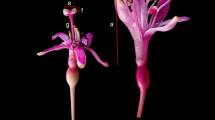
Abstract
Dioecy, the separation of sex at the individual level, evolved in angiosperms from hermaphroditic ancestors. One of the postulated evolutionary pathways is by way of the gynodioecious stage, i.e., the coexistence of female and hermaphroditic individuals. In a morphologically gynodioecious species, Daphne jezoensis., seed fertility in hermaphrodites is limited compared to that of females, indicating that its sexual system is close to dioecy. The aim of this study was to clarify whether the gynodioecy in this species is a stable sexual system or a transitional state leading to dioecy. We evaluated variations in the degree of pollen limitation, sexual and mating systems, floral morphology, sex ratio, and the genetic structure of 14 populations across the species’ distribution range in Japan. Sexual systems were similar among populations throughout the distribution range although the southernmost populations had unique genetic structures to some extent. Hermaphrodites were commonly self-compatible, but their fruiting capacity was low in every population. This suggests that gynodioecy composed of females and hermaphrodites having low seed fertility is a consistent sexual system in D. jezoensis. Low seed fertility in hermaphrodites throughout the distribution range suggests that this sexual system is not necessarily a transitional state from gynodioecy to dioecy if occasional seed production in hermaphrodites has any advantage. One possible ecological advantage for the maintenance of hermaphrodites is that seed production by autonomous self-pollination may be beneficial in the colonization process after long-distance seed dispersal.





Similar content being viewed by others
Availability of data and material
The datasets used in the current study are available from the corresponding author on reasonable request.
References
Adamack AT, Gruber B (2014) PopGenReport: Simplifying basic population genetic analyses in R. Meth Ecol Evol 5:384–387. https://doi.org/10.1111/2041-210X.12158
Alonso C, Herrera CM (2011) Back-and-forth hermaphroditism: phylogenetic context of reproductive system evolution in subdioecious Daphne laureola. Evolution 65:1680–1692. https://doi.org/10.1111/j.1558-5646.2011.01246.x
Alonso C, Mutikainen P, Herrera CM (2007) Ecological context of breeding system variation: sex, size and pollination in a (predominantly) gynodioecious shrub. Ann Bot (Oxford) 100:1547–1556. https://doi.org/10.1093/aob/mcm254
Arroyo MTK, Raven PH (1975) The evolution of subdioecy in morphologically gynodioecious species of Fuchsia sect. encliandra (Onagraceae). Evolution 29:500–511. https://doi.org/10.2307/2407262
Austerlitz F, Garnier-Géré PH (2003) Modelling the impact of colonisation on genetic diversity and differentiation of forest trees: interaction of life cycle, pollen flow and seed long-distance dispersal. Heredity 90:282–290. https://doi.org/10.1038/sj.hdy.6800243
Bai YP, Zhang YW, Gituru RW, Zhao JM, Li JD (2011) Sexual differences in reproductive characters and pollinator attractiveness in gynodioecious Glechoma longituba (Lamiaceae). Pl Spec Biol 26:33–42. https://doi.org/10.1111/j.1442-1984.2010.00298.x
Baker H (1955) Self-compatible and establishment after “long dinstance” dispersal. Evolution 9:347–349
Baker H, Cox P (1984) Further thoughts on dioecious and islands. Ann Missouri Bot Gard 71:244–253. https://doi.org/10.2307/2399068
Bell G (1985) On the function of flowers. Proc Roy Soc B Biol Sci 224:223–265. https://doi.org/10.1098/rspb.1985.0031
Blacket MJ, Robin C, Good RT, Lee SF, Miller AD (2012) Universal primers for fluorescent labelling of PCR fragments-an efficient and cost-effective approach to genotyping by fluorescence. Molec Ecol Resources 12:456–463. https://doi.org/10.1111/j.1755-0998.2011.03104.x
