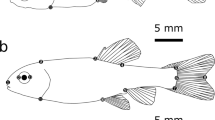
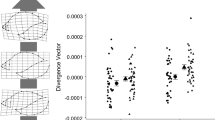
Abstract
Early life environments have important effects on phenotype development, but it can be difficult to disentangle the relative influences of genotype and environment on phenotypic variation within and among populations. Mangrove rivulus fish (Kryptolebias marmoratus) reproduce by self-fertilization and can generate isogenic lineages, which provides opportunities to resolve how the environment shapes the phenotype independent of genetic variation. Rivulus’ ecology is not well understood, but mangrove water snakes (Nerodia clarkii compressicauda) are thought to be a major predator. To test developmental responses to predator-related cues, four rivulus lineages (two that naturally co-exist with snakes; two that do not) were exposed to one of three treatments for 30 days post-hatching: cues from snakes that were fasted, fed rivulus, or fed heterospecifics. One week after exposure, fear and boldness responses were quantified. Individuals were photographed at 2 and 6 months of age for body size, growth, and body shape analysis. Animals that have historically encountered snakes were more risk averse and had wider heads than animals that historically have not encountered snakes. Rivulus exposed to cues from snakes fed conspecifics or heterospecifics grew faster than those exposed to fasted snake cues. Body shape was more streamlined in animals exposed to cues from snakes fed conspecifics, which may facilitate increased jumping performance as a way to escape aquatic predators. Our results suggest that rivulus exhibit phenotypic plasticity in response to cues associated with predator threat and that historical effects from selection or other evolutionary processes also are important determinants of behavioral and morphological variation.





Similar content being viewed by others
References
Adams DC, Otárola-Castillo E (2013) Geomorph: an R package for the collection and analysis of geometric morphometric shape data. Methods Ecol Evol 4:393–399
Ali M, Nicieza A, Wootton RJ (2003) Compensatory growth in fishes: a response to growth depression. Fish Fish 4:147–190
Ashley-Ross MA, Perlman BM, Gibb AC, Long JH Jr. (2014) Jumping sans legs: does elastic energy storage by the vertebral column power terrestrial jumps in bony fishes? Zoology 117:7–18
Auer SK, Arendt JD, Chandramouli R, Reznick DN (2010) Juvenile compensatory growth has negative consequences for reproduction in Trinidadian guppies (Poecilia reticulata). Ecol Lett 13:998–1007
Avise JC (2015) Evolutionary perspectives on clonal reproduction in vertebrate animals. Proc Natl Acad Sci USA 112:8867–8873
Berthé R, Lehmann FO (2015) Body appendages fine-tune posture and moments in freely manoeuvring fruit flies. J Exp Biol 218:3295–3307
Bookstein FL (1997) Morphometric tools for landmark data: geometry and biology. Cambridge University Press, Cambridge
Bookstein F, Schäfer K, Prossinger H, Seidler H, Fieder M, Stringer C, Weber GW, Arsuaga JL, Slice DE, Rohlf FJ (1999) Comparing frontal cranial profiles in archaic and modern Homo by morphometric analysis. Anat Rec 257:217–224
Bourdeau PE (2009) Prioritized phenotypic responses to combined predators in a marine snail. Ecology 90:1659–1669
Brown GE, Ferrari MC, Elvidge CK, Ramnarine I, Chivers DP (2013) Phenotypically plastic neophobia: a response to variable predation risk. Proc R Soc Lond B 280:20122712
Burrows M, Cullen DA, Dorosenko M, Sutton GP (2015) Mantises exchange angular momentum between three rotating body parts to jump precisely to targets. Curr Biol 25:786–789
Capellan E, Nicieza A (2007) Non-equivalence of growth arrest induced by predation risk or food limitation: context-dependent compensatory growth in anuran tadpoles. J Anim Ecol 76:1026–1035
Chivers DP, Smith RJF (1998) Chemical alarm signalling in aquatic predator–prey systems: a review and prospectus. Ecoscience 5:338–352
