Abstract
Purpose
Radiotherapy is the main therapeutic approach besides surgery of localized prostate cancer. It relies on risk stratification and exact staging. This report analyses the potential of [68Ga]Glu-urea-Lys(Ahx)-HBED-CC (68Ga-PSMA-11), a new positron emission tomography (PET) tracer targeting prostate-specific membrane antigen (PSMA) for prostate cancer staging and individualized radiotherapy planning.
Methods
A cohort of 57 patients with prostate cancer scanned with 68Ga-PSMA-11 PET/CT for radiotherapy planning was retrospectively reviewed; 15 patients were at initial diagnosis and 42 patients at time of biochemical recurrence. Staging results of conventional imaging, including bone scintigraphy, CT or MRI, were compared with 68Ga-PSMA ligand PET/CT results and the influence on radiotherapeutic management was quantified.
Results
68Ga-PSMA ligand PET/CT had a dramatic impact on radiotherapy application in the presented cohort. In 50.8 % of the cases therapy was changed.
Conclusion
The presented imaging technique of 68Ga-PSMA PET/CT could be a key technology for individualized radiotherapy management in prostate cancer.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Radiotherapy plays an essential role in the curative treatment of prostate cancer. While moderate doses of radiotherapy with conventional techniques seem to result in slightly inferior results compared to radical surgery, modern technology has the potential to overcome this limitation [1].
New strategies in the management of localized prostate cancer incorporate more and more individualized approaches based on innovative imaging to enable dose-escalated treatment. Individualized approaches already start at the point of diagnostic workup, where multiparametric prostate MRI has shown the possibilities of identifying special regions at risk and guiding biopsy. A recent investigation presented high detection rates even when previous biopsies had been negative [2]. Overall such a targeted diagnostic procedure resulted in a higher percentage of cancer detection compared to classical serial biopsy [3]. Generally MRI-guided biopsies are most useful for improving the diagnosis of clinically significant prostate cancers and reducing the diagnosis of clinically insignificant cancers. Thus, overall there is usually only a minimal overall increase in all prostate cancer detection.
The importance of exact imaging and staging of course continues in the therapeutic decision-making to tailor the individual treatment of a patient. This goes beyond finding stage IV patients; furthermore, modern techniques in radiation oncology allow an individualized risk-adapted therapy with high dose application to tumour lesions in the prostate or in lymph nodes. Instead of treating the entire prostate homogeneously, selective boosting of areas bearing the most tumour cells has been proposed. These ideas go back to the late 1990s, when a selective dose escalation to intraprostatic tumour areas as detected in MRI was shown to be feasible without increasing the dose to organs at risk [4]. This specific targeting of the so-called dominant intraprostatic lesion has been shown to be dosimetrically feasible in confirmative publications with doses up to 200 % of the regular therapeutic doses [5, 6]. Using calculation models and tumour control probabilities, this method in theory increases the therapeutic ratio in selected patients [7]. Yet its efficacy in terms of outcome still has to be proven. In analogy to this intraprostatic approach, several groups have conducted trials to selectively increase the dose in lymph node metastases as opposed to giving a homogeneous dose to the entire pelvis.
All these individualized approaches rely on the sensitivity and the specificity of the underlying imaging modality. Since conventional CT offers poor detection and differentiation possibilities here, the quest for a more precise and reliable tool has a long history now. MRI approaches using ultrasmall particles of iron oxide that are trapped in lymph nodes were used and high sensitivity and specificity for detection of lymph node metastases from 65 to 75 % and 93 to 96 % were reported [8]. 11C-Choline positron emission tomography (PET) was analysed with regard to the potential of finding the site of relapse at biochemical relapse and showed a high detection rate in 78 % of their patients as demonstrated by Lépinoy et al. [9]. Similar findings were reported by other groups enabling a more individualized radiotherapy approach [10–12].
Intense overexpression of the cell surface peptidase prostate-specific membrane antigen (PSMA) in the majority of both primary and metastatic prostate cancers as well as a positive correlation of PSMA with traditional adverse prognostic factors makes this structure a promising target for molecular imaging [1, 2]. Using the novel 68Ga-labelled PSMA ligand Glu-urea-Lys(Ahx)-HBED-CC (68Ga-PSMA-11), which presents outstanding affinity to PSMA [13], a highly selective approach of imaging prostate cancer, lymph node metastases and distant metastases is available. This has been shown to be possible in hybrid PET/CT and PET/MRI [14, 15]. Early reports found a better contrast than with choline PET [16].
