomol Chem 9(2):463–472. https://doi.org/10.1039/c0ob00303d
Akintola AA, van Heemst D (2015) Insulin, aging, and the brain: mechanisms and implications. Front Endocrinol (lausanne) 6:13. https://doi.org/10.3389/fendo.2015.00013
Alzheimer’s Association (2019) 2019 Alzheimer’s disease facts and figures. Alzheimer’s Dementia 15(3):321–387. https://doi.org/10.1016/j.jalz.2019.01.010
Amani M, Shokouhi G, Salari AA (2019) Minocycline prevents the development of depression-like behavior and hippocampal inflammation in a rat model of Alzheimer’s disease. Psychopharmacology 236(4):1281–1292. https://doi.org/10.1007/s00213-018-5137-8
Amin AR, Attur MG, Thakker GD et al (1996) A novel mechanism of action of tetracyclines: effects on nitric oxide synthases. Proc Natl Acad Sci USA 93(24):14014–14019. https://doi.org/10.1073/pnas.93.24.14014
Amin AR, Pattel RN, Thakker GD et al (1997) Post-transcriptional regulation of inducible nitric oxide synthase mRNA in murine macrophages by doxycycline and chemically modified tetracyclines. FEBS Lett 410(2–3):259–264. https://doi.org/10.1016/S0014-5793(97)00605-4
Angelucci F, Cechova K, Amlerova J et al (2019) Antibiotics, gut microbiota, and Alzheimer’s disease. J Neuroinflammation 16(1):1–10. https://doi.org/10.1186/s12974-019-1494-4
Antonenko YN, Rokitskaya TI, Cooper AJL et al (2010) Minocycline chelates Ca2+, binds to membranes, and depolarizes mitochondria by formation of Ca2+-dependent ion channels. J Bioenerg Biomembr 42(2):151–163. https://doi.org/10.1007/s10863-010-9271-1
Appleby BS, Nacopoulos D, Milano N et al (2013) A review: treatment of Alzheimer’s disease discovered in repurposed agents. Dement Geriatr Cogn Disord 35:1–22. https://doi.org/10.1159/000345791
Athauda D, Foltynie T (2016) The glucagon-like peptide 1 (GLP) receptor as a therapeutic target in Parkinson’s disease: mechanisms of action. Drug Discov Today 21(5):802–818. https://doi.org/10.1016/j.drudis.2016.01.013
Avgerinos KI, Kalaitzdis G, Malli A et al (2018) Intranasal insulin in Alzheimer’s dementia or mild cognitive impairment: a systematic review. J Neurol 265(7):1497–1510. https://doi.org/10.1007/s00415-018-8768-0
Avila CL, Chaves S, Socias SB et al (2017) Lessons learned from protein aggregation: toward technological and biomedical applications. Biophys Rev 9(5):501–515. https://doi.org/10.1007/s12551-017-0317-z
Azevedo EP, Ledo JH, Barbosa G et al (2013) Activated microglia mediate synapse loss and short-term memory deficits in a mouse model of transthyretin-related oculoleptomeningeal amyloidosis. Cell Death Dis 4(9):e789. https://doi.org/10.1038/cddis.2013.325
Bahrami F, Morris DL, Pourgholami MH (2011) Tetracyclines: drugs with huge therapeutic potential. Mini Rev Med Chem 12(1):44–52. https://doi.org/10.2174/138955712798868977
Balducci C, Forloni G (2019) Doxycycline for Alzheimer’s disease: fighting β-amyloid oligomers and neuroinflammation. Front Pharmacol 10:738. https://doi.org/10.3389/fphar.2019.00738
Balducci C, Santamaria G, La Vitola P et al (2018) Doxycycline counteracts neuroinflammation restoring memory in Alzheimer’s disease mouse models. Neurobiol Aging 70:128–139. https://doi.org/10.1016/j.neurobiolaging.2018.06.002
Banks WA, Owen JB, Erickson MA (2012) Insulin in the brain: there and back again. Pharmacol Ther 136(1):82–93. https://doi.org/10.1016/j.pharmthera.2012.07.006
Barilar JO, Knezovic A, Babic Perhoc A et al (2020) Shared cerebral metabolic pathology in non-transgenic animal models of Alzheimer’s and Parkinson’s disease. J Neural Transm (vienna) 127(2):231–250. https://doi.org/10.1007/s00702-020-02152-8
Barrientos RM, Kitt MM, Watkins LR et al (2015) Neuroinflammation in the normal aging hippocampus. Neuroscience 309:84–99. https://doi.org/10.1016/j.neuroscience.2015.03.007
Bartlett JG (2004) Antibiotic use in relation to the risk of breast cancer. Infect Dis Clin Pract 12(4):263. https://doi.org/10.1097/01.idc.0000130890.12611.f3
Beauduy CEWL (2018) Tetracyclines, macrolides, clindamycin, chloramphenicol, streptogramins, & oxazolidinones. In: Katzung BG (ed) Basic and clinical pharmacology, 14th edn. McGraw Hill-Education, New York, pp 815–825
Beinenfeld A, Nagler AR, Orlow SJ (2017) Oral antibacterial therapy for Acne vulgaris: an evidence-based review. Am J Clin Dermatol 18(4):469–490. https://doi.org/10.1007/s40257-017-0267-z
BerenS SC, Bird CM, Harrison NA (2020) Minocycline differentially modulates human spatial memory systems. Neuropsychopharmacology 45(13):2162–2169. https://doi.org/10.1038/s41386-020-00811-8
Biscaro B, Lindvall O, Tesco G et al (2012) Inhibition of microglial activation protects hippocampal neurogenesis and improves cognitive deficits in a transgenic mouse model for Alzheimer’s disease. Neurodegener Dis 9(4):187–198. https://doi.org/10.1159/000330363
Böcker R, Mühlberg W, Platt D et al (1986) Serum level, half-life and apparent volume of distribution of doxycycline in geriatric patients. Eur J Clin Pharmacol 30(1):105–108. https://doi.org/10.1007/BF00614205
Bonelli RM, Hödl AK, Hofmann P et al (2004) Neuroprotection in Huntington’s disease : a 2-year study on minocycline. Int Clin Psychopharmacol 19(6):337–342. https://doi.org/10.1097/00004850-200411000-00004
Bortolanza M, Nascimento GC, Socias SB et al (2018) Tetracycline repurposing in neurodegeneration: focus on Parkinson’s disease. J Neural Transm (vienna) 125(10):1403–1415. https://doi.org/10.1007/s00702-018-1913-1
Bortolanza M, Nascimento GC, Raisman-Vozari R et al (2021) Doxycycline and its derivative, COL-3, decrease dyskinesia induced by l-DOPA in hemiparkinsonian rats. Br J Pharmacol 178(13):2595–2616. https://doi.org/10.1111/bph.15439
Bradley-Whitman MA, Lovell MA (2013) Epigenetic changes in the progression of Alzheimer’s disease. Mech Ageing Dev 134(10):486–495. https://doi.org/10.1016/j.mad.2013.08.005
Brochard V, Comadiere B, Prigent A et al (2009) Infiltration of CD4+ lymphocytes into the brain contributes to neurodegeneration in a mouse model of Parkinson disease. J Clin Invest 119(1):182–192. https://doi.org/10.1172/JCI36470
Brown GC (2007) Mechanisms of inflammatory neurodegeneration: INOS and NADPH oxidase. Biochem Soc Trans 35(5):1119–1121. https://doi.org/10.1042/BST0351119
Butterfield AD, Boyd-Kimball D (2018) Oxidative stress, amyloid-β peptide, and altered key molecular pathways in the pathogenesis and progression of Alzheimer’s disease. J Alzheimers Dis 62(3):1345–1367. https://doi.org/10.3233/JAD-170543
Cai H, Cong W, Ji S et al (2012) Metabolic dysfunction in Alzheimers disease and related neurodegenerative disorders. Curr Alzheimer Res 9(1):5–17. https://doi.org/10.2174/156720512799015064
Cai Z, Yan Y, Wang Y (2013) Minocycline alleviates beta-amyloid protein and tau pathology via restraining neuroinflammation induced by diabetic metabolic disorder. Clin Interv Aging 8:1089–1095. https://doi.org/10.2147/CIA.S46536