Brickell C, Marhew B (1976) Daphne. The genus in the wild and cultivation, 1 st edn. Alpine Garden Society, Birmingham
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Mächler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400
Busch JW, Delph LF (2012) The relative importance of reproductive assurance and automatic selection as hypotheses for the evolution of self-fertilization. Ann Bot (Oxford) 109:553–562. https://doi.org/10.1093/aob/mcr219
Caruso CM, Case AL (2007) Sex ratio variation in gynodioecious Lobelia siphilitica: effects of population size and geographic location. J Evol Biol 20:1396–1405. https://doi.org/10.1111/j.1420-9101.2007.01361.x
Case AL, Barrett S (2004) Environmental stress and the evolution of dioecy: Wurmbea dioica (Colchicaceae) in Western Australia. Evol Ecol 18:145–164. https://doi.org/10.1023/B:EVEC.0000021152.34483.77
Cavalli-Sforza LL, Edwards AW (1967) Phylogenetic analysis. Models and estimation procedures. Amer J Human Genet 19:233–257. https://doi.org/10.2307/2406616
Charlesworth B, Charlesworth D (1978) A model for the evolution of dioecy and gynodioecy. Amer Naturalist 112:975–997
Charlesworth D (1989) Allocation to male and female function in hermaphrodites, in sexually polymorphic populations. J Theor Biol 139:327–342. https://doi.org/10.1016/S0022-5193(89)80212-7
Charlesworth D (2003) Effects of inbreeding on the genetic diversity of populations. Philos Trans Roy Soc B Biol Sci 358:1051–1070. https://doi.org/10.1098/rstb.2003.1296
Charlesworth D (2018) Does sexual dimorphism in plants promote sex chromosome evolution? Environm Exp Bot 146:5–12. https://doi.org/10.1016/j.envexpbot.2017.11.005
Charlesworth D, Pannell JR (2001) Mating systems and population genetic structure in the light of coalescent theory. In: Silvertown J, Antonovics J (eds) Integrating Ecology and Evolution in a Spatial Context. Blackwell, Oxford, pp 73–96
Cossard GG, Pannell JR (2019) A functional decomposition of sex inconstancy in the dioecious, colonizing plant Mercurialis annua. Amer J Bot 106:722–732. https://doi.org/10.1002/ajb2.1277
Covarrubias-Pazaran G, Diaz-Garcia L, Schlautman B, Salazar W, Zalapa J (2016) Fragman: An R package for fragment analysis. BMC Genet 17:1–8. https://doi.org/10.1186/s12863-016-0365-6
Darwin CR (1877) The different forms of flowers on plants of the same species. John Murray, London
Del Castillo RF, Argueta ST (2009) Reproductive implications of combined and separate sexes in a trioecious population of Opuntia robusta (Cactaceae). Amer J Bot 96:1148–1158. https://doi.org/10.3732/ajb.0800301
Delph LF, Wolf DE (2005) Evolutionary consequences of gender plasticity in genetically dimorphic breeding systems. New Phytol 166:119–128
Dufay M, Champelovier P, Käfer J, Henry JP, Mousset S, Marais GAB (2014) An angiosperm-wide analysis of the gynodioecy-dioecy pathway. Ann Bot (Oxford) 114:539–548. https://doi.org/10.1093/aob/mcu134
Ehlers BK, Bataillon T (2007) “Inconstant males” and the maintenance of labile sex expression in subdioecious plants. New Phytol 174:194–211. https://doi.org/10.1111/j.1469-8137.2007.01975.x
Felsenstein J (2013) PHYLIP (Phylogeny Inference Package) version 3.695. Abailable at: https://evolution.genetics.washington.edu/phylip.html. Accessed 15 May 2020
Fleming TH, Maurice S, Hamrick JL (1998) Geographic variation in the breeding system and the evolutionary stability of trioecy in Pachycereus pringlei (Cactaceae). Evol Ecol 12:279–289. https://doi.org/10.1023/A:1006548132606
Garnock-Jones PJ, Brockie RE, FitzJohn RG (2007) Gynodioecy, sexual dimorphism and erratic fruiting in Corynocarpus laevigatus (Corynocarpaceae). Austral J Bot 55:803–808. https://doi.org/10.1071/BT07054
