Abstract
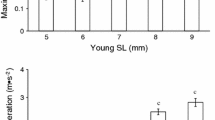
Innate antipredator responses are integral for survival in many species, particularly those which lack parental care. Antipredator responses include both active (fight or flight) and passive behaviours (immobility). As the success of antipredator responses directly relates to survival and fitness, investigating the drivers that explain variance in these traits is key to understanding how predation shapes the instinctive behaviour of animals. We quantified innate antipredator behaviour of hatchling Australian water dragons (Intellagama lesueurii) immediately after hatching using a model snake to simulate a series of attacks, and scored their behaviour using a fight or flight index. Then we explored which factors were related to dragon antipredator behaviour, such as habitat disturbance, origin population, morphology, and parental genetic effects and phenotype (clutch effects). We developed multiple hypotheses and used model selection to determine which factors drive variation in hatchling antipredator behaviour. Clutch effects explained a significant proportion of variation in innate antipredator responses, suggesting a heritable component. We also found an effect of body size on innate antipredator behaviour: larger hatchlings were more prone to flight behaviour (e.g. short-distance runs and long-distance sprinting), while smaller individuals were more prone to standing their ground and being aggressive (e.g. throat puffing, mouth gaping, biting). Clutch effects also explained a significant proportion of the variance in dragon body size. Our study provides evidence that the innate antipredator responses of water dragons are heritable in origin (directly through clutch effects, and indirectly through body size) and not associated with particular populations or habitat types. We suggest future research examine the survival implications of these responses.
Significance statement
The action an animal takes in response to a predator is a life or death decision, and can be required immediately after birth. These innate antipredator behaviours may be genetically linked, and enable individuals to emerge into their environment with the necessary behaviour to promote survival. We examined what factors drive hatchling lizards to exhibit different innate antipredator behaviour. Our study found that body size affected their innate behaviour: larger hatchlings were more prone to flee and smaller hatchlings were more likely to fight. Interestingly, parental genetics and phenotype (clutch effects) also significantly explained the variation in innate antipredator behaviour, which supports the hypothesis that these behaviours are heritable. Understanding what drives variation is a cornerstone of evolutionary biology, and our findings raise questions about how selection acts on antipredator behaviour and the degree to which they are plastic.



Similar content being viewed by others
References
Åbjörnsson K, Hansson LA, Brönmark C (2004) Responses of prey from habitats with different predator regimes: local adaptation and heritability. Ecology 85:1859–1866. https://doi.org/10.1890/03-0074
Ariyomo TO, Carter M, Watt PJ (2013) Heritability of boldness and aggressiveness in the zebrafish. Behav Genet 43:161–167. https://doi.org/10.1007/s10519-013-9585-y
Arnold SJ, Bennett AF (1984) Behavioural variation in natural populations. III: antipredator displays in the garter snake Thamnophis radix. Anim Behav 32:1108–1118
de Barros FC, de Carvalho JE, Abe AS, Kohlsdorf T (2010) Fight versus flight: the interaction of temperature and body size determines antipredator behaviour in tegu lizards. Anim Behav 79:83–88. https://doi.org/10.1016/j.anbehav.2009.10.006
Barton K (2009) MuMIn: multi-model inference. In: R package version 0.12.0. http://r-forge.r-project.org/projects/mumin/