Calsolaro V, Edison P (2016) Neuroinflammation in Alzheimer’s disease: current evidence and future directions. Alzheimers Dement 12(6):719–732. https://doi.org/10.1016/j.jalz.2016.02.010
Cankaya S, Cankaya B, Kilic U et al (2019) The therapeutic role of minocycline in Parkinson’s disease. Drugs Context 8:212553. https://doi.org/10.7573/dic.212553
Cardoso I, Merlini G, Saraiva MJ (2003) 4′-Iodo-4′-deoxydoxorubicin and tetracyclines disrupt transthyretin amyloid fibrils in vitro producing noncytotoxic species: screening for TTR fibril disrupters. FASEB J 17(8):803–809. https://doi.org/10.1096/fj.02-0764com
Chalmers JR, Wojnarowska F, Kirtsching G et al (2017) A randomised controlled trial to compare the safety, effectiveness and cost-effectiveness of doxycycline (200 mg/day) with that of oral prednisolone (0.5 mg/kg/day) for initial treatment of bullous pemphigoid: the Bullous Pemphigoid Steroids and Tetracyclines (BLISTER) trial. Health Technol Assess 21(10):1–90. https://doi.org/10.3310/hta21100
Cheignon C, Tomas M, Bonnefont-Rousselot D et al (2018) Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Biol 14:450–464. https://doi.org/10.1016/j.redox.2017.10.014
Chen M, Ona VO, Li M et al (2000) Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nat Med 6(7):797–801
Chen S, Yin JH, Hwang CH et al (2012) Anti-apoptotic and anti-oxidative mechanisms of minocycline against sphingomyelinase/ceramide neurotoxicity: implication in Alzheimer’s disease and cerebral ischemia. Free Radic Res 46(8):940–950. https://doi.org/10.3109/10715762.2012.674640
Chen WW, Zhang X, Huang WJ (2016) Role of neuroinflammation in neurodegenerative diseases (Review). Mol Med Rep 13(4):3391–3396. https://doi.org/10.3892/mmr.2016.4948
Chêne G, Beiser A, Au R et al (2015) Gender and incidence of dementia in the Framingham Heart Study from mid-adult life. Alzheimers Dement 11(3):310–320. https://doi.org/10.1016/j.jalz.2013.10.005
Choi Y, Kim HS, Shin KY et al (2007) Minocycline attenuates neuronal cell death and improves cognitive impairment in Alzheimer’s disease models. Neuropsychopharmacology 32(11):2393–2404. https://doi.org/10.1038/sj.npp.1301377
Chopra I, Hawkey PM, Hinton M (1992) Tetracyclines, molecular and clinical aspects. J Antimicrob Chemother 29(3):245–277. https://doi.org/10.1093/jac/29.3.245
Chow HM, Herrup K (2015) Genomic integrity and the ageing brain. Nat Rev Neurosci 16(11):672–684. https://doi.org/10.1038/nrn4020
Chtarto A, Humbert-Claude M, Bockstael O et al (2016) A regulatable AAV vector mediating GDNF biological effects at clinically-approved sub-antimicrobial doxycycline doses. Mol Ther Methods Clin Dev 5:16027. https://doi.org/10.1038/mtm.2016.27
Colaizzi JL, Klink PR (1969) pH-partition behavior of tetracyclines. J Pharm Sci 58(10):1184–1189. https://doi.org/10.1002/jps.2600581003
Costa R, Speretta E, Crowther D et al (2011) Testing the therapeutic potential of doxycycline in a Drosophila melanogaster model of Alzheimer disease. J Biol Chem 286(48):41647–41655. https://doi.org/10.1074/jbc.M111.274548
Craft S, Watson GS (2004) Insulin and neurodegenerative disease: shared and specific mechanisms. Lancet Neurol 3(3):169–178. https://doi.org/10.1016/S1474-4422(04)00681-7
Craft S, Peskind E, Schwartz, MW et al (1998) Cerebrospinal fluid and plasma insulin levels in Alzheimer’s disease: relationship to severity of dementia and apolipoprotein E genotype. Neurology 50(1):164–168. https://doi.org/10.1212/WNL.50.1.164
Cryan JF, O’Riordan KJ, Cowan CSM et al (2019) The microbiota-gut-brain axis. Physiol Rev 99(4):1877–2013. https://doi.org/10.1152/physrev.00018.2018
Cuello AC, Ferretti MT, Leon WC et al (2010) Early-stage inflammation and experimental therapy in transgenic models of the Alzheimer-like amyloid pathology. Neurodegener Dis 7(1–3):96–98. https://doi.org/10.1159/000285514
De Felice FG, Lourenco MV, Ferreira ST (2014) How does brain insulin resistance develop in Alzheimer’s disease? Alzheimers Dement 10(1 Suppl):S26–S32. https://doi.org/10.1016/j.jalz.2013.12.004
De La Monte SM (2017) Insulin resistance and neurodegeneration: progress towards the development of new therapeutics for Alzheimer’s disease. Drugs 77(1):47–65. https://doi.org/10.1007/s40265-016-0674-0
De La MonteTong SMM (2014) Brain metabolic dysfunction at the core of Alzheimer’s disease. Biochem Pharmacol 88(4):548–559. https://doi.org/10.1016/j.bcp.2013.12.012
Di Caprio R, Lembo S, Di Costanzo L et al (2015) Anti-inflammatory properties of low and high doxycycline doses: an in vitro study. Mediators Inflamm 2015:329418. https://doi.org/10.1155/2015/329418
Diguet E, Fernagut PO, Wei X et al (2004) Deleterious effects of minocycline in animal models of Parkinson’s disease and Huntington’s disease. Eur J Neurosci 19(12):3266–3276. https://doi.org/10.1111/j.0953-816X.2004.03372.x
Dobson CM, Knowles TPJ, Vendruscolo M (2020) The amyloid phenomenon and its significance in biology and medicine. Cold Spring Harb Perspect Biol 12(2):a033878. https://doi.org/10.1101/cshperspect.a033878
Dodel R, Spottke A, Gerhard A et al (2010) Minocycline 1-year therapy in multiple-system-atrophy: effect on clinical symptoms and [11C] (R)-PK11195 PET (MEMSA-trial). Mov Disord 25(1):97–107. https://doi.org/10.1002/mds.22732
Dominguez-Meijide A, Parrales V, Vasili E et al (2021) Doxycycline inhibits α-synuclein-associated pathologies in vitro and in vivo. Neurobiol Dis 151:105256. https://doi.org/10.1016/j.nbd.2021.105256
Dorsey R, Sherer T, Okun MS (2018) The emerging evidence of the Parkinson pandemic. J Parkinsons Dis 8(Suppl 1):S3–S8. https://doi.org/10.3233/JPD-181474
Dotevall L, Hagberg L (1989) Penetration of doxycycline into cerebrospinal fluid in patients treated for suspected lyme neuroborreliosis. Antimicrob Agents Chemother 33(7):1078–1080. https://doi.org/10.1128/AAC.33.7.1078
Dotevall L, Hagberg L (1999) Successful oral doxycycline treatment of lyme disease-associated facial palsy and meningitis. Clin Infect Dis 28(3):569–574. https://doi.org/10.1086/515145
Du Y, Ma Z, Lin S et al (2001) Minocycline prevents nigrostriatal dopaminergic neurodegeneration in the MPTP model of Parkinson’s disease. Proc Natl Acad Sci USA 98(25):14669–14674. https://doi.org/10.1073/pnas.251341998
Dugger BN, Dickson DW (2017) Pathology of neurodegenerative diseases. Cold Spring Harb Perspect Biol 9(7):a028035. https://doi.org/10.1101/cshperspect.a028035
Egeberg A, Hansen PR, Gislason GH et al (2016) Exploring the association between Rosacea and Parkinson disease: a Danish Nationwide Cohort Study. JAMA Neurol 73(5):529–534. https://doi.org/10.1001/jamaneurol.2016.0022
Elbaz A, Carcaillon L, Kab S et al (2015) Epidemiology of Parkinson ’ s disease. Rev Neurol (paris) 1580(1):1–13. https://doi.org/10.1016/j.neurol.2015.09.012
