Abstract
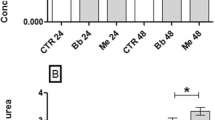
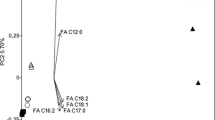
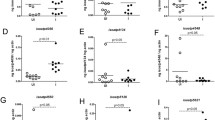
Despite the importance of fat body in metabolism of arthropods, studies in ticks are scarce. This study evaluated the lipid composition and activation of extracellular signal-regulated protein kinase (ERK) and AMP-activated protein kinase (AMPK) enzymes in Rhipicephalus microplus fat body after infection with different isolates of the fungus Metarhizium anisopliae sensu lato (Metschnikoff, 1879) Sorokin, 1883. The isolates CG 32, GC 112, GC 148, GC 347, and GC 629 were inoculated as viable or non-viable conidia in the ticks. The engorged females were dissected, and their fat bodies were collected 24 and 48 h after infection. The lipid composition was assessed by thin layer chromatography, and enzyme activation was detected by Western blotting with antibodies against p-AMPK and p-ERK. The study showed increased levels of triacylglycerol 24 and 48 h and fatty acid after 48 h after inoculation with different isolates of viable fungi in the tick’s hemocoel. Detection of the active form of ERK was demonstrated only after inoculation with non-viable conidia of all isolates tested. The active form of AMPK, only isolate CG 112 was able to activate with viable or non-viable conidia, whereas isolates CG 32 and CG 629 were able to activate with non-viable conidia. This study provides the first report about changes in important metabolic pathways in ticks infected with entomopathogenic fungi and suggests that the lipid content is modulated by non-usual pathways. However, further studies may be necessary for a better elucidation of this interaction.



Similar content being viewed by others
References
Alves SB (1998) Controle microbiano de insetos. FEALQ, Piracicaba
Angelo IC, Golo PS, Perinotto WMS, Camargo MG, Quinelato S, Sá FA, Pontes EG, Bittencourt VREP (2013) Neutral lipid composition changes in the fat bodies of engorged females Rhipicephalus microplus ticks in response to fungal infections. Parasitol Res 112(2):501–509. https://doi.org/10.1007/s00436-012-3159-4
Atella GC, Bittencourt-Cunha PR, Nunes RD, Shahabuddin M, Silva-Neto MAC (2009) The major insect lipoprotein is a lipid source to mosquito stages of malaria parasite. Acta Trop 109(2):159–162. https://doi.org/10.1016/j.actatropica.2008.10.004
Azeez OI, Meintjes R, Chamunorwa JP (2014) Fat body, fat pad and adipose tissues in invertebrates and vertebrates: the nexus. Lipids Health Dis 13(1):1–13. https://doi.org/10.1186/1476-511X-13-71
Benelli G, Pavela R, Canale A, Mehlhorn H (2016) Tick repellents and acaricides of botanical origin: a green roadmap to control tick-borne diseases? Parasitol Res 115(7):2545–2560. https://doi.org/10.1007/s00436-016-5095-1
Bi L, Chiang JY, Ding WX, Dunn W, Roberts B, Li T (2013) Saturated fatty acids activate ERK signaling to downregulate hepatic sortilin 1 in obese and diabetic mice. J Lipid Res 54(10):2754–2762. https://doi.org/10.1194/jlr.M039347
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Med Sci 37(8):911–917. https://doi.org/10.1139/o59-099
Charroux B, Royet J (2010) Drosophila immune response: from systemic antimicrobial peptide production in fat body cells to local defense in the intestinal tract. Fly 4(1):40–47. https://doi.org/10.4161/fly.4.1.10810
