Abstract
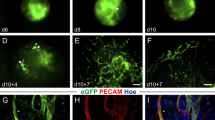
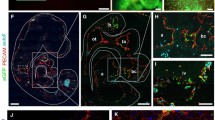
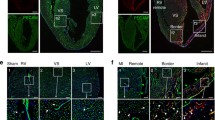
Vessel formation is of critical importance for organ function in the normal and diseased state. In particular, the labeling and quantitation of small vessels prove to be technically challenging using current approaches. We have, therefore, established a transgenic embryonic stem (ES) cell line and a transgenic mouse model where the vascular endothelial growth factor receptor VEGFR-1 (flt-1) promoter drives the expression of the live reporter eGFP. Fluorescence microscopy and immunostainings revealed endothelial-specific eGFP labeling of vascular networks. The expression pattern recapitulates that of the endogenous flt-1 gene, because small and large vessels are labeled by eGFP during embryonic development; after birth, the expression becomes more restricted to small vessels. We have explored this in the cardiovascular system more in detail and found that all small vessels and capillaries within the heart are strongly eGFP+. In addition, myocardial injuries have been induced in transgenic mice and prominent vascular remodeling, and an increase in endothelial cell area within the peri-infarct area could be observed underscoring the utility of this mouse model. Thus, the transgenic flt-1/eGFP models are powerful tools to investigate and quantify vascularization in vivo and to probe the effect of different compounds on vessel formation in vitro.






Similar content being viewed by others
References
Bernatchez PN, Soker S, Sirois MG (1999) Vascular endothelial growth factor effect on endothelial cell proliferation, migration, and platelet-activating factor synthesis is flk-1-dependent. J Biol Chem 274:31047–31054. doi:10.1074/jbc.274.43.31047
Breier G, Clauss M, Risau W (1995) Coordinate expression of vascular endothelial growth factor receptor-1 (flt-1) and its ligand suggests a paracrine regulation of murine vascular development. Dev Dyn 204:228–239. doi:10.1002/aja.1002040303
Bremer S, Worth AP, Paparella M, Bigot K, Kolossov E, Fleischmann BK, Hescheler J, Balls M (2001) Establishment of an in vitro reporter gene assay for developmental cardiac toxicity. Toxicol In Vitro 15:215–223. doi:10.1016/S0887-2333(01)00007-8
Dor Y, Camenisch TD, Itin A, Fishman GI, McDonald JA, Carmeliet P, Keshet E (2001) A novel role for VEGF in endocardial cushion formation and its potential contribution to congenital heart defects. Development 128:1531–1538
Fong GH, Klingensmith J, Wood CR, Rossant J, Breitman ML (1996) Regulation of flt-1 expression during mouse embryogenesis suggests a role in the establishment of vascular endothelium. Dev Dyn 207:1–10. doi:10.1002/(SICI)1097-0177(199609)207:1<1:AID-AJA1>3.0.CO;2-M
Frank NY, Schatton T, Kim S, Zhan Q, Wilson BJ, Ma J, Saab KR, Osherov V, Widlund HR, Gasser M, Waaga-Gasser AM, Kupper TS, Murphy GF, Frank MH (2011) VEGFR-1 expressed by malignant melanoma-initiating cells is required for tumor growth. Cancer Res 71:1474–1485
George SH, Gertsenstein M, Vintersten K, Korets-Smith E, Murphy J, Stevens ME, Haigh JJ, Nagy A (2007) Developmental and adult phenotyping directly from mutant embryonic stem cells. Proc Natl Acad Sci U S A 104:4455–4460. doi:10.1073/pnas.0609277104
Hazarika S, Dokun AO, Li Y, Popel AS, Kontos CD, Annex BH (2007) Impaired angiogenesis after hindlimb ischemia in type 2 diabetes mellitus: differential regulation of vascular endothelial growth factor receptor 1 and soluble vascular endothelial growth factor receptor 1. Circ Res 101:948–956. doi:10.1161/CIRCRESAHA.107.160630