Elewa HF, Hilali H, Hess DC et al (2006) Minocycline for short-term neuroprotection. Pharmacotherapy 26(4):515–521
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35(4):495–516. https://doi.org/10.1080/01926230701320337
Erekat NS (2018) Apoptosis and its role in Parkinson’s disease. In: Stoker TB, Greenland JC (eds) Parkinson’s disease: pathogenesis and clinical aspects, chapter 4. Codon Publications, Brisbane
Ethell DW, Buhler LA (2003) Fas ligand-mediated apoptosis in degenerative disorders of the brain. J Clin Immunol 23(6):439–446. https://doi.org/10.1023/B:JOCI.0000010420.96419.a8
Familian A, Boshuizen RS, Eikelenboom P et al (2006) Inhibitory effect of minocycline on amyloid β fibril formation and human microglial activation. Glia 53(3):233–240. https://doi.org/10.1002/glia.20268
Fan R, Xu F, Previti ML et al (2007) Minocycline reduces microglial activation and improves behavioral deficits in a transgenic model of cerebral microvascular amyloid. J Neurosci 27(12):3057–3063. https://doi.org/10.1523/JNEUROSCI.4371-06.2007
Ferretti MT, Allard S, Partridge V et al (2012) Minocycline corrects early, pre-plaque neuroinflammation and inhibits BACE-1 in a transgenic model of Alzheimer’s disease-like amyloid pathology. J Neuroinflammation 9:62. https://doi.org/10.1186/1742-2094-9-62
Forloni G, Colombo L, Girola L et al (2001) Anti-amyloidogenic activity of tetracyclines: studies in vitro. FEBS Lett 487(3):404–407. https://doi.org/10.1016/S0014-5793(00)02380-2
Forloni G, Terreni L, Bertani I et al (2002) Protein misfolding in Alzheimer’s and Parkinson’s disease: genetics and molecular mechanisms. Neurobiol Aging 23(5):957–976. https://doi.org/10.1016/S0197-4580(02)00076-3
Forloni G, Salmona M, Marcon G (2009) Tetracyclines and prion infectivity. Infect Disord Drug Targets 9(1):23–30. https://doi.org/10.2174/1871526510909010023
Forloni G, Salmona M, Marcon G et al (2012) Tetracyclines and prion infectivity. Infect Disord Drug Targets 9(1):23–30. https://doi.org/10.2174/1871526510909010023
Forloni G, Tettamanti M, Lucca U et al (2015) Preventive study in subjects at risk of fatal familial insomnia: Innovative approach to rare diseases. Prion 9(2):75–79. https://doi.org/10.1080/19336896.2015.1027857
Forloni G, Roiter I, Tagliavini F (2019) Clinical trials of prion disease therapeutics. Curr Opin Pharmacol 44:53–60. https://doi.org/10.1016/j.coph.2019.04.019
Forloni G, La Vitola P, Cerovic M et al (2021) Inflammation and Parkinson’s disease pathogenesis: mechanisms and therapeutic insight. Prog Mol Biol Transl Sci 177:175–202. https://doi.org/10.1016/bs.pmbts.2020.11.001
Friedman GD, Oestreicher N, Chan J et al (2006) Antibiotics and risk of breast cancer: up to 9 years of follow-up of 2.1 million women. Cancer Epidemiol Biomarkers Prev 15(11):2102–2106. https://doi.org/10.1158/1055-9965.EPI-06-0401
Frölich L, Blum-Degen D, Bernstein HG et al (1998) Brain insulin and insulin receptors in aging and sporadic Alzheimer’s disease. J Neural Transm (vienna) 105(4–5):423–438. https://doi.org/10.1007/s007020050068
Fuoco D (2012) Classification framework and chemical biology of tetracycline-structure-based drugs. Antibiotics 1:1–13. https://doi.org/10.3390/antibiotics1010001
Galasko D, Montine TJ (2010) Biomarkers of oxidative damage and inflammation in Alzheimers disease. Biomark Med 4(1):27–36. https://doi.org/10.2217/bmm.09.89
Gandhi S, Abramov AY (2012) Mechanism of oxidative stress in neurodegeneration. Oxid Med Cell Longev 2012:428010. https://doi.org/10.1155/2012/428010
Garcez ML, Mina F, Bellettini-Santos T et al (2017) Minocycline reduces inflammatory parameters in the brain structures and serum and reverses memory impairment caused by the administration of amyloid β (1–42) in mice. Prog Neuropsychopharmacol Biol Psychiatry 77:23–31. https://doi.org/10.1016/j.pnpbp.2017.03.010
Garcia-Martinez EM, Sanz-Blasco S, Karachitos A et al (2010) Mitochondria and calcium flux as targets of neuroprotection caused by minocycline in cerebellar granule cells. Biochem Pharmacol 79(2):239–250. https://doi.org/10.1016/j.bcp.2009.07.028
Garrett JPD, Margolis DJ (2012) Impact of long-term antibiotic use for acne on bacterial ecology and health outcomes: a review of observational studies. Curr Dermatol Rep 1(1):23–28. https://doi.org/10.1007/s13671-011-0001-7
Garrido-Mesa N, Zarzuelo A, Gálvez J (2013) Minocycline: far beyond an antibiotic. Br J Pharmacol 169(2):337–352. https://doi.org/10.1111/bph.12139
Garwood CJ, Cooper JD, Hanger DP et al (2010) Anti-inflammatory impact of minocycline in a mouse model of tauopathy. Front Psychiatry 1:136. https://doi.org/10.3389/fpsyt.2010.00136
Gasparrini AJ, Markley JL, Kumar H (2020) Tetracycline-inactivating enzymes from environmental, human commensal, and pathogenic bacteria cause broad-spectrum tetracycline resistance. Commun Biol 3(1):241. https://doi.org/10.1038/s42003-020-0966-5
Gautieri A, Beeg M, Gobbi M et al (2019) The anti-amyloidogenic action of doxycycline: a molecular dynamics study on the interaction with Aβ42. Int J Mol Sci 20(18):4641. https://doi.org/10.3390/ijms20184641
GBD (2016) Neurology Collaborators (2019) Global, regional, and national burden of Parkinson’s disease, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol 18(5):459–480. https://doi.org/10.1016/S1474-4422(18)30295-3
Giorgetti S, Raimondi S, Pagano K et al (2011) Effect of tetracyclines on the dynamics of formation and destructuration of β2-microglobulin amyloid fibrils. J Biol Chem 286(3):2121–2131. https://doi.org/10.1074/jbc.M110.178376
Giovannini MG, Scali C, Prosperi C et al (2002) β-amyloid-induced inflammation and cholinergic hypofunction in the rat brain in vivo: involvement of the p38MAPK pathway. Neurobiol Dis 11(2):257–274. https://doi.org/10.1006/nbdi.2002.0538
González-Lizárraga F, Socias S, Avila CL et al (2017) Repurposing doxycycline for synucleinopathies: remodelling of α-synuclein oligomers towards non-toxic parallel beta-sheet structured species. Sci Rep 7:41755. https://doi.org/10.1038/srep41755
González-Lizárraga F, Ploper D, Avila CL et al (2020) CMT-3 targets different α-synuclein aggregates mitigating their toxic and inflammogenic effects. Sci Rep 10(1):20258. https://doi.org/10.1038/s41598-020-76927-0
Good ML, Hussey DL (2003) Minocycline: stain devil? Brit J Dermatol 149(2):237–239. https://doi.org/10.1046/j.1365-2133.2003.05497.x
Gordon PH, Moore DH, Miller RG et al (2007) Efficacy of minocycline in patients with amyotrophic lateral sclerosis: a phase III randomised trial. Lancet Neurol 6(12):1045–1053. https://doi.org/10.1016/S1474-4422(07)70270-3
Griffin MO, Fricovsky E, Ceballos G et al (2010) tetracyclines: a pleitropic family of compounds with promising therapeutic properties. Review of the literature. Am J Physiol Cell Physiol 299(3):C539–C548. https://doi.org/10.1152/ajpcell.00047.2010