Fernandes ÉKK, Moraes ÁML, Pacheco RS, Rangel DEN, Miller MP, Bittencourt VREP, Roberts DW (2009) Genetic diversity among Brazilian isolates of Beauveria bassiana: comparisons with non-Brazilian isolates and other Beauveria species. J Appl Microbiol 107(3):760–774. https://doi.org/10.1111/j.1365-2672.2009.04258.x
Fernandes ÉKK, Angelo IC, Rangel DEN, Bahiense TC, Moraes ÁML, Roberts DW, Bittencourt VREP (2011) An intensive search for promising fungal biological control agents of ticks, particularly Rhipicephalus microplus. Vet Parasitol 182:307–318. https://doi.org/10.4161/fly.4.1.10810
Folly E, Silva NCE, Lopes AH, Silva-Neto MAC, Atella GC (2003) Trypanosoma rangeli uptakes the main lipoprotein from the hemolymph of its invertebrate host. Biochem Bioph Res Co 310(2):555–561. https://doi.org/10.1016/j.bbrc.2003.09.038
Gehart H, Kumpf S, Ittner A, Ricci R (2010) MAPK signalling in cellular metabolism: stress or wellness? EMBO Rep 11(11):834–840. https://doi.org/10.1038/embor.2010.160
George JE, Pound JM, Davey RB (2004) Chemical control of ticks on cattle and the resistance of these parasites to acaricides. Parasitology 129(7):S353–S366. https://doi.org/10.1017/S0031182003004682
Grisi L, Leite RC, Martins JRS, Barros ATM, Andreotti R, Cançado PD, Leon AAP, Pereira JB, Villela HS (2014) Reassessment of the potential economic impact of cattle parasites in Brazil. Rev Bras Parasitol Vet 23(2):150–156. https://doi.org/10.1590/S1984-29612014042
Hardie DG (2014) AMPK–sensing energy while talking to other signaling pathways. Cell Metab 20(6):939–952. https://doi.org/10.1016/j.cmet.2014.09.013
Jiang X, Huang L, Xing D (2015) Photoactivation of Dok1/ERK/PPARγ signaling axis inhibits excessive lipolysis in insulin-resistant adipocytes. Cell Signal 27(7):1265–1275. https://doi.org/10.1016/j.cellsig.2015.03.010
Jiao P, Feng B, Li Y, He Q, Xu H (2013) Hepatic ERK activity plays a role in energy metabolism. Mol Cell Endocrinol 375(1-2):157–166. https://doi.org/10.1016/j.mce.2013.05.021
Johns R, Sonenshine DE, Hynes WL (1998) Control of bacterial infections in the hard tick Dermacentor variabilis (Acari: Ixodidae): evidence for the existence of antimicrobial proteins in tick hemolymph. J Med Entomol 35(4):458–464. https://doi.org/10.1093/jmedent/35.4.458
Kawooya JK, Law JH (1988) Role of lipophorin in lipid transport to the insect egg. J Biol Chem 263(18):8748–8753
Khovidhunkit W, Kim MS, Memon RA, Shingenaga JK, Moser AH, Feingold KR, Grunfeld C (2004) Effects of infection and inflammation on lipid and lipoprotein metabolism: mechanisms and consequences to the host. J Lipid Res 45(7):1169–1186. https://doi.org/10.1194/jlr.R300019-JLR200
Kujiraoka T, Satoh Y, Ayaori M, Shiraishi Y, Arai-Nakaya Y, Hakuno D, Yada H, Kuwada N, Endo S, Isoda K, Adachi T (2013) Hepatic extracellular signal-regulated kinase 2 suppresses endoplasmic reticulum stress and protects from oxidative stress and endothelial dysfunction. J Am Heart Assoc 2(4):1–18. https://doi.org/10.1161/JAHA.113.000361
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685. https://doi.org/10.1038/227680a0
Lowry OH, Rosebrough NJ, Farr AL, Randall RJJ (1951) Protein measurement with the Folin phenol reagent. Biol Chem 193:265–275
Luz C, Tigano MS, Silva IG, Cordeiro CMT, Aljanabi SM (1998) Selection of Beauveria bassiana and Metarhizium anisopliae isolates to control Triatoma infestans. Mem I Oswaldo Cruz 93(6):839–846. https://doi.org/10.1590/S0074-02761998000600026