Bateman PW, Fleming PA (2014) Living on the edge: effects of body size, group density and microhabitat selection on escape behaviour of southern leopard frogs Lithobates sphenocephalus. Curr Zool 60:712–718. https://doi.org/10.1093/czoolo/60.6.712
Bateman AW, Vos M, Anholt BR (2014) When to defend: antipredator defenses and the predation sequence. Am Nat 183:847–855. https://doi.org/10.1086/675903
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Bellairs A (1970) The life of reptiles (volume 1). Universe Books, New York, USA
Brodie ED III (1989) Genetic correlations between morphology and antipredator behaviour in natural populations of the garter snake Thamnophis ordinoides. Nature 342:542–543. https://doi.org/10.1038/342542a0
Brown C, Laland KN (2003) Social learning in fishes: a review. Fish Fish 4:280–288. https://doi.org/10.1046/j.1467-2979.2003.00122.x
Burnham KP, Anderson DR (2003) Model selection and multimodel inference: a practical information-theoretic approach. Springer Science & Business Media, New York, USA
Canty A, Ripley B (2017) Boot: bootstrap R (S-plus) functions. R package version 1:3–20 https://cran.r-project.org/web/packages/boot/boot.pdf
Caro T (2005) Antipredator defenses in birds and mammals. University of Chicago Press, Chicago
Caro T (2014) Antipredator deception in terrestrial vertebrates. Curr Zool 60:16–25. https://doi.org/10.1093/czoolo/60.1.16
Cogger HG (2014) Reptiles and amphibians of Australia, 7th edn. Reed Books, Chatswood, NSW
Cooper WE Jr, Wilson DS (2008) Thermal cost of refuge use affects refuge entry and hiding time by striped plateau lizards Sceloporus virgatus. Herpetologica 64:406–412. https://doi.org/10.1655/07-083R2.1
Dalesman S, Rundle SD, Coleman RA, Cotton PA (2006) Cue association and antipredator behaviour in a pulmonate snail, Lymnaea stagnalis. Anim Behav 71:789–797. https://doi.org/10.1016/j.anbehav.2005.05.028
Davison AC, Hinkley DV (1997) Bootstrap methods and their applications. Cambridge University Press, Cambridge, UK
Doody JS, Guarino E, Georges A, Corey B, Murray G, Ewert M (2006) Nest site choice compensates for climate effects on sex ratios in a lizard with environmental sex determination. Evol Ecol 20:307–330
Doody JS, Harlow P, Douglass D, Thiem JD, Broadhurst B, Trembath D, Olsen J, Fuentes E, Rose T (2014) Patterns of predation and antipredator behavior in the australian water dragon, Physignathus lesueurii. Herpetol Conserv Biol 9:48–56
Eilam D (2005) Die hard: a blend of freezing and fleeing as a dynamic defense—implications for the control of defensive behavior. Neurosci Biobehav Rev 29:1181–1191. https://doi.org/10.1016/j.neubiorev.2005.03.027
Ferrari MC, Sih A, Chivers DP (2009) The paradox of risk allocation: a review and prospectus. Anim Behav 78:579–585. https://doi.org/10.1016/j.anbehav.2009.05.034
Foster KL, Collins CE, Higham TE, Garland TJ Jr (2015) Determinants of lizard escape performance: decision, motivation, ability, and opportunity. In: Cooper WE, Blumstein DT (eds) Escaping from predators: an integrative view of escape decisions and refuge use. Cambridge University Press, Cambridge, UK, pp 287–321
Giesing E R, Suski CD, Warner RE, Bell AM (2010) Female sticklebacks transfer information via eggs: effects of maternal experience with predators on offspring. Proc R Soc Lond B Biol Sci, rspb20101819
Greene HW (1988) Antipredator mechanisms in reptiles. In: Gans C, Huey RB (eds) Biology of the Reptilia. Alan R. Liss, New York, USA, pp 1–152
Harlow PS (2001) Ecology of sex-determining mechanisms in Australian agamid lizards. PhD thesis, School of Biological Sciences, Macquarie University, Sydney, Australia
Harlow PS, Harlow MF (1997) Captive reproduction and longevity in the eastern water dragon (Physignathus lesueurii). Herpetofauna 27:14–19