Grossman TH (2016) Tetracycline antibiotics and resistance. Cold Spring Harb Perspect Med 6(4):a025387. https://doi.org/10.1101/cshperspect.a025387
Gu Y, Walker C, Ryan ME et al (2012) Non-antibacterial tetracycline formulations: clinical applications in dentistry and medicine. J Oral Microbiol 4:1–14. https://doi.org/10.3402/jom.v4i0.19227
Gubellini P, Kachidian P (2015) Animal models of Parkinson’s disease: an updated overview. Rev Neurol (paris) 171(11):750–761. https://doi.org/10.1016/j.neurol.2015.07.011
Gyengesi E, Münch G (2020) In search of an anti-inflammatory drug for Alzheimer disease. Nat Rev Neurol 16(3):131–132. https://doi.org/10.1038/s41582-019-0307-9
Hagelmaier T, Lebbing M, Duscha A et al (2020) interventional influence of the intestinal microbiome through dietary intervention and bowel cleansing might improve motor symptoms in Parkinson’s disease. Cells 9(2):376
Haïk S, Marcon G, Mallet A et al (2014) Doxycycline in Creutzfeldt-Jakob disease: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet Neurol 13(2):150–158. https://doi.org/10.1016/S1474-4422(13)70307-7
He Y, Appel S, Le W (2001) Minocycline inhibits microglial activation and protects nigral cells after 6-hydroxydopamine injection into mouse striatum. Brain Res 909(1–2):187–193. https://doi.org/10.1016/S0006-8993(01)02681-6
He Y, Li H, Sun D et al (2020) Gut microbiota: implications in Alzheimer’s disease. J Clin Med 9(7):2042. https://doi.org/10.3390/jcm9072042
Hebert LE, Weuve J, Scherr PA et al (2013) Alzheimer diease in the US (2010–2050) estimated using the 2010 census. Neurology 80(19):1778–1783
Hirsch EC, Standaert DG (2021) Ten unsolved questions about neuroinflammation in Parkinson’s disease. Mov Disord 36(1):16–24. https://doi.org/10.1002/mds.28075
Holmes C (2017) Inflammation in Alzheimer’s disease. In: Ames D, O’Brien JT, Burns A (eds) Dementia, 5th edn. CRC Press, Boca Raton
Holmes WM, Klaips CL, Serio TR (2014) Defining the limits: protein aggregation and toxicity in vivo. Crit Rev Biochem Mol Biol 49(4):294–303. https://doi.org/10.3109/10409238.2014.914151
Hou Y, Dan X, Babbar M et al (2019) Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 15(10):565–581. https://doi.org/10.1038/s41582-019-0244-7
Howard R, Zubko O, Bradley R et al (2020) Minocycline at 2 different dosages vs placebo for patients with mild alzheimer disease: a randomized clinical trial. JAMA Neurol 77(2):164–174. https://doi.org/10.1001/jamaneurol.2019.3762
Hoyer S (2002) The aging brain. Changes in the neuronal insulin/insulin receptor signal transduction cascade trigger late-onset sporadic Alzheimer disease (SAD). A mini-review. J Neural Transm 109(7–8):991–1002. https://doi.org/10.1007/s007020200082
Hoyer S (2004) Glucose metabolism and insulin receptor signal transduction in alzheimer disease. Eur J Pharmacol 490(1–3):115–125. https://doi.org/10.1016/j.ejphar.2004.02.049
https://clinicaltrials.gov/ct2/show/NCT01463384 (no date). Accessed 9 Decembre 2021
https://www.clinicaltrials.gov/ct2/show/NCT03958708?term=antibiotics&cond=Parkinson+Disease&draw=2&rank=8 (no date). Accessed 9 December 2021
https://www.clinicaltrials.gov/ct2/show/record/NCT03575195?term=antibiotics&cond=Parkinson+Disease&draw=2&rank=3 (no date). Accessed 9 December 2021
https://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-alzheimers-drug. Accessed 9 December 2021
Huang WJ, Zhang X, Chen WW (2016) Role of oxidative stress in Alzheimer’s disease (review). Biomed Rep 4(5):519–522. https://doi.org/10.3892/br.2016.630
Huang LK, Chao SP, Hu CJ (2020) Clinical trials of new drugs for Alzheimer disease. J Biomed Sci 27(1):18. https://doi.org/10.1186/s12929-019-0609-7
Hunter CL, Quintero EM, Gilstrap L et al (2004) Minocycline protects basal forebrain cholinergic neurons from mu p75-saporin immunotoxic lesioning. Eur J Neurosci 19(12):3305–3316. https://doi.org/10.1111/j.0953-816X.2004.03439.x
Huntington Study Group DOMINO Investigators (2010) A futility study of minocycline in Huntington’s disease. Mov Disord 25(13):2219–2224. https://doi.org/10.1002/mds.23236
Ian C, Marilyn R (2001) Tetracycline antibiotics: mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol Mol Biol Rev 65(3):232–260. https://doi.org/10.1128/MMBR.65.2.232
Jagmag SA, Tripathi N, Shukla S et al (2016) Evaluation of models of Parkinson’s disease. Front Neurosci 9:503. https://doi.org/10.3389/fnins.2015.00503
Jenkins EC, O’Connell MJ, Manfredi G et al (2021) Doxycycline promotes proteasome fitness in the central nervous system. Sci Rep 11(1):17003. https://doi.org/10.1038/s41598-021-96540-z
Jones RN, Flonta M, Gurler N et al (2014) Resistance surveillance program report for selected European nations (2011). Diagn Microbiol Infect Dis 78(4):429–436. https://doi.org/10.1016/j.diagmicrobio.2013.10.008
Kaeberlein M, Galvan V (2019) Rapamycin and Alzheimer’s disease: time for a clinical trial? Sci Transl Med 11(476):1–6. https://doi.org/10.1126/scitranslmed.aar4289
Kamat PK, Kalani A, Rai SR et al (2016) Mechanism of oxidative stress and synapse dysfunction in the pathogenesis of Alzheimer’s disease: understanding the therapeutics strategies. Mol Neurobiol 53(1):648–661. https://doi.org/10.1007/s12035-014-9053-6
Karlsson M, Hammers S, Nilsson-Ehle I et al (1996) Concentrations of doxycycline and penicillin G in sera and cerebrospinal fluid of patients treated for neuroborreliosis. Antimicrob Agents Chemother 40(5):1104–1107. https://doi.org/10.1128/aac.40.5.1104
Keerthisinghe TP, Wang M, Zhang Y et al (2019) Low-dose tetracycline exposure alters gut bacterial metabolism and host-immune response: “Personalized” effect? Environ Int 131:104989. https://doi.org/10.1016/j.envint.2019.104989
Keijmel SP, Delsing CE, Bleijenberg G et al (2017) Effectiveness of long-term doxycycline treatment and cognitive-behavioral therapy on fatigue severity in patients with Q fever fatigue syndrome (Qure Study): a randomized controlled trial. Clin Infect Dis 64(8):998–1005. https://doi.org/10.1093/cid/cix013
Keller AF, Gravel M, Kriz J (2011) Treatment with minocycline after disease onset alters astrocyte reactivity and increases microgliosis in SOD1 mutant mice. Exp Neurol 228(1):69–79. https://doi.org/10.1016/j.expneurol.2010.12.010
Kempuraj D, Thangavel R, Natteru PA et al (2016) Neuroinflammation induces neurodegeneration. J Neurol Neurosurg Spine 1(1):1003
Kim HS, Suh YH (2009) Minocycline and neurodegenerative diseases. Behav Brain Res 196(2):168–179. https://doi.org/10.1016/j.bbr.2008.09.040
Kim DS, Ko BS, Ryuk JA et al (2020a) Tetragonia tetragonioides protected against memory dysfunction by elevating hippocampal amyloid-β deposition through potentiating insulin signaling and altering gut microbiome composition. Int J Mol Sci 21(8):2900. https://doi.org/10.3390/ijms21082900
Kim MS, Kim Y, Choi H et al (2020b) Transfer of a healthy microbiota reduces amyloid and tau pathology in an Alzheimer’s disease animal model. Gut 69:283–294. https://doi.org/10.1136/gutjnl-2018-317431
