Abstract
Purpose
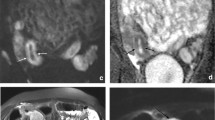
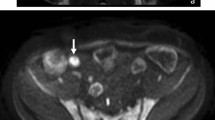
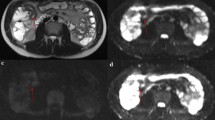
To compare the accuracy of MR enterography (MRE) using combined T2-weighted and contrast-enhanced (CE) sequences with that of combined T2- and diffusion-weighted (DW) sequences for the detection of complex enteric Crohn’s disease (CD).
Materials
Thirty-eight patients who underwent surgery for CD complications and preoperative MRE from 2011 to 2016 were included. MRE examinations were blindly analyzed independently by one junior and one senior abdominal radiologist for the presence of fistula, stenosis and abscesses. During a first reading session, T2-weighted images (WI), steady-state sequences and DW-MRE were reviewed (set 1). During a separate distant session, T2-WI, True-FISP and CE-MRE were reviewed (set 2). Performance of each reader was evaluated by comparison with the standard of reference established using intraoperative and pathological findings.
Results
Forty-eight fistulas, 43 stenoses and 11 abscesses were found. For the senior radiologist, sensitivity for the detection of fistula, stenosis and abscess ranged from 80% to 100% for set 1 and 88% to 100% for set 2 and specificity ranged from 56% to 70% for set 1 and 53% to 93% for set 2, with no significant difference between the sets (p = 0.342–0.429). For the junior radiologist, sensitivity ranged from 53% to 63% for set 1 and 64% to 88% for set 2 and specificity ranged from 0% to 25% for set 1 and 17% to 40% for set 2 (p = 0.001 and 0.007, respectively).
Conclusion
For a senior radiologist, DW-MRE has similar sensitivity as CE-MRE for the detection of CD complications. For a junior radiologist, CE-MRE yields the best results compared with DW-MRE.
Key Points
• For experienced readers, DWI has similar diagnostic capability as contrast-enhanced MR imaging for the diagnosis of Crohn’s disease complications.
• For senior radiologists, gadolinium chelate injection could be waived for the diagnosis of Crohn’s disease complications.
• The interpretation of DWI for Crohn’s disease complications requires some experience.



Similar content being viewed by others
Abbreviations
- CD:
-
Crohn’s disease
- CE:
-
Contrast-enhanced
- DWI:
-
Diffusion-weighted imaging
- HASTE:
-
Half-Fourier acquisition single-shot turbo spin-echo
- MRE:
-
Magnetic resonance enterography
- MRI:
-
Magnetic resonance imaging
- NA:
-
Non applicable
- NPV:
-
Negative predictive value
- PACS:
-
Picture archiving and communication system
- PPV:
-
Positive predictive value
- SD:
-
Standard deviation
- TNF:
-
Tumor necrosis factor
- TP:
-
True positive
- True-FISP:
-
True fast imaging with steady-state precession
- VIBE:
-
Volumetric interpolated breath-hold examination
- WI:
-
Weighted imaging
References
Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L (2017) Crohn’s disease. Lancet 389:1741–1755
Molodecky NA, Soon IS, Rabi DM et al (2012) Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 142:46–54 e42 quiz e30
Pariente B, Cosnes J, Danese S et al (2011) Development of the Crohn's disease digestive damage score, the Lemann score. Inflamm Bowel Dis 17:1415–1422
Barral M, Dohan A, Allez M et al (2016) Gastrointestinal cancers in inflammatory bowel disease: an update with emphasis on imaging findings. Crit Rev Oncol Hematol 97:30–46
Hristova L, Soyer P, Hoeffel C et al (2013) Colorectal cancer in inflammatory bowel diseases: CT features with pathological correlation. Abdom Imaging 38:421–435
Goyer P, Alves A, Bretagnol F, Bouhnik Y, Valleur P, Panis Y (2009) Impact of complex Crohn's disease on the outcome of laparoscopic ileocecal resection: a comparative clinical study in 124 patients. Dis Colon Rectum 52:205–210