Hidaka M, Stanford WL, Bernstein A (1999) Conditional requirement for the flk-1 receptor in the in vitro generation of early hematopoietic cells. Proc Natl Acad Sci USA 96:7370–7375. doi:10.1073/pnas.96.13.7370
Kaplan RN, Riba RD, Zacharoulis S, Bramley AH, Vincent L, Costa C, MacDonald DD, Jin DK, Shido K, Kerns SA, Zhu Z, Hicklin D, Wu Y, Port JL, Altorki N, Port ER, Ruggero D, Shmelkov SV, Jensen KK, Rafii S, Lyden D (2005) VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438:820–827. doi:10.1038/nature04186
Kolossov E, Bostani T, Roell W, Breitbach M, Pillekamp F, Nygren JM, Sasse P, Rubenchik O, Fries JW, Wenzel D, Geisen C, Xia Y, Lu Z, Duan Y, Kettenhofen R, Jovinge S, Bloch W, Bohlen H, Welz A, Hescheler J, Jacobsen SE, Fleischmann BK (2006) Engraftment of engineered ES cell-derived cardiomyocytes but not BM cells restores contractile function to the infarcted myocardium. J Exp Med 203:2315–2327. doi:10.1084/jem.20061469
Krijnen PA, Hahn NE, Kholova I, Baylan U, Sipkens JA, van Alphen FP, Vonk AB, Simsek S, Meischl C, Schalkwijk CG, van Buul JD, van Hinsberg VW, Niessen HW (2012) Loss of DPP4 activity is related to a prothrombogenic status of endothelial cells: implications for the coronary microvasculature of myocardial infarction patients. Basic Res Cardiol 107:1–13. doi:10.1007/s00395-011-0233-5
Lahteenvuo JE, Lahteenvuo MT, Kivela A, Rosenlew C, Falkevall A, Klar J, Heikura T, Rissanen TT, Vahakangas E, Korpisalo P, Enholm B, Carmeliet P, Alitalo K, Eriksson U, Yla-Herttuala S (2009) Vascular endothelial growth factor-B induces myocardium-specific angiogenesis and arteriogenesis via vascular endothelial growth factor receptor-1- and neuropilin receptor-1-dependent mechanisms. Circulation 119:845–856. doi:10.1161/CIRCULATIONAHA.108.8164
Lassaletta AD, Chu LM, Sellke FW (2011) Therapeutic neovascularization for coronary disease: current state and future prospects. Basic Res Cardiol 106:897–909. doi:10.1007/s00395-011-0200-1
Li J, Brown LF, Hibberd MG, Grossman JD, Morgan JP, Simons M (1996) VEGF, flk-1, and flt-1 expression in a rat myocardial infarction model of angiogenesis. Am J Physiol 270:H1803–H1811
Luttun A, Tjwa M, Moons L, Wu Y, Angelillo-Scherrer A, Liao F, Nagy JA, Hooper A, Priller J, De KB, Compernolle V, Daci E, Bohlen P, Dewerchin M, Herbert JM, Fava R, Matthys P, Carmeliet G, Collen D, Dvorak HF, Hicklin DJ, Carmeliet P (2002) Revascularization of ischemic tissues by PlGF treatment, and inhibition of tumor angiogenesis, arthritis and atherosclerosis by anti-flt1. Nat Med 8:831–840. doi:10.1038/nm731
Malan D, Wenzel D, Schmidt A, Geisen C, Raible A, Bolck B, Fleischmann BK, Bloch W (2012) Endothelial beta-1 integrins regulate sprouting and network formation during vascular development. Development 137:993–1002. doi:10.1242/dev.045377
Marchetti S, Gimond C, Iljin K, Bourcier C, Alitalo K, Pouyssegur J, Pages G (2002) Endothelial cells genetically selected from differentiating mouse embryonic stem cells incorporate at sites of neovascularization in vivo. J Cell Sci 115:2075–2085
Marti HH, Risau W (1998) Systemic hypoxia changes the organ-specific distribution of vascular endothelial growth factor and its receptors. Proc Natl Acad Sci USA 95:15809–15814. doi:10.1073/pnas.95.26.15809
Milberg P, Klocke R, Frommeyer G, Quang TH, Dieks K, Stypmann J, Osada N, Kuhlmann M, Fehr M, Milting H, Nikol S, Waltenberger J, Breithardt G, Eckardt L (2011) G-CSF therapy reduces myocardial repolarization reserve in the presence of increased arteriogenesis, angiogenesis and connexin 43 expression in an experimental model of pacing-induced heart failure. Basic Res Cardiol 106:995–1008. doi:10.1007/s00395-011-0230-8