Korting HC, Schöllmann C (2009) Tetracycline actions relevant to rosacea treatment. Skin Pharmacol Physiol 22(6):287–294. https://doi.org/10.1159/000235550
Kovacs GG (2019) Molecular pathology of neurodegenerative diseases: principles and practice. J Clin Pathol 72(11):725–735. https://doi.org/10.1136/jclinpath-2019-205952
Kraus RL, Pasieczny R, Lariosa-Willingham K et al (2005) Antioxidant properties of minocycline: neuroprotection in an oxidative stress assay and direct radical-scavenging activity. J Neurochem 94(3):819–827. https://doi.org/10.1111/j.1471-4159.2005.03219.x
Kriz J, Nguyen MD, Julien JP (2002) Minocycline slows disease progression in a mouse model of amyotrophic lateral sclerosis. Neurobiol Dis 10(3):268–278. https://doi.org/10.1006/nbdi.2002.0487
Kupsch K, Hertel S, Kreutzmann P et al (2009) Impairment of mitochondrial function by minocycline. FEBS J 276(6):1729–1738. https://doi.org/10.1111/j.1742-4658.2009.06904.x
Ladewig P (1945) Double-refringence of the amyloid-Congo-red-complex in histological sections. Nature 156:81–82
Lavigna G, Masone A, Bouybayoune I et al (2021) Doxycycline rescues recognition memory and circadian motor rhythmicity but does not prevent terminal disease in fatal familial insomnia mice. Neurobiol Dis 158:105455. https://doi.org/10.1016/j.nbd.2021.105455
Lazzarini M, Martin S, Mitkovski M et al (2013) Doxycycline restrains glia and confers neuroprotection in a 6-OHDA Parkinson model. Glia 61(7):1084–1100. https://doi.org/10.1002/glia.22496
Levy RM, Huang EY, Roling D et al (2003) Effect of antibiotics on the oropharyngeal flora in patients with acne. Arch Dermatol 139(4):467–471. https://doi.org/10.1001/archderm.139.4.467
Li C, Yuan K, Schluesener H (2013) Impact of minocycline on neurodegenerative diseases in rodents: a meta-analysis. Rev Neurosci 24(5):553–562. https://doi.org/10.1515/revneuro-2013-0040
Liguori I, Russo G, Curcio F et al (2018) Oxidative stress, aging, and diseases. Clin Interv Aging 13:757–772. https://doi.org/10.2147/CIA.S158513
Liu H, Su W, Li S et al (2017) Eradication of Helicobacter pylori infection might improve clinical status of patients with Parkinson’s disease, especially on bradykinesia. Clin Neurol Neurosurg 160:101–104. https://doi.org/10.1016/j.clineuro.2017.07.003
Loeb MB, Molloy DW, Smieja M et al (2004) A randomized, controlled trial of doxycycline and rifampin for patients with Alzheimer’s disease. J Am Geriatr Soc 52(3):381–387. https://doi.org/10.1111/j.1532-5415.2004.52109.x
Lucchetti J, Fracasso C, Balducci C et al (2019) Plasma and brain concentrations of doxycycline after single and repeated doses in wild-type and APP23 mices. J Pharmacol Exp Ther 368(1):32–40. https://doi.org/10.1124/jpet.118.252064
Luo J, Mills K, Le Cessi S et al (2020) Ageing, age-related diseases and oxidative stress: what to do next? Ageing Res Rev 57:100982. https://doi.org/10.1016/j.arr.2019.100982
Ma T (2014) GSK3 in Alzheimer’s disease: mind the isoforms. J Alzheimers Dis 39(4):707–710. https://doi.org/10.3233/JAD-131661
Macdonald H, Kelly RG, Allen ES et al (1973) Pharmacokinetic studies on minocycline in man. Clin Pharmacol Ther 14(5):852–861. https://doi.org/10.1002/cpt1973145852
Maciejczyk M, Żebrowska E, Chabowski A (2019) Insulin resistance and oxidative stress in the brain: what’s new? Int J Mol Sci 20(4):874. https://doi.org/10.3390/ijms20040874
Mantzavinos V, Alexiou A (2017) Biomarkers for Alzheimer’s disease diagnosis. Curr Alzheimer Res 14(11):1149–1154. https://doi.org/10.2174/1567205014666170203125942
Margolis DJ, Fanelli M, Hoffstad O et al (2005) Antibiotic treatment of acne may be associated with upper respiratory tract infections. Arch Dermatol 141(9):1132–1136. https://doi.org/10.1001/archderm.141.9.1132
Margolis DJ, Hoffstad O, Bilker W (2007) Association or lack of association between tetracycline class antibiotics used for acne vulgaris and lupus erythematosus. Br J Dermatol 157(3):540–546. https://doi.org/10.1111/j.1365-2133.2007.08056.x
Margolis DJ, Fanelli M, Hoffstad O et al (2010) Potential association between the oral tetracycline class of antimicrobials used to treat acne and inflammatory bowel disease. Am J Gastroenterol 105(12):2610–2616. https://doi.org/10.1038/ajg.2010.303
Margolis DJ, Fanelli M, Kupperman E et al (2012) Association of pharyngitis with oral antibiotic use for the treatment of acne: a cross-sectional and prospective cohort study. Arch Dermatol 148(3):326–332. https://doi.org/10.1001/archdermatol.2011.355
Matthews KA, Xu W, Gaglioti A et al (2019) Racial and ethnic estimates of Alzheimer’s disease and related dementias in the United States (2015–2060) in adults aged ≥65 years. Alzheimers Dement 15(1):17–24. https://doi.org/10.1016/j.jalz.2018.06.3063
McGeer PL, Itagaki S, Boyes BE et al (1988) Reactive microglia are positive for HLA-DR in the: substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 38(8):1285–1291. https://doi.org/10.1212/wnl.38.8.1285
McKeage K, Deeks ED (2010) Doxycycline 40 mg capsules (30 mg immediate-release/10 mg delayed-release beads): anti-inflammatory dose in rosacea. Am J Clin Dermatol 11(3):217–222. https://doi.org/10.2165/11204850-000000000-00000
Medina L, González-Lizárraga F, Dominguez-Meijide A et al (2021) Doxycycline interferes with tau aggregation and reduces its neuronal toxicity. Front Aging Neurosci 13:635760. https://doi.org/10.3389/fnagi.2021.635760
Mendes RE, Farrell DJ, Sader HS et al (2015) Update of the telavancin activity in vitro tested against a worldwide collection of Gram-positive clinical isolates (2013), when applying the revised susceptibility testing method. Diagn Microbiol Infect Dis 81(4):275–279. https://doi.org/10.1016/j.diagmicrobio.2014.12.011
Merati Z, Farshad A, Farzinpour A et al (2018) Anti-apoptotic effects of minocycline on ram epididymal spermatozoa exposed to oxidative stress. Theriogenology 114:266–272. https://doi.org/10.1016/j.theriogenology.2018.03.041
Merlini G, Ascari E, Amboldi N et al (1995) Interaction of the anthracycline 4’-iodo-4’-deoxydoxorubicin with amyloid fibrils: inhibition of amyloidogenesis. Proc Natl Acad Sci USA 92(7):2959–2963. https://doi.org/10.1073/pnas.92.7.2959
Merlini G, Dispenzieri A, Sanchorawala V et al (2018) Systemic immunoglobulin light chain amyloidosis. Nat Rev Dis Primers 4(1):38. https://doi.org/10.1038/s41572-018-0034-3
Mertsalmi TH, Pekkonen E, Scheperjans F (2020) Antibiotic exposure and risk of Parkinson’s disease in Finland: a nationwide case-control study. Mov Disord 35(3):431–442. https://doi.org/10.1002/mds.27924
Młynarczyk-Bonikowska B, Majewska A, Malejczyk M et al (2020) Multiresistant Neisseria gonorrhoeae: a new threat in second decade of the XXI century. Med Microbiol Immunol 209(2):95–108. https://doi.org/10.1007/s00430-019-00651-4