This study analyses the impact of PSMA-11 PET/CT on a cohort of prostate cancer patients treated with radiotherapy with respect to changes of staging and radiotherapeutic management [17, 18].
Materials and methods
Patients
A total of 57 patients (Table 1) with prostate cancer were retrospectively analysed with conventional CT and PSMA-11 PET/CT imaging; 15 patients presented at initial diagnosis, while 42 patients presented with recurrence after total prostatectomy. The median age was 70 years (range 53–83) with a median Gleason score of 8.0 (range 5–9) and a median prostate-specific antigen (PSA) level of 3.0 ng/ml (range 0.16–113). According to the D’Amico criteria 22 patients (38.6 %) had intermediate-risk and 31 patients (54.4 %) high-risk cancer [19]. Table 1 displays the characteristics of the examined patient cohort. It is possible that our cohort presents some overlap with a previous analysis about the diagnostic accuracy of PSMA PET [16].
Imaging
PSMA-11 PET/CT
PET and CT imaging were performed for every patient on a Biograph 6 PET/CT scanner (Siemens, Erlangen, Germany) 60 ± 10 min after intravenous injection of 68Ga-PSMA-11 (median 175 MBq, range 77–350). The target activity was 2–3 MBq/kg body weight. Due to the short half-life of 68Ga (68 min) only a wider range of injected activity than commonly seen with 18F tracers was achievable in the clinical workflow; however, the shorter half-life simultaneously limits radiation exposure. 68Ga-PSMA-11 was synthesized as previously described [13]. On conventional CT, lymph nodes were regarded as pathologically involved if their short axis diameter was ≥10 mm. Lymph node involvement in 68Ga-PSMA ligand PET was diagnosed when a node on CT demonstrated a maximum standardized uptake value (SUVmax) >2. This cut-off was chosen after evaluating the blood pool remainder at 60 min post-injection in the aorta, which was found to be a mean SUVmax of 1.49 ± standard deviation of 0.49. Lymph nodes were assessed as distant metastases if their localization was beyond the true pelvis. TNM classification was performed by two experienced readers in consensus. Lesions that were visually considered as suggestive of prostate cancer were counted and analysed in localization (local relapses, lymph node, soft tissue and bone metastases) and SUVmax.
CT/MRI/bone scan
Additional imaging modalities were chosen depending on the result of the preceding PSMA-11 PET/CT. If suspect bone lesions were found, a bone scan was done as a second modality to confirm “true-positive”. All patients with primary tumour inside the prostate additionally received multiparametric MRI, otherwise only if PSMA-11 PET/CT was suspicious for local recurrence. MRI was focused on the prostate bed [T2-weighted turbo spin echo (TSE) px 0.7 × 0.7 mm; repetition time (TR) 9.0; echo time (TE) 97; T1-weighted TSE px 0.54 ×0.54 mm; TR 1.38; TE 11 post CM], but contained at least one series covering the pelvis/abdomen. Dual-phase contrast-enhanced CT pelvis/abdomen was routinely performed for radiation planning. PSMA-11 PET/low-dose CT and contrast-enhanced CT were done within <3 weeks of each other.
Results
In 12 of 57 (21.1 %) patients at least 1 lesion characteristic for prostate cancer was detected in conventional CT. This included ten patients at the time of recurrence and two patients with initial diagnosis. According to the D’Amico criteria, two patients had low-risk, four intermediate-risk and six high-risk cancer. Using 68Ga PSMA ligand PET/CT, 85 lesions characteristic for prostate cancer were detected in 34 patients, 25 (73.5 %) patients with recurrence and 9 (26.5 %) at initial diagnosis. Of 34 patients, 21 (61.7 %) had high-risk, 9 (26.5 %) intermediate-risk and 4 (11.6 %) low-risk cancer. In 19 (32.8 %) patients no lesions were found with both methods. Amongst the 85 lesions characteristic for prostate cancer, 62 were defined as lymph node metastases, 9 as bone metastases, 8 as local relapses and 6 as primary tumours.