Gutiérrez-Flores C, García-De León FJ, León-de la luz JL, Cota-Sánchez JH (2016) Microsatellite genetic diversity and mating systems in the columnar cactus Pachycereus pringlei (Cactaceae). Perspect Pl Ecol Evol Syst 22:1–10. https://doi.org/10.1016/j.ppees.2016.06.003
Hamrick JL, Godt MJW, Sherman-Broyles SL (1992) Factors influencing levels of genetic diversity in woody plant species. New forests 6:95–124. https://doi.org/10.1007/BF00120641
Hedrick PW (2005) A standardized genetic differentiation measure. Evolution 59:1633–1638. https://doi.org/10.1111/j.0014-3820.2005.tb01814.x
Hiraoka K, Tomaru N (2009) Genetic divergence in nuclear genomes between populations of Fagus crenata along the Japan Sea and Pacific sides of Japan. J Pl Res 122:269–282. https://doi.org/10.1007/s10265-009-0217-9
Iwaizumi MG, Tsuda Y, Ohtani M, Tsumura Y, Takahashi M (2013) Recent distribution changes affect geographic clines in genetic diversity and structure of Pinus densiflora natural populations in Japan. Forest Ecol Managem 304:407–416. https://doi.org/10.1016/j.foreco.2013.05.026
Iwatsuki K, Boufford DE, Oba H (1999) Flora of Japan, vol. IIc. Kodansha, Tokyo
Keenan K, Mcginnity P, Cross TF, Crozier WW, Prodöhl PA (2013) DiveRsity: An R package for the estimation and exploration of population genetics parameters and their associated errors. Meth Ecol Evol 4:782–788. https://doi.org/10.1111/2041-210X.12067
Kikuzawa K (1989) Floral biology and evolution of gynodioecism in Daphne kamtchatica var. jezoensis. Oikos 56:196–202
Kosman E, Leonard KJ (2005) Similarity coefficients for molecular markers in studies of genetic relationships between individuals for haploid, diploid, and polyploid species. Molec Ecol 14:415–424. https://doi.org/10.1111/j.1365-294X.2005.02416.x
Kudo G, Kameyama Y, Shirosaka YH (2015) Population structure of Daphne jezoensis Maxim. Bull Bihor Mu 22:1–6 (in Japanese)
Le Corre V, Kremer A (1998) Cumulative effects of founding events during colonisation on genetic diversity and differentiation in an island and stepping-stone model. J Evol Biol 11:495–512. https://doi.org/10.1007/s000360050102
Lewis D (1941) Male sterility in natural populations of hermaphrodite plants: the equilibrium between females and hermaphrodites to be expected with different types of inheritance. New Phytol 40:56–63. https://doi.org/10.1111/j.1469-8137.1941.tb07028.x
Lloyd DG (1974) Theoretical sex ratios of dioecious and gynodioecious angiosperms. Heredity 32:11–34. https://doi.org/10.1038/hdy.1974.2
Lloyd DG (1975) The maintenance of gynodioecy and androdioecy in angiosperms. Genetica 45:325–339. https://doi.org/10.1007/BF01508307
Lloyd DG (1976) The transmission of genes via pollen and ovules in gynodioecious angiosperms. Theor Populat Biol 9:299–316. https://doi.org/10.1016/0040-5809(76)90050-2
Meglécz E, Costedoat C, Dubut V, Gilles A, Malausa T, Pech N, Martin JF (2010) QDD: A user-friendly program to select microsatellite markers and design primers from large sequencing projects. Bioinformatics 26:403–404. https://doi.org/10.1093/bioinformatics/btp670
Meglécz E, Pech N, Gilles A, Dubut V, Hingamp P, Trilles A, Grenier R, Martin JF (2014) QDD version 3.1: A user-friendly computer program for microsatellite selection and primer design revisited: Experimental validation of variables determining genotyping success rate. Molec Ecol Resources 14:1302–1313. https://doi.org/10.1111/1755-0998.12271
Nilsson E, Ågren J (2006) Population size, female fecundity, and sex ratio variation in gynodioecious Plantago maritima. J Evol Biol 19:825–833. https://doi.org/10.1111/j.1420-9101.2005.01045.x
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2019) Vegan: community ecology package.