Chivers DP, Wisenden BD, Hindman CJ, Michalak TA, Kusch RC, Kaminskyj SG, Jack KL, Ferrari MC, Pollock RJ, Halbgewachs CF (2007) Epidermal ‘alarm substance’ cells of fishes maintained by non-alarm functions: possible defence against pathogens, parasites and UVB radiation. Proc R Soc Lond B 274:2611–2619
Costa WJ, Lima SM, Bartolette R (2010) Androdioecy in Kryptolebias killifish and the evolution of self-fertilizing hermaphroditism. Biol J Linn Soc 99:344–349
Davis WP, Taylor DS, Turner BJ (1990) Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyol Explor Freshw 1:123–134
Earley RL, Hanninen AF, Fuller A, Garcia MJ, Lee EA (2012) Phenotypic plasticity and integration in the mangrove rivulus (Kryptolebias marmoratus): a prospectus. Integr Comp Biol 52:814–827
Ferrari MC, Gonzalo A, Messier F, Chivers DP (2007) Generalization of learned predator recognition: an experimental test and framework for future studies. Proc R Soc Lond B 274:1853–1859
Ferrari MC, Wisenden BD, Chivers DP (2010) Chemical ecology of predator–prey interactions in aquatic ecosystems: a review and prospectus. Can J Zool 88:698–724
Frommen JG, Herder F, Engqvist L, Mehlis M, Bakker TCM, Schwarzer J, Thünken T (2011) Costly plastic morphological responses to predator specific odour cues in three-spined sticklebacks (Gasterosteus aculeatus). Evol Ecol 25:641–656
Fu J, MacCulloch RD, Murphy RW, Darevsky IS, Kupriyanova LA, Danielyan F (1998) The parthenogenetic rock lizard Lacerta unisexualis: an example of limited genetic polymorphism. J Mol Evol 46:127–130
Gibb AC, Ashley-Ross MA, Hsieh ST (2013) Thrash, flip, or jump: the behavioral and functional continuum of terrestrial locomotion in teleost fishes. Integr Comp Biol 53:295–306
Harrington RW Jr. (1967) Environmentally controlled induction of primary male gonochorists from eggs of the selffertilizing hermaphroditic fish, Rivulus marmoratus Poey. Biol Bull 132:174–199
Jansen KP, Mushinsky HR, Karl SA (2008) Population genetics of the mangrove salt marsh snake, Nerodia clarkii compressicauda, in a linear, fragmented habitat. Conserv Genet 9:401–410
Langerhans RB (2009) Trade-off between steady and unsteady swimming underlies predator-driven divergence in Gambusia affinis. J Evol Biol 22:1057–1075
Lugert V, Thaller G, Tetens J, Schulz C, Krieter J (2016) A review on fish growth calculation: multiple functions in fish production and their specific application. Rev Aquacult 8:30–42
Madison DM, Sullivan AM, Maerz JC, McDarby JH, Rohr JR (2002) A complex, cross-taxon, chemical releaser of antipredator behavior in amphibians. J Chem Ecol 28:2271–2282
Maher JM, Werner EE, Denver RJ (2013) Stress hormones mediate predator-induced phenotypic plasticity in amphibian tadpoles. Proc R Soc Lond B 280:20123075
Miller DE, Mushinsky HR (1990) Foraging ecology and prey size in the mangrove water snake, Nerodia fasciata compressicauda. Copeia 4:1099–1106
Mirza RS, Chivers DP (2001) Do juvenile yellow perch use diet cues to assess the level of threat posed by intraspecific predators? Behaviour 138:1249–1258
Mitteroecker P, Gunz P (2009) Advances in geometric morphometrics. Evol Biol 36:235–247
Moczek AP, Sultan S, Foster S, Ledón-Rettig C, Dworkin I, Nijhout HF, Abouheif E, Pfennig DW (2011) The role of developmental plasticity in evolutionary innovation. Proc R Soc Lond B 278:2705–2713
Parejko K, Dodson SI (1991) The evolutionary ecology of an antipredator reaction norm: Daphnia pulex and Chaoborus americanus. Evolution 45:1665–1674
Pfennig DW, Wund MA, Snell-Rood EC, Cruickshank T, Schlichting CD, Moczek AP (2010) Phenotypic plasticity’s impacts on diversification and speciation. Trends Ecol Evol 25:459–467
R Core Team (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. ISBN 3-900051-07-0
Relyea RA (2004) Fine-tuned phenotypes: tadpole plasticity under 16 combinations of predators and competitors. Ecology 85:172–179