Harb WJ (2015) Crohn's Disease of the Colon, Rectum, and Anus. Surg Clin North Am 95(1195-1210):vi
Malgras B, Soyer P, Boudiaf M et al (2012) Accuracy of imaging for predicting operative approach in Crohn's disease. Br J Surg 99:1011–1020
Stoddard PB, Ghazi LJ, Wong-You-Cheong J, Cross RK, Vandermeer FQ (2015) Magnetic resonance enterography: state of the art. Inflamm Bowel Dis 21:229–239
Van Assche G, Dignass A, Panes J et al (2010) The second European evidence-based Consensus on the diagnosis and management of Crohn's disease: Definitions and diagnosis. J Crohns Colitis 4:7–27
Zappa M, Stefanescu C, Cazals-Hatem D et al (2011) Which magnetic resonance imaging findings accurately evaluate inflammation in small bowel Crohn's disease? A retrospective comparison with surgical pathologic analysis. Inflamm Bowel Dis 17:984–993
Pola S, Santillan C, Levesque BG, Feagan BG, Sandborn WJ (2014) An overview of magnetic resonance enterography for Crohn's disease. Dig Dis Sci 59:2040–2049
Erden A, Ünal S, Akkaya HE et al (2017) MR Enterography in Crohn's disease complicated with enteroenteric fistula. Eur J Radiol. https://doi.org/10.1016/j.ejrad.2017.06.012
You-Ling Shyu J, Chick JF, Adackapara CA, Adduci AJ (2016) Urachal-colonic fistula: MR imaging and MDCT features. Diagn Interv Imaging 97:105–108
Dohan A, Eveno C, Oprea R et al (2014) Diffusion-weighted MR imaging for the diagnosis of abscess complicating fistula-in-ano: preliminary experience. Eur Radiol 24:2906–2915
Monnier L, Dohan A, Amara N et al (2017) Anoperineal disease in hidradenitis suppurativa: MR imaging distinction from perianal Crohn's disease. Eur Radiol 27:4100–4109
Dohan A, Taylor S, Hoeffel C et al (2016) Diffusion-weighted MRI in Crohn's disease: Current status and recommendations. J Magn Reson Imaging 44:1381–1396
Seo N, Park SH, Kim KJ et al (2016) MR enterography for the evaluation of small-bowel inflammation in Crohn disease by using diffusion-weighted imaging without intravenous contrast material: a prospective noninferiority study. Radiology 278:762–772
Sinha R, Rajiah P, Ramachandran I, Sanders S, Murphy PD (2013) Diffusion-weighted MR imaging of the gastrointestinal tract: technique, indications, and imaging findings. Radiographics 33:655–676
Tielbeek JA, Ziech ML, Li Z et al (2014) Evaluation of conventional, dynamic contrast enhanced and diffusion weighted MRI for quantitative Crohn's disease assessment with histopathology of surgical specimens. Eur Radiol 24:619–629
Schmid-Tannwald C, Agrawal G, Dahi F, Sethi I, Oto A (2012) Diffusion-weighted MRI: role in detecting abdominopelvic internal fistulas and sinus tracts. J Magn Reson Imaging 35:125–131
Cavusoglu M, Duran S, Sozmen Ciliz D et al (2017) Added value of diffusion-weighted magnetic resonance imaging for the diagnosis of perianal fistula. Diagn Interv Imaging 98:401–408
Olchowy C, Cebulski K, Łasecki M et al (2017) The presence of the gadolinium-based contrast agent depositions in the brain and symptoms of gadolinium neurotoxicity - A systematic review. PLoS One 12:e0171704
Soyer P, Dohan A, Patkar D, Gottschalk A (2017) Observational study on the safety profile of gadoterate meglumine in 35,499 patients: The SECURE study. J Magn Reson Imaging 45:988–997
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Seastedt KP, Trencheva K, Michelassi F et al (2014) Accuracy of CT enterography and magnetic resonance enterography imaging to detect lesions preoperatively in patients undergoing surgery for Crohn's disease. Dis Colon Rectum 57:1364–1370
Schmidt S, Chevallier P, Bessoud B et al (2007) Diagnostic performance of MRI for detection of intestinal fistulas in patients with complicated inflammatory bowel conditions. Eur Radiol 17:2957–2963