Pannell J (1997) The maintenance of gynodioecy and androdioecy in angiosperms. Evolution 51:10–20. https://doi.org/10.1007/BF01508307
Pannell JR (2015) Evolution of the mating system in colonizing plants. Molec Ecol 24:2018–2037. https://doi.org/10.1111/mec.13087
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. Available at: https://www.R-project.org/. Accessed 20 Apr 2021
Ritland K (2002) Extensions of models for the estimation of mating systems using n independent loci. Heredity 88:221–228. https://doi.org/10.1038/sj/hdy/6800029
Roccotiello E, Casazza G, Galli L, Cornara L, Minuto MA (2009) The flower biology of Daphne gnidium L. (Thymelaeaceae). Pl Syst Evol 279:41–49. https://doi.org/10.1007/s00606-009-0144-1
Ross MD (1978) The evolution of gynodioecy and subdioecy. Evolution 32:174–188. https://doi.org/10.2307/2407418
Rodrıguez-Perez J, Traveset A (2011) Influence of reproductive traits on pollination success in two Daphne species (Thymelaeaceae). J Pl Res 124:277–287. https://doi.org/10.1007/s10265-010-0373-y
Rousset F (2008) GENEPOP’007: a complete re-implementation of the GENEPOP software for Windows and Linux. Molec Ecol Resources 8:103–106. https://doi.org/10.1111/j.1471-8286.2007.01931.x
Sakaguchi S, Takeuchi Y, Yamasaki M, Sakurai S, Isagi Y (2011) Lineage admixture during postglacial range expansion is responsible for the increased gene diversity of Kalopanax septemlobus in a recently colonised territory. Heredity 107:338–348. https://doi.org/10.1038/hdy.2011.20
Sakata Y, Nakahama N (2018) Flexible pollination system in an unpalatable shrub Daphne miyabeana (Thymelaeaceae). Pl Spec Biol 33:239–247. https://doi.org/10.1111/1442-1984.12212
Schultz ST (2003) Sexual dimorphism in gynodioecious Sidalcea hirtipes (Malvaceae). II. Floral Traits Int J Plant Sci 164:175–180. https://doi.org/10.1086/344551
Shibata A (2020) Maintenance mechanism of gynodioecy in a summer-deciduous shrub, Daphne jezoensis. PhD Thesis, Hokkaido University, Hokkaido
Shibata A, Kameyama Y, Kudo G (2018) Restricted female function of hermaphrodites in a gynodioecious shrub, Daphne jezoensis (Thymelaeaceae). J Pl Res 131:245–254. https://doi.org/10.1007/s10265-017-0978-5
Shibata A, Kudo G (2017) Size-dependent sex allocation and reproductive investment in a gynodioecious shrub. AoB Pl 9:1–11. https://doi.org/10.1093/aobpla/plw0899
Shykoff JA, Kolokotronis S-O, Collin CL, López-Villavicencio M (2003) Effects of male sterility on reproductive traits in gynodioecious plants: a meta-analysis. Oecologia 135:1–9. https://doi.org/10.1007/s00442-002-1133-z
Sinclair JP, Kameyama Y, Shibata A, Kudo G (2016) Male-biased hermaphrodites in a gynodioecious shrub, Daphne jezoensis. Plant Biol 18:859–867. https://doi.org/10.1111/plb.12463
Spigler RB, Ashman T-L (2011) Sex ratio and subdioecy in Fragaria virginiana: the roles of plasticity and gene flow examined. New Phytol 190:1058–1068. https://doi.org/10.1111/j.1469-8137.2011.03657.x
Spigler RB, Ashman T-L (2012) Gynodioecy to dioecy: Are we there yet? Ann Bot (Oxford) 109:531–543. https://doi.org/10.1093/aob/mcr170
Stewart CN, Via LE (1993) A rapid CTAB DNA isolation technique useful for RAPD fingerprinting and other PCR applications. Bio Tech 14:748–750
Suzuki T (2016) Fruit- and seed-eating agents for the poisonous shrub, Daphne pseudomezereum (Thymelaeaceae), in southern Kanto district, Japan. Yamashina Inst Ornithol 48:1–11 (in Japanese)