Rohlf F (2010) tpsDig, version 2.16. Department of Ecology and Evolution, State University of New York, Stony Brook
Royle NJ, Lindström J, Metcalfe NB (2005) A poor start in life negatively affects dominance status in adulthood independent of body size in green swordtails Xiphophorus helleri. Proc R Soc Lond B 272:1917–1922
Royle NJ, Lindström J, Metcalfe NB (2006) Effect of growth compensation on subsequent physical fitness in green swordtails Xiphophorus helleri. Biol Lett 2:39–42
Scarsella GE, Gresham JD, Earley RL (2018) Relationships between external sexually dimorphic characteristics and internal gonadal morphology in a sex-changing fish. J Zool. https://doi.org/10.1111/jzo.12546 (in press)
Schoeppner NM, Relyea RA (2005) Damage, digestion, and defence: the roles of alarm cues and kairomones for inducing prey defences. Ecol Lett 8:505–512
Schoeppner NM, Relyea RA (2009) Interpreting the smells of predation: how alarm cues and kairomones induce different prey defences. Funct Ecol 23:1114–1121
Smee DL, Weissburg MJ (2006) Hard clams (Mercenaria mercenaria) evaluate predation risk using chemical signals from predators and injured conspecifics. J Chem Ecol 32:605–619
Smith RJF (1992) Alarm signals in fishes. Rev Fish Biol Fish 2:33–63
Stabell OB, Ogbebo F, Primicerio R (2003) Inducible defences in Daphnia depend on latent alarm signals from conspecific prey activated in predators. Chem Senses 28:141–153
Styga JM, Houslay TM, Wilson AJ, Earley RL (2018) Ontogeny of the morphology-performance axis in an amphibious fish (Kryptolebias marmoratus). J Exp Zool. https://doi.org/10.1002/jez.2150
Tatarenkov A, Lima SM, Taylor DS, Avise JC (2009) Long-term retention of self-fertilization in a fish clade. Proc Natl Acad Sci USA 106:14456–14459
Tatarenkov A, Earley RL, Taylor DS, Avise JC (2012) Microevolutionary distribution of isogenicity in a self-fertilizing fish (Kryptolebias marmoratus) in the Florida Keys. Integr Comp Biol 52:743–752
Tatarenkov A, Earley RL, Perlman BM, Taylor DS, Turner BJ, Avise JC (2015) Genetic subdivision and variation in selfing rates among central American populations of the mangrove rivulus, Kryptolebias marmoratus. J Hered 106:276–284
Taylor DS (2012) Twenty-four years in the mud: what have we learned about the natural history and ecology of the mangrove rivulus, Kryptolebias marmoratus? Integr Comp Biol 52:724–736
Turko AJ, Earley RL, Wright PA (2011) Behaviour drives morphology: voluntary emersion patterns shape gill structure in genetically identical individuals of the self-fertilizing mangrove rivulus, Kryptolebias marmoratus. Anim Behav 82:39–47
West-Eberhard MJ (2003) Developmental plasticity and evolution. Oxford University Press, Oxford
Wisenden BD, Pollock MS, Tremaine RJ, Webb JM, Wismer ME, Chivers DP (2003) Synergistic interactions between chemical alarm cues and the presence of conspecific and heterospecific fish shoals. Behav Ecol Sociobiol 54:485–490
Zelditch ML, Swiderski DL, Sheets HD (2004) Geometric morphometrics for biologists: a primer. Academic Press, London
Acknowledgements
This study was performed in accordance with the University of Alabama Institutional Animal Care and Use Committee (Protocol #’s 08-312 [13-10-0048] and [14-05-0071]). The authors would like to thank D. Scott Taylor, Yvonne Wielhouwer, Mark Garcia, Benjamin Perlman, and the staff at the Keys Marine Laboratory for assistance in field collections and logistics, Matthew Symonds for his patience during the revision process, and two reviewers for very insightful comments that significantly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
James, W.R., Styga, J.M., White, S. et al. Phenotypically plastic responses to predation threat in the mangrove rivulus fish (Kryptolebias marmoratus): behavior and morphology. Evol Ecol 32, 453–468 (2018). https://doi.org/10.1007/s10682-018-9952-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-018-9952-5