Nagy A, Gocza E, Diaz EM, Prideaux VR, Ivanyi E, Markkula M, Rossant J (1990) Embryonic stem cells alone are able to support fetal development in the mouse. Development 110:815–821
Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z (1999) Vascular endothelial growth factor (VEGF) and its receptors. FASEB J 13:9–22
Oerlemans MI, Goumans MJ, van MB, Clevers H, Doevendans PA, Sluijter JP (2012). Active Wnt signaling in response to cardiac injury. Basic Res Cardiol 105:631–641. doi:10.1007/s00395-010-0100-9
Ohtani K, Dimmeler S (2011) Control of cardiovascular differentiation by microRNAs. Basic Res Cardiol 106:5–11. doi:10.1007/s00395-010-0139-7
Peters KG, De VC, Williams LT (1993) Vascular endothelial growth factor receptor expression during embryogenesis and tissue repair suggests a role in endothelial differentiation and blood vessel growth. Proc Natl Acad Sci USA 90:8915–8919. doi:10.1073/pnas.90.19.8915
Quinn G, Ochiya T, Terada M, Yoshida T (2000) Mouse flt-1 promoter directs endothelial-specific expression in the embryoid body model of embryogenesis. Biochem Biophys Res Commun 276:1089–1099. doi:10.1006/bbrc.2000.3602
Redick SD, Bautch VL (1999) Developmental platelet endothelial cell adhesion molecule expression suggests multiple roles for a vascular adhesion molecule. Am J Pathol 154:1137–1147. doi:10.1016/S0002-9440(10)65366-7
Roell W, Lewalter T, Sasse P, Tallini YN, Choi BR, Breitbach M, Doran R, Becher UM, Hwang SM, Bostani T, von MJ, Hofmann A, Reining S, Eiberger B, Gabris B, Pfeifer A, Welz A, Willecke K, Salama G, Schrickel JW, Kotlikoff MI, Fleischmann BK (2007) Engraftment of connexin 43-expressing cells prevents post-infarct arrhythmia. Nature 450:819–824. doi:10.1038/nature06321
Roell W, Lu ZJ, Bloch W, Siedner S, Tiemann K, Xia Y, Stoecker E, Fleischmann M, Bohlen H, Stehle R, Kolossov E, Brem G, Addicks K, Pfitzer G, Welz A, Hescheler J, Fleischmann BK (2002) Cellular cardiomyoplasty improves survival after myocardial injury. Circulation 105:2435–2441. doi:10.1161/01.CIR.0000016063.66513.BB
Sacchetti A, El ST, Nasr AF, Alberti S (2001) Efficient GFP mutations profoundly affect mRNA transcription and translation rates. FEBS Lett 492:151–155. doi:10.1016/S0014-5793(01)02246-3
Stankunas K, Ma GK, Kuhnert FJ, Kuo CJ, Chang CP (2010) VEGF signaling has distinct spatiotemporal roles during heart valve development. Dev Biol 347:325–336
Ulyatt C, Walker J, Ponnambalam S (2011) Hypoxia differentially regulates VEGFR1 and VEGFR2 levels and alters intracellular signaling and cell migration in endothelial cells. Biochem Biophys Res Commun 404:774–779
Wenzel D, Knies R, Matthey M, Klein AM, Welschoff J, Stolle V, Sasse P, Roll W, Breuer J, Fleischmann BK (2009) Beta(2)-adrenoceptor antagonist ICI 118,551 decreases pulmonary vascular tone in mice via a G(i/o) protein/nitric oxide-coupled pathway. Hypertension 54:157–163. doi:10.1161/HYPERTENSIONAHA.109.130468
Wenzel D, Rieck S, Vosen S, Mykhaylyk O, Trueck C, Eberbeck D, Trahms L, Zimmeremann K, Pfeifer A, Fleischmann BK (2012) Identification of magnetic nanoparticles for combined positioning and lentiviral transduction of endothelial cells. Pharm Res. doi:10.1007/s11095-011-0657-5
Witmer AN, Dai J, Weich HA, Vrensen GF, Schlingemann RO (2002) Expression of vascular endothelial growth factor receptors 1, 2, and 3 in quiescent endothelia. J Histochem Cytochem 50:767–777. doi:10.1177/002215540205000603
Wobus AM, Wallukat G, Hescheler J (1991) Pluripotent mouse embryonic stem cells are able to differentiate into cardiomyocytes expressing chronotropic responses to adrenergic and cholinergic agents and Ca2+ channel blockers. Differentiation 48:173–182