Molloy DW, Standish TI, Zhou Q et al (2013) A multicenter, blinded, randomized, factorial controlled trial of doxycycline and rifampin for treatment of Alzheimer’s disease: the DARAD trial. Int J Geriatr Psychiatry 28(5):463–470. https://doi.org/10.1002/gps.3846
Montagna P, Gambetti P, Cortelli P (2003) Familial and sporadic fatal insomnia. Lancet Neurol 2(3):167–176. https://doi.org/10.1016/s1474-4422(03)00323-5
Moroo I, Yamada T, Makino H et al (1994) Loss of insulin receptor immunoreactivity from the substantia nigra pars compacta neurons in Parkinson’s disease. Acta Neuropathol 87(4):343–348. https://doi.org/10.1007/BF00313602
Mozafari H, Amiri S, Mehr SE et al (2020) Minocycline attenuates depressive-like behaviors in mice treated with the low dose of intracerebroventricular streptozotocin; the role of mitochondrial function and neuroinflammation. Mol Biol Rep 47(8):6143–6153. https://doi.org/10.1007/s11033-020-05696-w
Nagatsu T, Sawada M (2005) Inflammatory process in Parkinsons disease: role for cytokines. Curr Pharm Des 11(8):999–1016. https://doi.org/10.2174/1381612053381620
Nau R, Sörgel F, Eiffert H (2010) Penetration of drugs through the blood-cerebrospinal fluid/blood-brain barrier for treatment of central nervous system infections. Clin Microbiol Rev 23(4):858–883. https://doi.org/10.1128/CMR.00007-10
Nikodemova M, Duncan ID, Watters JJ (2006) Minocycline exerts inhibitory effects on multiple mitogen-activated protein kinases and IκBα degradation in a stimulus-specific manner in microglia. J Neurochem 96(2):314–323. https://doi.org/10.1111/j.1471-4159.2005.03520.x
NINDS NET-PD Investigators (2006) A randomized, double-blind, futility clinical trial of creatine and minocycline in early Parkinson disease. Neurology 66(5):664–671
NINDS NET-PD Investigators (2008) A pilot clinical trial of creatine and minocycline in early Parkinson disease: 18-month results. Clin Neuropharmacol 31(3):141–150. https://doi.org/10.1097/WNF.0b013e3181342f32
Niu H, Alvarez-Alvarez I, Guillen-Grima F et al (2017) Prevalence and incidence of Alzheimer’s disease in Europe: a meta-analysis. Neurologia SEGO 32(8):523–532. https://doi.org/10.1016/j.nrl.2016.02.016
Noble W, Garwood C, Stephenson J et al (2009) Minocycline reduces the development of abnormal tau species in models of Alzheimer’s disease. FASEB J 23(3):739–750. https://doi.org/10.1096/fj.08-113795
OIE (2021). Fifth OIE annual report on the use of antimicrobial agents in animals. Better understanding of the global situation. https://www.oie.int/en/document/fifth-oie-annual-report-on-antimicrobial-agents-intended-for-use-in-animals/. Accessed 9 December 2021
Orsucci D, Calsolaro V, Mancuso M et al (2009) Neuroprotective effects of tetracyclines: molecular targets, animal models and human disease. CNS Neurol Disord Drug Targets 8(3):222–231
Parachikova A, Vasilevko V, Cribbs DH et al (2010) Reductions in amyloid-beta-derived neuroinflammation, with minocycline, restore cognition but do not significantly affect tau hyperphosphorylation. J Alzheimers Dis 21(2):527–542
Perdue BE, Standiford HC (1999) Tetracyclines. In: Yu VL, Merigan TC, Barriere SL (eds) Antimicrobial therapy and Vaccines. Williams and Wilkins, Baltimore, p 981
Persson T, Popescu BO, Cedazo-Minguez A (2014) Oxidative stress in Alzheimer’s disease: Why did antioxidant therapy fail? Oxi Med Cell Longev 2014:427318. https://doi.org/10.1155/2014/427318
Pfeiffer RF (2016) Non-motor symptoms in Parkinson’s disease. Parkinsonism Rel Dis 22(Suppl 1):S119–S122. https://doi.org/10.1016/j.parkreldis.2015.09.004
Picca A, Calvani R, Coelho-Junior H et al (2020) Mitochondrial dysfunction, oxidative stress, and neuroinflammation: intertwined roads to neurodegeneration. Antioxidants (basel) 9(8):647. https://doi.org/10.3390/antiox9080647
Pierantozzi M, Pietroiusti A, Brusa L et al (2006) Helicobacter pylori eradication and L-dopa absorption in patients with PD and motor fluctuations. Neurology 66(12):1824–1829. https://doi.org/10.1212/01.wnl.0000221672.01272.ba
Pinto Bezerra Soter AC, Bezerra TF, Pezato R et al (2017) Prospective open-label evaluation of long-term low-dose doxycycline for difficult-to-treat chronic rhinosinusitis with nasal polyps. Rhinology 55(2):175–180
Piras S, Furfaro AL, Domenicotti C et al (2016) RAGE Expression and ROS generation in neurons: differentiation versus damage. Oxid Med Cell Longev 2016:9348651. https://doi.org/10.1155/2016/9348651
Prince M (2015) World Alzheimer report. Available at: https://www.alz.co.uk/research/WorldAlzheimerReport2015.pdf
Pringsheim T, Jette N, Frolkis A et al (2014) The prevalence of Parkinson’s disease: a systematic review and meta-analysis. Mov Disord 29(13):1583–1590. https://doi.org/10.1002/mds.25945
Prusiner SB, Scott MR, De Armond SJ et al (1998) Prion protein biology. Cell 93:337–348. https://doi.org/10.1016/S0092-8674(00)81163-0
Quigley EMM (2017) Microbiota-brain-gut axis and neurodegenerative diseases. Curr Neurol Neurosci Rep 17(12):94. https://doi.org/10.1007/s11910-017-0802-6
Quintero EM, Willis L, Singleton R et al (2006) Behavioral and morphological effects of minocycline in the 6-hydroxydopamine rat model of Parkinson’s disease. Brain Res 1093(1):198–207. https://doi.org/10.1016/j.brainres.2006.03.104
Rao VS, Nagaraj RK, Hebbagodi S et al (2011) Association of inflammatory and oxidative stress markers with metabolic syndrome in Asian Indians in India. Cardiol Res Pract 2011:295976. https://doi.org/10.4061/2011/295976
Roberts MC (2003) Tetracycline therapy: update. Clin Infec Dis 36(4):462–467. https://doi.org/10.1086/367622
Ross JI, Eady EA, Cove JH et al (1998) 16S rRNA mutation associated with tetracycline resistance in a gram- positive bacterium. Antimicrob Agents Chemother 42(7):1702–1705. https://doi.org/10.1128/aac.42.7.1702
Sampson TR, Debelius JW, Thron T et al (2016) Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell 167(6):1469-1480.e12. https://doi.org/10.1016/j.cell.2016.11.018
Santa-Cecília FV, Socias B, Ouidja MO et al (2016) Doxycycline suppresses microglial activation by inhibiting the p38 MAPK and NF-kB signaling pathways. Neurotox Res 29(4):447–459. https://doi.org/10.1007/s12640-015-9592-2
Santa-Cecília FV, Leite CA, Del-Bel E et al (2019) The neuroprotective effect of doxycycline on neurodegenerative diseases. Neurotox Res 35(4):981–986. https://doi.org/10.1007/s12640-019-00015-z
Sapadin AN, Fleischmajer R (2006) Tetracyclines: nonantibiotic properties and their clinical implications. J Am Acad Dermatol 54(2):258–265. https://doi.org/10.1016/j.jaad.2005.10.004
Schapira AHV, Cooper JM, Dexter D et al (1990) Mitochondrial complex I deficiency in Parkinson’s disease. J Neurochem 54(3):823–827. https://doi.org/10.1111/j.1471-4159.1990.tb02325.x
Schlienger RG, Bircher AJ, Meier CR (2003) Minocycline-induced lupus. A systematic review. Dermatology 200(3):223–231