Harlow PS, Taylor JE (2000) Reproductive ecology of the jacky dragon (Amphibolurus muricatus): an agamid lizard with temperature-dependent sex determination. Aust Ecol 25:640–652. https://doi.org/10.1111/j.1442-9993.2000.tb00070.x
Herzog HA, Burghardt GM (1986) Development of antipredator responses in snakes: I. Defensive and open-field behaviors in newborns and adults of three species of garter snakes (Thamnophis melanogaster, T. sirtalis, T. butleri). J Comp Psychol 100:372–379. https://doi.org/10.1037/0735-7036.100.4.372
Jenssen TA, Decourcy KR, Congdon JD (2005) Assessment in contests of male lizards (Anolis carolinensis): how should smaller males respond when size matters? Anim Behav 69:1325–1336. https://doi.org/10.1016/j.anbehav.2004.07.023
Johnson MT, Munshi-South J (2017) Evolution of life in urban environments. Science 358:eaam8327. https://doi.org/10.1126/science.aam8327
Just W, Morris MR (2003) The Napoleon complex: why smaller males pick fights. Evol Ecol 17:509–522. https://doi.org/10.1023/B:EVEC.0000005629.54152.83
Kar F, Whiting MJ, Noble DW (2016) Influence of prior contest experience and level of escalation on contest outcome. Behav Ecol Sociobiol 70:1679–1687. https://doi.org/10.1007/s00265-016-2173-4
Kissner KJ, Forbes MR, Secoy DM (1997) Rattling behavior of prairie rattlesnakes (Crotalus viridis viridis, Viperidae) in relation to sex, reproductive status, body size, and body temperature. Ethology 103:1042–1050. https://doi.org/10.1111/j.1439-0310.1997.tb00146.x
Kuo CY, Irschick DJ (2016) Ecology drives natural variation in an extreme antipredator trait: a cost–benefit analysis integrating modelling and field data. Funct Ecol 30:953–963. https://doi.org/10.1111/1365-2435.12593
Laurila A, Pakkasmaa S, Merilä J (2006) Population divergence in growth rate and antipredator defences in Rana arvalis. Oecologia 147:585–595. https://doi.org/10.1007/s00442-005-0301-3
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Lind J, Cresswell W (2005) Determining the fitness consequences of antipredation behavior. Behav Ecol 16:945–956. https://doi.org/10.1093/beheco/ari075
Littleford-Colquhoun BL, Clemente C, Whiting MJ, Ortiz-Barrientos D, Frère CH (2017) Archipelagos of the Anthropocene: rapid and extensive differentiation of native terrestrial vertebrates in a single metropolis. Mol Ecol 26:2466–2481. https://doi.org/10.1111/mec.14042
Losos JB (1990) The evolution of form and function: morphology and locomotor performance in West Indian Anolis lizards. Evolution 44:1189–1203. https://doi.org/10.1111/j.1558-5646.1990.tb05225.x
Lynch M, Walsh B (1998) Genetics and analysis of quantitative traits. Sinauer, Sunderland, MA, USA
Mader DR (1996) Reproductive surgery in the green iguana. Sem Avian Exot Pet 5:214–221
Martín J, López P (2003) Ontogenetic variation in antipredator behavior of Iberian rock lizards (Lacerta monticola): effects of body-size-dependent thermal-exchange rates and costs of refuge use. Can J Zool 81:1131–1137. https://doi.org/10.1139/z03-094
Martín J, Luque-Larena JJ, López P (2005) Factors affecting escape behavior of Iberian green frogs (Rana perezi). Can J Zool 83:1189–1194. https://doi.org/10.1139/z05-114
Morrell LJ, Lindström J, Ruxton GD (2005) Why are small males aggressive? Proc R Soc Lond B 272:1235–1241. https://doi.org/10.1098/rspb.2005.3085
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed effects models. Methods Ecol Evol 4:133–142. https://doi.org/10.1111/j.2041-210x.2012.00261.x
Noble DWA, McFarlane SE, Keogh JS, Whiting MJ (2014) Maternal and additive genetic effects contribute to variation in offspring traits in a lizard. Behav Ecol 25:633–640. https://doi.org/10.1093/beheco/aru032
Petelle MB, Martin JG, Blumstein DT (2015) Heritability and genetic correlations of personality traits in a wild population of yellow-bellied marmots (Marmota flaviventris). J Evol Biol 28:1840–1848. https://doi.org/10.1111/jeb.12700