Manzano-Roman R, Almazán C, Naranjo V, Blouin EF, Kocan KM, de la Fuente J (2008) Expression of perilipin in human promyelocytic cells in response to Anaplasma phagocytophilum infection results in modified lipid metabolism. J Med Microbiol 57(2):159–163. https://doi.org/10.1099/jmm.0.47504-0
Monteiro CMO, Araujo LX, Matos RS, Golo P, Angelo IC, Perinotto WMS, Rodrigues CAC, Furlong J, Bittencourt VREP, Prata MCA (2013) Association between entomopathogenic nematodes and fungi for control of Rhipicephalus microplus (Acari: Ixodidae). Parasitol Res 112(10):3645–3651. https://doi.org/10.1007/s00436-013-3552-7
Moret Y, Schmid-Hempel P (2000) Survival for immunity: the price of immune system activation for bumblebee workers. Science 290(5494):1166–1168. https://doi.org/10.1126/science.290.5494.1166
Nassaji M, Ghorbani R (2012) Plasma lipid levels in patients with acute bacterial infections. Turk J Med Sci 42:465–469. https://doi.org/10.3906/sag-1011-1278
Pavela R, Canale A, Mehlhorn H, Benelli G (2016) Application of ethnobotanical repellents and acaricides in prevention, control and management of livestock ticks: a review. Res Vet Sci 109:1–9. https://doi.org/10.1016/j.rvsc.2016.09.001
Quinelato S, Golo PS, Perinotto WMS, Sá FA, Camargo MG, Angelo IC, Moraes AML, Bittencourt VREP (2012) Virulence potential of Metarhizium anisopliae s.l. isolates on Rhipicephalus (Boophilus) microplus larvae. Vet Parasitol 190(3-4):556–565. https://doi.org/10.1016/j.vetpar.2012.06.028
Schmid MR, Anderl I, Vesala L, Vanha-aho LM, Deng XJ, Rämet M, Hultmark D (2014) Control of Drosophila blood cell activation via toll signaling in the fat body. PLoS One 9(8):1–10. https://doi.org/10.1371/journal.pone.0102568
Towler MC, Hardie DG (2007) AMP-activated protein kinase in metabolic control and insulin signaling. Circ Res 100(3):328–341. https://doi.org/10.1161/01.RES.0000256090.42690.05
Ziegler R, van Antwerpen R (2006) Lipid uptake by insect oocytes. Insect Biochem Molec 36(4):264–272. https://doi.org/10.1016/j.ibmb.2006.01.014
Acknowledgments
We thank Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and CNPq for providing graduation scholarships for Fillipe Sá and Caio Coutinho-Rodrigues during the elaboration of this paper. V.R.E.P. Bittencourt, G.C. Atella and M.A.C Silva-Neto (In memorian) are CNPq researchers.
Funding
This research was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) of Brazil, Instituto Nacional de Ciência e Tecnologia em Entomologia Molecular (INCT-EM), and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) from Rio de Janeiro State, Brazil.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mário Alberto C. Silva-Neto is deceased.
Electronic supplementary material
Supplementary Table 1
. Isolate, host, or substrate and localization of M. anisopliae s.l. isolates are used in this study. All were generously provided by Embrapa Recursos Genéticos e Biotecnologia. CG = isolates. (DOCX 13 kb).
Rights and permissions
About this article
Cite this article
Sá, F.A., Coutinho-Rodrigues, C.J.B., Angelo, I.C. et al. Metarhizium anisopliae s.l. modulation of lipid metabolism during tick infection is independent of AMPK and ERK pathways. Parasitol Res 117, 793–799 (2018). https://doi.org/10.1007/s00436-018-5753-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-5753-6