The TNM staging of 29 of 57 (50.8 %) patients was changed after PSMA-11 PET/CT imaging. This included 4 (13.7 %) patients with changes at initial diagnosis and 25 (86.3 %) patients at the time of recurrence. According to the D’Amico criteria, 3 (10.3 %) patients with change in TNM staging had low-risk, 11 (37.9 %) intermediate-risk and 15 (51.8 %) high-risk cancer. Of 29 patients, 15 (51.8 %) were changed from N0 to N1, 9 patients (31.0 %) were changed from M0 to M1a, 4 patients (13.8 %) were changed from M0 to M1b and 6 patients (20.7 %) were changed from Tx to T2a.
Within the group of high-risk patients, 15 of 31 (48.4 %) were changed in TNM staging. Of 16 patients, 10 had no findings in conventional CT and bone scan (Tx N0 M0). Four patients (26.7 %) were changed to N1 M0, three (20.0 %) to N0 M1a, two (13.3 %) to N1 M1a, two (13.3 %) to N1 M1b, two (13.3 %) to T2a N0 M0. One (6.7 %) patient was changed from N1 M0 to N1 M1a and one (7.1 %) from N1 M1a to N1 M1b.
Of 22 patients among the group of patients with intermediate-risk cancer, 11 (50.0 %) had a change in TNM staging after PSMA-11 PET/CT. This included 9 of 11 (81.8 %) patients with changes from N0 to N1, 3 patients from M0 to M1a (27.3 %) and 1 patient from Tx to T2 (9.1 %). Figure 1 illustrates the changes of TNM stage with respect to the different imaging modalities.
The reported changes in TNM stage resulted in the following alteration of radiotherapeutic management: based on PSMA-11 PET/CT 29/57 patients (50.8 %) received a different therapeutic approach compared to the initial plan based upon conventional staging. Of 29 patients, 18 (62.1 %) received an additional simultaneous integrated lymph node boost due to a change from N0 to N1, while the extent of pelvic lymphatic irradiation remained the same. Eight patients (27.5 %) were changed from M0 to M1a, which resulted in an enlarged lymphatic field irradiation with either para-aortic node region or inguinal node region plus an additional simultaneous integrated lymph node boost. Four patients (13.8 %) were changed from M0 to M1b and radiotherapy was cancelled and systemic therapy was given. Figures 2, 3 and 4 display cases at primary diagnosis and disease relapse in which PSMA PET/CT changed the radiotherapeutic regimen.
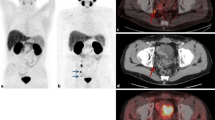
Example of the impact of PSMA imaging on radiotherapeutic management at initial diagnosis of intermediate-risk prostate cancer. a PSMA PET/CT with tracer uptake in a pararectal lymph node (SUVmax 3.1; arrow) which was not clearly pathological in conventional CT (b). Accordingly the irradiation plan was changed with coverage of perirectal space and a simultaneous boost to the lymph node (c: IMRT in 34 fractions with 51 Gy to lymphatic pathways, 76.5 Gy to the prostate and 61.2 Gy to the pathological lymph node). d IMRT plan prior to PSMA PET information without sufficient coverage of the pathological lymph node
Prostate cancer recurrence in the intermediate-risk situation. a, b PSMA PET/CT with a pathological presacral lymph node that was unclear in conventional CT (c). The corresponding IMRT plan is shown in d with 34 fractions and 51 Gy to the lymphatic pathways (arrow) and a simultaneous integrated boost to the lymph node to 61.2 Gy. For comparison e displays the IMRT plan without PET information
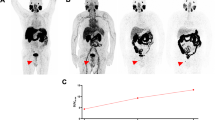
Example of PSA relapse 3 years after prostatectomy and prostate bed irradiation for high-risk prostate cancer. Only the PSMA PET/CT (a) reveals the iliac lymph node metastases with an SUVmax of 9.7, whereas the conventional CT (b) could show nothing. The corresponding IMRT plan is shown in c with 26 fractions to the pelvic lymphatic pathways to 46.8 Gy and a simultaneous integrated boost to the lymph node to a dose of 57.2 Gy. After prostate bed irradiation 3 years before radiotherapy would not have been possible without the PET information
Of the 16 PSMA-negative patients 5 had a PSA level <0.5, 8 had a PSA level between 0.51 < 1.0, 2 had a PSA level between 1.1 and < 2.0 and 1 patient had a PSA level of 21.7 and a Gleason score of 7.