Taylor DR, Trimble S, McCauley DE (1999) Ecological genetics of gynodioecy in Silene vulgaris: relative fitness of females and hermaphrodites during the colonization process. Evolution 53:745–751. https://doi.org/10.2307/2640714
Vaughton G, Ramsey M (1998) Floral display, pollinator visitation and reproductive success in the dioecious perennial herb Wurmbea dioica (Liliaceae). Oecologia 115:93–101. https://doi.org/10.1007/s004420050495
Venables WN, Ripley BD (2002) Modern Applied Statistics with S, 4th edn. Springer, New York
Venkatasamy S, Khittoo G, Keeley S (2007) Leaky dioecy in Diospyros (Ebenaceae) endemic to the Island of Mauritius. Pl Ecol 189:139–146. https://doi.org/10.1007/s11258-006-9171-y
Wang Q, Deng HP, Ding B, Zhou GL (2014) Flower structure and sex expression of leaky dioecy in Eurya obtusifolia. Nordic J Bot 32:314–319. https://doi.org/10.1111/j.1756-1051.2013.00226.x
Wang X, Wen M, Wu M, Xu Y, Zhang K, Zhang D (2020) Gynodioecy or leaky dioecy? The unusual sexual system of a coral dune-habitant Tournefortia argentea (Boraginaceae). Pl Syst Evol 306:1–11. https://doi.org/10.1007/s00606-020-01698-0
Wolfe LM, Shmida A (1997) The ecology of sex expression in a gynodioecious isreali desert shrub (Ochradenus baccatus). Ecology 78:101–110. https://doi.org/10.1890/0012-9658(1997)078[0101:TEOSEI]2.0.CO;2
Wright S (1943) Isolation by distance. Genetics 28:114–138. https://doi.org/10.1109/IGARSS.2006.90
Acknowledgements
We thank all members of the Plant Ecology Lab of the Graduate School of Environmental Science, Hokkaido University, for their kind assistance in the fieldwork and helpful discussions on this study. We appreciate the following persons for their help with the permission procedure and their practical supports for the field survey: K. Narita, N. Nakamura, K. Horie, I. Honda, M. Masuda, and M. Tada. We would like to thank Editage (www.editage.com) for English editing.
Funding
This study was supported by JSPS KAKENHI (19J10583, 18H02504).
Author information
Authors and Affiliations
Contributions
A.S. and G.K. designed the study, and collected and analyzed the data. Y.K. developed microsatellite markers. A.S. wrote the first draft of the manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling editor: Martin A. Lysak.
Contribution to “Living at its dry limits - Tillandsiales in the Atacama Desert”.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Information on Electronic Supplementary Material
Information on Electronic Supplementary Material
Online Resource 1. Mean fruit number in females (F) and hermaphrodites (H) and relative fruit number in F to H under open pollination and outcross pollination conditions.
Online Resource 2. The number of flowers used for floral measurements.
Online Resource 3. Sequences for newly developed primer pairs.
Online Resource 4. Relationships between the degree of pollen limitation and fruit-set rate of hermaphrodites, selfing rate of hermaphrodites, allelic richness, and FIS.
Online Resource 5. Unrooted neighbor-joining tree using Cavalli-Sforza and Edwards’ chord distances among 14 Daphne jezoensis populations.
Rights and permissions
About this article
Cite this article
Shibata, A., Kameyama, Y. & Kudo, G. Low seed fertility of hermaphrodites is maintained in a gynodioecious species throughout the distribution range in Japan. Plant Syst Evol 307, 55 (2021). https://doi.org/10.1007/s00606-021-01780-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00606-021-01780-1