Taylor SA, Avni F, Cronin CG et al (2017) The first joint ESGAR/ESPR consensus statement on the technical performance of cross-sectional small bowel and colonic imaging. Eur Radiol 27:2570–2582
Fiorino G, Bonifacio C, Peyrin-Biroulet L et al (2011) Prospective comparison of computed tomography enterography and magnetic resonance enterography for assessment of disease activity and complications in ileocolonic Crohn's disease. Inflamm Bowel Dis 17:1073–1080
Baik J, Kim SH, Lee Y, Yoon JH (2017) Comparison of T2-weighted imaging, diffusion-weighted imaging and contrast-enhanced T1-weighted MR imaging for evaluating perianal fistulas. Clin Imaging 44:16–21
Yoshizako T, Wada A, Takahara T et al (2012) Diffusion-weighted MRI for evaluating perianal fistula activity: feasibility study. Eur J Radiol 81:2049–2053
Rimola J, Planell N, Rodríguez S et al (2015) Characterization of inflammation and fibrosis in Crohn's disease lesions by magnetic resonance imaging. Am J Gastroenterol 110:432–440
Li XH, Sun CH, Mao R et al (2017) Diffusion-weighted MRI enables to accurately grade inflammatory activity in patients of ileocolonic Crohn's disease: results from an observational study. Inflamm Bowel Dis 23:244–253
Nguyen TL, Soyer P, Barbe C et al (2013) Diagnostic value of diffusion-weighted magnetic resonance imaging in pelvic abscesses. J Comput Assist Tomogr 37:971–979
Norris DG, Niendorf T, Leibfritz D (1994) Health and infarcted brain tissues studied at short diffusion times: the origins of apparent restriction and the reduction in apparent diffusion coefficient. NMR Biomed 7:304–310
Sugahara T, Korogi Y, Kochi M et al (1999) Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 9:53–60
Chan JH, Tsui EY, Luk SH et al (2001) Diffusion-weighted MR imaging of the liver: distinguishing hepatic abscess from cystic or necrotic tumor. Abdom Imaging 26:161–165
Le Bihan D, Breton E, Lallemand D, Aubin ML, Vignaud J, Laval-Jeantet M (1988) Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology 168:497–505
Le Bihan D, Breton E, Lallemand D, Grenier P, Cabanis E, Laval-Jeantet M (1986) MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurologic disorders. Radiology 161:401–407
Rosenkrantz AB, Padhani AR, Chenevert TL et al (2015) Body diffusion kurtosis imaging: Basic principles, applications, and considerations for clinical practice. J Magn Reson Imaging 42:1190–1202
Yantiss RK, Odze RD (2006) Diagnostic difficulties in inflammatory bowel disease pathology. Histopathology 48:116–132
Nasu K, Kuroki Y, Nawano S et al (2006) Hepatic metastases: diffusion-weighted sensitivity-encoding versus SPIO-enhanced MR imaging. Radiology 239:122–130
Oto A, Schmid-Tannwald C, Agrawal G et al (2011) Diffusion-weighted MR imaging of abdominopelvic abscesses. Emerg Radiol 18:515–524
Lu Y, Jin H, Genant HK (2003) On the non-inferiority of a diagnostic test based on paired observations. Stat Med 22:3029–3044
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is P. Soyer, MD, PhD.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and Biometry
Statistical analysis was performed by an expert senior statistician (Dr. AS Jannot)
Informed Consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical Approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnosis method
• performed at one institution
Rights and permissions
About this article
Cite this article
Barat, M., Hoeffel, C., Bouquot, M. et al. Preoperative evaluation of small bowel complications in Crohn’s disease: comparison of diffusion-weighted and contrast-enhanced MR imaging. Eur Radiol 29, 2034–2044 (2019). https://doi.org/10.1007/s00330-018-5734-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5734-2