Discussion
The presented analysis examines the value and impact of 68Ga-PSMA ligand PET/CT for the radiotherapeutic management of prostate cancer patients. We presented a cohort of 57 patients both in the setting of primary diagnosis and disease relapse. With a percentage of 38.6 % intermediate-risk and 54.4 % high-risk constellations, this was a selected cohort with high probability of metastatic disease—locoregional or distant. It is important to emphasize the characteristics of this patient group before interpretations of the results can be made. In addition, this work has inherent limitations due to its retrospective nature: the follow-up of this new approach of imaging and selective treatment is too short to report outcome; due to the short follow-up, additionally applied androgen deprivation, which was given to all lymph node-positive patients for at least 2 years; and the lack of a control group.
Today, exact data on sensitivity and specificity of PSMA-11 PET/CT are still limited. However, PSMA-11 PET correlated well with the histological findings in succeeding surgery in 42 patients that have been recently published [24] and is more sensitive than CT based 3D volumetric lymph node evaluation in determining the node status of patients with recurrent prostate cancer [25]. A total of 319 patients have been evaluated with follow-up as standard of reference and sensitivity/specificity was reported with 76.6/100 %; the negative predictive value (NPV) and positive predictive value (PPV) were 91.4 and 100 % [24]. Especially the high PPV provides a good rationale to administer local radiation boosts to PET-positive lymph nodes. Our report clearly demonstrates the potential role of this method that can enable selective and custom-tailored radiotherapy for patients with prostate cancer. Today, radiotherapy techniques can perform these selective dose escalations of tumour-bearing parts of the prostate or metastatic lymph nodes [5, 6]. With the development of intensity-modulated radiotherapy (IMRT), new possibilities have been opened up for the treatment of prostate cancer. Using a simultaneous integrated boost (SIB) concept, many centres treat the whole pelvis in high-risk patients and apply a SIB to the prostate or prostate bed [20]. Some groups have taken this concept further and have used the SIB technique for dose escalation to macroscopic lymph node metastases: Fonteyne et al. [21] reported on 80 patients with T1–4 N1 M0 prostate cancer who received androgen deprivation for 2–3 years and whole-pelvis radiotherapy (45 Gy) and a SIB to the prostate (69.3 Gy), to the intraprostatic lesion (72 Gy) and to enlarged pelvic lymph nodes (65 Gy) in 25 fractions. Radiotherapy was tolerated well with 3-year rates of late gastrointestinal toxicity of 8 % (grade 3) and 20 % (grade 3) and late genitourinary toxicity of 6 % (grade 3–4) and 34 % (grade 2). Actuarial 3-year biochemical recurrence-free survival was 81 %. Engels et al. [22] treated 28 high-risk patients with whole-pelvis radiotherapy (54 Gy) with a SIB to the prostate (70.5 Gy) in 30 fractions. Among these, macroscopic lymph node metastases received a SIB (60 Gy) in eight patients. Acute toxicity rates were low, and no difference in toxicity was seen between patients with and without lymph node boost.
An Italian publication [23] described high-dose hypofractionated IMRT treatment with SIB to pathological nodes in 83 patients with a lymph node recurrence after primary treatment which was detected by 11C-choline PET/CT. The treatment was tolerated well with only patients experiencing acute grade 3 genitourinary side effects, and in 83 % of patients, a complete or partial biochemical response was achieved [24].
The results of these approaches still have to be shown with longer follow-up in controlled trials. Yet, the toxicity of such individualized approaches reported so far is very promising [26]. The key to this method, however, is a reliable, sensitive and specific diagnostic tool.
In comparison to other diagnostic options, our results compare favourably with those reported for 11C-choline PET in a similar setting. Jereczek-Fossa reported a ratio of 22 of 74 cases when 11C-choline PET/CT altered the treatment approach compared with the treatment that would have been adopted in the absence of 11C-choline PET/CT analysis [11]. However, a new technology can also introduce now pitfalls. In regard to PSMA PET, physiological uptake in coeliac ganglia which could be mistaken as M1a should carefully be considered [27].