Schoonen WM, Thomas SL, Somers EC et al (2010) Do selected drugs increase the risk of lupus? A matched case-control study. Br J Clin Pharmacol 70(4):588–596. https://doi.org/10.1111/j.1365-2125.2010.03733.x
Seabrook TJ, Jiang L, Maier M et al (2006) Minocycline affects microglia activation, Abeta deposition, and behavior in APP-tg mice. Glia 53(7):776–782
Singh A, Kukreti R, Saso L et al (2019) Oxidative stress: A key modulator in neurodegenerative diseases. Molecules 24(8):1583. https://doi.org/10.3390/molecules24081583
Singhal G, Jaehne EJ, Corrigan F et al (2014) Inflammasomes in neuroinflammation and changes in brain function: a focused review. Front Neurosci 8:315. https://doi.org/10.3389/fnins.2014.00315
Sinha M, Bhowmick P, Banerjee A et al (2013) Antioxidant role of amyloid β protein in cell-free and biological systems: Implication for the pathogenesis of Alzheimer disease. Free Radic Biol Med 56:184–192. https://doi.org/10.1016/j.freeradbiomed.2012.09.036
Skoumalová A, Hort J (2012) Blood markers of oxidative stress in Alzheimer’s disease. J Cell Mol Med 16(10):291–2300. https://doi.org/10.1111/j.1582-4934.2012.01585.x
Smith DL, Woodman B, Mahal A et al (2003) Minocycline and doxycycline are not beneficial in a model of Huntington’s disease. Ann Neurol 54(2):186–196. https://doi.org/10.1002/ana.10614
Socias SB, González-Lizárraga F, Avila CL (2018) Exploiting the therapeutic potential of ready-to-use drugs: repurposing antibiotics against amyloid aggregation in neurodegenerative diseases. Prog Neurobiol 162:17–36. https://doi.org/10.1016/j.pneurobio.2017.12.002
Speer BS, Shoemaker NB, Salyers AA (1992) Bacterial resistance to tetracycline: mechanisms, transfer, and clinical significance. Clin Microbiol Rev 5(4):387–399. https://doi.org/10.1128/CMR.5.4.387
Steen E, Terry BM, Rivera EJ et al (2005) Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer’s disease—is this type 3 diabetes? J Alzheimers Dis 7(1):63–80. https://doi.org/10.3233/JAD-2005-7107
Stoilova T, Colombo L, Forloni G et al (2013) A new face for old antibiotics: tetracyclines in treatment of amyloidoses. J Med Chem 56(15):5987–6006. https://doi.org/10.1021/jm400161p
Stretton C, Hoffmann TM, Munson MJ et al (2015) GSK3-mediated raptor phosphorylation supports amino-acid-dependent mTORC1-directed signalling. Biochem J 470(2):207–221. https://doi.org/10.1042/BJ20150404
Sturkenboom MC, Meier CR, Jick H et al (1999) Minocycline and lupus like syndrome in acne patients. Arch Int Med 159(5):493–497. https://doi.org/10.1001/archinte.159.5.493
Sun MF, Shen YQ (2018) Dysbiosis of gut microbiota and microbial metabolites in Parkinson’s disease. Ageing Res Rev 45:53–61. https://doi.org/10.1016/j.arr.2018.04.004
Sun C, Wang Y, Mo M et al (2019) Minocycline protects against rotenone-induced neurotoxicity correlating with upregulation of Nurr1 in a Parkinson’s disease rat model. Biomed Res Int 2019:6843265. https://doi.org/10.1155/2019/6843265
Swerdlow RH, Burns JM, Khan SM (2010) The Alzheimer’s disease mitochondrial cascade hypothesis. J Alzheimers Dis 20(SUPPL 2):265–279. https://doi.org/10.3233/JAD-2010-100339
Szeto MD, Boissel SJS, Baker D et al (2011) Mining endonuclease cleavage determinants in genomic sequence data. J Biol Chem 286(37):32617–32627. https://doi.org/10.1074/jbc.M111.259572
Tagliavini F, McArthur RA, Canciani B et al (1997) Effectiveness of anthracycline against experimental prion disease in Syrian hamsters. Science 276(5315):1119–1122. https://doi.org/10.1126/science.276.5315.1119
Tagliavini F, Forloni G, Colombo L et al (2000) Tetracycline affects abnormal properties of synthetic PrP peptides and PrP(Sc) in vitro. J Mol Biol 300(5):1309–1322. https://doi.org/10.1006/jmbi.2000.3840
Takahashi M, Yamada T, Tooyama I et al (1996) Insulin receptor mRNA in the substantia nigra in Parkinson’s disease. Neurosci Lett 204(3):201–204. https://doi.org/10.1016/0304-3940(96)12357-0
Tan AH, Mahadeva S, Marras C et al (2015) Helicobacter pylori infection is associated with worse severity of Parkinson’s disease. Parkinsonism Relat Dis 21(3):221–225. https://doi.org/10.1016/j.parkreldis.2014.12.009
Tarafdar A, Pula G (2018) The role of NADPH oxidases and oxidative stress in neurodegenerative disorders. Int J Mol Sci 19(12):3824. https://doi.org/10.3390/ijms19123824
Taylor KSM, Cook JA, Counsell CE (2007) Heterogeneity in male to female risk for Parkinson’s disease. J Neurol Neurosurg Psychiatry 78(8):905–906. https://doi.org/10.1136/jnnp.2006.104695
Teo E, Ravi S, Barardo D et al (2019) Metabolic stress is a primary pathogenic event in transgenic Caenorhabditis elegans expressing pan-neuronal human amyloid beta. Elife 8:e50069. https://doi.org/10.7554/eLife.50069
Thaker M, Spanogiannopoulos P, Wright GD (2010) The tetracycline resistome. Cell Mol Life Sci 67(3):419–431. https://doi.org/10.1007/s00018-009-0172-6
Thomas JG, Metheny RJ, Karakiozis JM et al (1998) Long-term sub-antimicrobial doxycycline (Periostat) as adjunctive management in adult periodontitis: effects on subgingival bacterial population dynamics. Adv Dent Res 12(2):32–39. https://doi.org/10.1177/08959374980120011601
Tieu K, Ischiropoulos H, Przedborski S (2003) Nitric oxide and reactive oxygen species in Parkinson’s disease. IUBMB Life 55(6):329–335. https://doi.org/10.1080/1521654032000114320
Tikka T, Fiebich BL, Goldsteins G et al (2001) Minocycline, a tetracycline derivative, is neuroprotective against excitotoxicity by inhibiting activation and proliferation of microglia. J Neurosci 21(8):2580–2588. https://doi.org/10.1523/jneurosci.21-08-02580.2001
Tong M, Dong M, De La Monte SM (2009) Brain Insulin-like growth factor and neurotrophin resistance in parkinson’s disease and dementia with Lewy bodies: potential role of manganese neurotoxicity. J Alzheimers Dis 16(3):585–599. https://doi.org/10.3233/JAD-2009-0995.Brain
Tönnies E, Trushina E (2017) Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. J Alzheimers Dis 57(4):1105–1121. https://doi.org/10.3233/JAD-161088
Tran TTT, Corsini S, Kellingray L et al (2019) APOE genotype influences the gut microbiome structure and function in humans and mice: relevance for Alzheimer’s disease pathophysiology. FASEB J 33(7):8221–8231. https://doi.org/10.1096/fj.201900071R
Trist BG, Hare DJ, Double KL (2019) Oxidative stress in the aging substantia nigra and the etiology of Parkinson’s disease. Aging Cell 18(6):e13031. https://doi.org/10.1111/acel.13031
Tsaluchidu S, Cocchi M, Tonello L et al (2008) Fatty acids and oxidative stress in psychiatric disorders. BMC Psychiatry 8(SUPPL. 1):S5. https://doi.org/10.1186/1471-244X-8-S1-S5
van Marum RJ (2009) Update on the use of memantine in Alzheimer’s disease. Neuropsychiatr Dis Treat 5(1):237–247. https://doi.org/10.2147/ndt.s4048