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria https://www.R-project.org/
Ripley B (2018) Package ‘MASS. R package version 7:3–49 https://cran.r-project.org/web/packages/MASS/MASS.pdf
Robert KA, Vleck C, Bronikowski AM (2009) The effects of maternal corticosterone levels on offspring behavior in fast-and slow-growth garter snakes (Thamnophis elegans). Horm Behav 55:24–32. https://doi.org/10.1016/j.yhbeh.2008.07.008
Santos MBD, Oliveira MCLMD, Verrastro L, Tozetti AM (2010) Playing dead to stay alive: death-feigning in Liolaemus occipitalis (Squamata: Liolaemidae). Biota Neotrop 10:361–364. https://doi.org/10.1590/S1676-06032010000400043
Sih A, Bell AM, Johnson JC, Ziemba RE (2004) Behavioral syndromes: an integrative overview. Q Rev Biol 79:241–277. https://doi.org/10.1086/422893
Storm JJ, Lima SL (2010) Mothers forewarn offspring about predators: a transgenerational maternal effect on behavior. Am Nat 175:382–390. https://doi.org/10.1086/650443
Thaker M, Lima SL, Hews DK (2009) Acute corticosterone elevation enhances antipredator behaviors in male tree lizard morphs. Horm Behav 56:51–57. https://doi.org/10.1016/j.yhbeh.2009.02.009
Thompson MB (1993) Estimate of the population structure of the eastern water dragon, Physignathus lesueurii (Reptilia: Agamidae), along riverside habitat. Wildlife Res 20:613–619. https://doi.org/10.1071/WR9930613
Uller T, Olsson M (2006) Direct exposure to corticosterone during embryonic development influences behaviour in an ovoviviparous lizard. Ethology 112:390–397
Vanhooydonck B, Van Damme R (2003) Relationships between locomotor performance, microhabitat use and antipredator behaviour in lacertid lizards. Funct Ecol 17:160–169. https://doi.org/10.1046/j.1365-2435.2003.00716.x
Webb JK, Brown GP, Shine R (2001) Body size, locomotor speed and antipredator behaviour in a tropical snake (Tropidonophis mairii, Colubridae): the influence of incubation environments and genetic factors. Funct Ecol 15:561–568. https://doi.org/10.1046/j.0269-8463.2001.00570.x
Ydenberg RC, Dill LM (1986) The economics of fleeing from predators. Adv Stud Behav 16:229–249. https://doi.org/10.1016/S0065-3454(08)60192-8
Zuur A, Ieno E, Walker N, Saveliev A, Smith G (eds) (2009) Mixed effects models and extensions in ecology with R. Spring Science and Business Media, New York, USA
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Acknowledgements
We would like to thank the many individuals who assisted in the fieldwork: P. Bolton, T. Damasio, C. Fryns, G. Hughes, F. Kar, S. Klopper, L. Monk-Whipp, M. Mühlenhaupt, and D. Noble, as well as Taronga Zoo and the rest of the members of Macquarie University Lizard Lab. We would also like to thank two anonymous reviewers whose contributions improved this article.
Funding
This research was supported by Macquarie University (scholarship awarded to JB-G) and Natural Sciences and Engineering Research Council of Canada (scholarship awarded to JB-G).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Lizard capture and our experimental protocols followed animal ethics guidelines that were approved by both the Macquarie University Animal Ethics Committee (ARA no. 2015/023) and Taronga Zoo Animal Ethics Committee (ARA no. 3b/08/15). Our research was approved by the New South Wales National Parks and Wildlife Service, Office of Environment and Heritage (Licence no. SL100570).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by T. Madsen
Electronic supplementary material
ESM 1
(DOCX 1.87 mb)
Rights and permissions
About this article
Cite this article
Baxter-Gilbert, J., Riley, J.L. & Whiting, M.J. Runners and fighters: clutch effects and body size drive innate antipredator behaviour in hatchling lizards. Behav Ecol Sociobiol 72, 97 (2018). https://doi.org/10.1007/s00265-018-2505-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-018-2505-7