Wu J, Li J, Zhang N, Zhang C (2011) Stem cell-based therapies in ischemic heart diseases: a focus on aspects of microcirculation and inflammation. Basic Res Cardiol 106:317–324. doi:10.1007/s00395-011-0168-x
Xiao J, Moon M, Yan L, Nian M, Zhang Y, Liu C, Lu J, Guan H, Chen M, Jiang D, Jiang H, Liu PP, Li H (2012) Cellular FLICE-inhibitory protein protects against cardiac remodelling after myocardial infarction. Basic Res Cardiol 107:1–21. doi:10.1007/s00395-011-0239-z
Zentilin L, Puligadda U, Lionetti V, Zacchigna S, Collesi C, Pattarini L, Ruozi G, Camporesi S, Sinagra G, Pepe M, Recchia FA, Giacca M (2010) Cardiomyocyte VEGFR-1 activation by VEGF-B induces compensatory hypertrophy and preserves cardiac function after myocardial infarction. FASEB J 24:1467–1478
Acknowledgments
We thank A. Nagy (Mount Sinai Hospital Toronto) and M. Gertsenstein (Toronto, Canada) for providing the G4 ES cell line, N. Copeland (NCI, Frederick, USA) for providing SW105 cells, R. Schneider-Kramann (University of Aachen) for help with teratoma analysis and Y. Matuschek (University of Bonn) for assistance in vector cloning. Funding was provided to the junior research group of D.W. by the Ministry of Innovation, Science, Research and Technology of the State of North Rhine-Westphalia. This work was further supported EU FP7 consortium CardioCell Grant No. 223372 (to BKF).
Author information
Authors and Affiliations
Corresponding author
Additional information
K. Herz and J. C. Heinemann contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Suppl. Fig. 1: Re-formation of vascular networks after dissociation of flt-1/eGFP EBs. a) After enzymatic dissociation of day 5 (d 5) EBs and re-plating on gelatine single eGFP+ cells were visible one day later. b) After 3 days eGFP+ sprouts have formed (arrow). c) The sprouts generated network-like vascular structures 7 days after dissociation. d) Re-formed vascular networks were found through day 12, bar= 300 μm.
Suppl. Fig. 2: EGFP+ vessels in adult organs. a-f) Immunohistochemistry revealed endothelial-specific eGFP expression in vessels of the thymus of chimeric (a-c) and of the kidney of heterozygous mice (d-f), inset: also vessels of the glomeruli were eGFP+, green=eGFP, red=PECAM, blue=Hoechst, magenta=autofluorescence, bars=50 μm.
Suppl. Fig. 3: EGFP expression in the endothelium of large adult vessels. a-i) Immunohistochemistry showed that adult aorta displayed no eGFP labeling in endothelial cells (a-c), this was also true for the carotid artery (d-f). In contrast, the endothelium of the murine jugular vein was highly eGFP+ (g-i), green=eGFP, red=PECAM, bar=50 μm.
Suppl. Fig. 4: EGFP+ structures and leukocytes invading the peri-infarct zone after cryoinfarction. a-c) Immunohistochemistry showed that eGFP+ vascular structures in the peri-infarct zone 3 days after cryoinfarction were CD45- (green=eGFP, magenta=CD45, blue=Hoechst), bar=50 μm.
Suppl. Video file: The video shows sprouting angiogenesis within an flt-1/eGFP BAC EB on day 5+5. Vascular sprouts elongated and connected to early vascular networks. Video covered app. 12 h (acquisition at 4 frames per hour (fph), display rate at 10 fps) and was recorded with a 20× objective.




Supplementary Video(MPEG 446 kb)
Rights and permissions
About this article
Cite this article
Herz, K., Heinemann, J.C., Hesse, M. et al. Live monitoring of small vessels during development and disease using the flt-1 promoter element. Basic Res Cardiol 107, 257 (2012). https://doi.org/10.1007/s00395-012-0257-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-012-0257-5