Varges D, Manthey H, Heinemann U et al (2017) Doxycycline in early CJD: a double-blinded randomised phase II and observational study. J Neurol Neurosurg Psychiatry 88(2):119–125. https://doi.org/10.1136/jnnp-2016-313541
Vendrik KEW, Ooijevaar RE, de Jong PRC et al (2020) Fecal microbiota transplantation in neurological disorders. Front Cell Infect Microbiol 10:98. https://doi.org/10.3389/fcimb.2020.00098
Vich Vila A, Collij V, Sanna S et al (2020) Impact of commonly used drugs on the composition and metabolic function of the gut microbiota. Nat Commun 11(1):362. https://doi.org/10.1038/s41467-019-14177-z
Von Wittenau MS, Twomey TM, Swindell AC (1971) The disposition of doxycycline by the rat. Chemotherapy 17(1):26–39
Walker C, Preshaw PM, Novak J et al (2005) Long-term treatment with sub-antimicrobial dose doxycycline has no antibacterial effect on intestinal flora. J Clin Periodontol 32(11):1163–1169. https://doi.org/10.1111/j.1600-051X.2005.00840.x
Wang Q, Miyakoda M, Yang W et al (2004) Stress-induced apoptosis associated with null mutation of ADAR1 RNA editing deaminase gene. J Biol Chem 279(6):4952–4961. https://doi.org/10.1074/jbc.M310162200
Wang N, Tian X, Chen Y et al (2017) Low dose doxycycline decreases systemic inflammation and improves glycemic control, lipid profiles, and islet morphology and function in db/db mice. Sci Rep 7(1):14707. https://doi.org/10.1038/s41598-017-14408-7
Wang J, Qang Q, Yu R et al (2020) Minocycline inhibition of microglial rescues nigrostriatal dopaminergic neurodegeneration caused by mutant alpha-synuclein overexpression. Aging (albany NY) 12(14):14232–14243. https://doi.org/10.18632/aging.103440
Ward JE, Ren R, Toraldo G et al (2011) Doxycycline reduces fibril formation in a transgenic mouse model of AL amyloidosis. Blood 118:6610–6617. https://doi.org/10.1182/blood-2011-04-351643
Williams HC, Wojnarowska F, Kirtschig G et al (2017) Doxycycline versus prednisolone as an initial treatment strategy for bullous pemphigoid: a pragmatic, non-inferiority, randomised controlled trial. Lancet 389(10079):1630–1638. https://doi.org/10.1016/S0140-6736(17)30560-3
Wojsiat J, Laskowska-Kaszub K, Mietelska-Porowska A et al (2017) Search for Alzheimer’s disease biomarkers in blood cells: hypotheses-driven approach. Biomark Med 11(10):917–931. https://doi.org/10.2217/bmm-2017-0041
Wu DC, Jackso-Lewis V, Vila M et al (2002) Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson disease. J Neuroscience 22(5):1763–1771. https://doi.org/10.1523/jneurosci.22-05-01763.2002
Xu J, Zhao C, Huang X et al (2020) Tetracycline derivatives resist the assembly behavior of human islet amyloid polypeptide. Biochimie 174:95–106. https://doi.org/10.1016/j.biochi.2020.04.012
Yang L, Sugama S, Chirichigno JW et al (2003) Minocycline enhances MPTP toxicity to dopaminergic neurons. J Neurosci Res 74(2):278–285. https://doi.org/10.1002/jnr.10709
Yang L, Wang H, Liu L et al (2018) The role of insulin/IGF-1/PI3K/Akt/GSK3 β signaling in Parkinson’s disease dementia. Front Neurosci 12:73. https://doi.org/10.3389/fnins.2018.00073
Yiannopoulou KG, Papageorgiou SG (2013) Current and future treatments for Alzheimer’s disease. Ther Adv Neurol Disord 6(1):19–33. https://doi.org/10.1177/1756285612461679
Yılmaz C, Özcengiz G (2017) Antibiotics: pharmacokinetics, toxicity, resistance and multidrug efflux pumps. Biochem Pharmacol 133:43–62. https://doi.org/10.1016/j.bcp.2016.10.005
Yim CW, Flynn NM, Fitzgerald FT (1985) Penetration of oral doxycycline into the cerebrospinal fluid of patients with latent of neurosyphilis. Antimicrob Agents Chemother 28(2):347–348. https://doi.org/10.1128/AAC.28.2.347
Yimer EM, Hishe HZ, Tuem KB (2019) Repurposing of the β-lactam antibiotic, ceftriaxone for neurological disorders: a review. Front Neurosci 13:236. https://doi.org/10.3389/fnins.2019.00236
Yin F, Sancheti H, Patil I et al (2016) Energy metabolism and inflammation in brain aging and Alzheimer’s disease. Free Radic Biol Med 100:108–122. https://doi.org/10.1016/j.freeradbiomed.2016.04.200
Yong VW, Power C, Edwards DR (2001) Metalloproteinases in biology and pathology of the nervous system. Nat Rev Neurosci 2(7):502–511. https://doi.org/10.1038/35081571
Yoon KW, Byun S, Kwon E et al (2015) Control of signaling-mediated clearance of apoptotic cells by the tumor suppressor p53. Science 349(6247):1261669. https://doi.org/10.1126/science.1261669
Yrjänheikki J, Tikka T, Keinänen R et al (1999) A tetracycline derivative, minocycline, reduces inflammation and protects against focal cerebral ischemia with a wide therapeutic window. Proc Natl Acad Sci USA 96(23):13496–13500. https://doi.org/10.1073/pnas.96.23.13496
Yulug B, Hanoglu L, Ozansoy M et al (2018) Therapeutic role of rifampicin in Alzheimer’s disease. Psychiatry Clin Neurosci 72(3):152–159. https://doi.org/10.1111/pcn.12637
Zhanel GG, Homenuik K, Nichol K et al (2004) The glycylcyclines: a comparative review with the tetracyclines. Drugs 64(1):63–88. https://doi.org/10.2165/00003495-200464010-00005
Zhang H, García Rodríguez LA, Hernández-Díaz S (2008) Antibiotic use and the risk of lung cancer. Cancer Epidemiol Biomarkers Prev 17(6):1308–1315. https://doi.org/10.1158/1055-9965.EPI-07-2817
Zhang J, Zhang J, Wang R (2018) Gut microbiota modulates drug pharmacokinetics. Drug Metab Rev 50(3):357–368. https://doi.org/10.1080/03602532.2018.1497647
Zhuang ZQ, Shen LL, Li WW et al (2018) Gut microbiota is altered in patients with Alzheimer’s disease. J Alzheimers Dis 63(4):1337–1346. https://doi.org/10.3233/JAD-180176
Zilka N, Kazmerova Z, Jadhav S et al (2012) Who fans the flames of Alzheimer’s disease brains? Misfolded tau on the crossroad of neurodegenerative and inflammatory pathways. J Neuroinflammation 9:47. https://doi.org/10.1186/1742-2094-9-47
Zink MC, Uhrlaub J, De Witt J et al (2011) Neuroprotective and Anti-Human Immunodeficiency Virus Activity of Minocycline. JAMA 293(16):2003–2011. https://doi.org/10.1001/jama.293.16.2003
Acknowledgements
This work was supported by the Scientific Centre of Excellence for Basic, Clinical, and Translational Neuroscience (project “Experimental and clinical research of hypoxic-ischemic damage in perinatal and adult brain”; GA KK01.1.1.01.0007 funded by the European Union through the European Regional Development Fund).
Author information
Authors and Affiliations
Contributions
IM and MM performed the literature search and participated in writing; VET participated in writing and critical reading; MS-P had the idea for the article, drafted and critically revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Markulin, I., Matasin, M., Turk, V.E. et al. Challenges of repurposing tetracyclines for the treatment of Alzheimer’s and Parkinson’s disease. J Neural Transm 129, 773–804 (2022). https://doi.org/10.1007/s00702-021-02457-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-021-02457-2