Geiger et al. reported that 51 % of patients with non-small cell lung cancer (NSCLC) had new findings in 18F-fluorodeoxyglucose (FDG) PET/CT [28]. In a prospective study incorporating 18F-FDG PET/CT for stage III NSCLC a probability of 54 % of changes of target volumes was reported [29]. For these reasons this PET/CT method is in routine use for NSCLC patients. Our results suggest a similar impact of PSMA-11 PET/CT staging for prostate cancer patients compared to 18F-FDG PET/CT at least in this selected risk group. If these findings can be reproduced in a prospective setting, the method has the potential to become a routine tool in the staging and therapy planning for prostate cancer patients.
Among the PSMA PET-negative patients were many patients with a PSA <1 ng/ml. This underlines that for particular patients with a low level of PSA the likelihood of detecting a PSMA-positive lesion is smaller. However, even with a PSA below 1 ng/ml the rate of positive PSMA PET was described to be around 50 % [24]. Furthermore, it has to be considered that PET resolution also has its physical limitation and therefore might also bring a further limitation to lesion detection even in a patient with PSMA-positive expression.
Conclusion
PSMA-11 PET/CT had a significant impact on the management of the presented cohort of radiotherapy patients with prostate cancer. Radiotherapeutic management was changed in 50.8 %. The treatment of lymph node areas in the pelvis has already become more individualized by the use of IMRT. Radiotherapy today delivers selective pelvic lymphatic irradiation rather than standardized whole-pelvis irradiation. More sensitive and specific staging helps to exclude stage IV patients from this approach and enables individualized dose escalation to lymph node metastases or subvolumes of the prostate. The consequences in terms of changes for progression-free survival and overall survival remain to be investigated.
References
Sooriakumaran P, Nyberg T, Akre O, Haendler L, Heus I, Olsson M, et al. Comparative effectiveness of radical prostatectomy and radiotherapy in prostate cancer: observational study of mortality outcomes. BMJ 2014;348:g1502.
Roethke M, Anastasiadis AG, Lichy M, Werner M, Wagner P, Kruck S, et al. MRI-guided prostate biopsy detects clinically significant cancer: analysis of a cohort of 100 patients after previous negative TRUS biopsy. World J Urol 2012;30(2):213–8.
Kuru TH, Roethke MC, Seidenader J, Simpfendörfer T, Boxler S, Alammar K, et al. Critical evaluation of magnetic resonance imaging targeted, transrectal ultrasound guided transperineal fusion biopsy for detection of prostate cancer. J Urol 2013;190(4):1380–6.
Pickett B, Vigneault E, Kurhanewicz J, Verhey L, Roach M. Static field intensity modulation to treat a dominant intra-prostatic lesion to 90 Gy compared to seven field 3-dimensional radiotherapy. Int J Radiat Oncol Biol Phys 1999;44(4):921–9.
Housri N, Ning H, Ondos J, Choyke P, Camphausen K, Citrin D, et al. Parameters favorable to intraprostatic radiation dose escalation in men with localized prostate cancer. Int J Radiat Oncol Biol Phys 2011;80(2):614–20.
Fellin F, Azzeroni R, Maggio A, Lorentini S, Cozzarini C, Di Muzio N, et al. Helical tomotherapy and intensity modulated proton therapy in the treatment of dominant intraprostatic lesion: a treatment planning comparison. Radiother Oncol 2013;107(2):207–12.
Riches SF, Payne GS, Desouza NM, Dearnaley D, Morgan VA, Morgan SC, et al. Effect on therapeutic ratio of planning a boosted radiotherapy dose to the dominant intraprostatic tumour lesion within the prostate based on multifunctional MR parameters. Br J Radiol 2014;87(1037):20130813.
Birkhäuser FD, Studer UE, Froehlich JM, Triantafyllou M, Bains LJ, Petralia G, et al. Combined ultrasmall superparamagnetic particles of iron oxide-enhanced and diffusion-weighted magnetic resonance imaging facilitates detection of metastases in normal-sized pelvic lymph nodes of patients with bladder and prostate cancer. Eur Urol 2013;64(6):953–60.
Lépinoy A, Cochet A, Cueff A, Cormier L, Martin E, Maingon P, et al. Pattern of occult nodal relapse diagnosed with (18)F-fluoro-choline PET/CT in prostate cancer patients with biochemical failure after prostate-only radiotherapy. Radiother Oncol 2014;111:120–5.
Alongi F, Comito T, Villa E, Lopci E, Cristina I, Mancosu P, et al. What is the role of [11C]choline PET/CT in decision making strategy before post-operative salvage radiation therapy in prostate cancer patients? Acta Oncol 2014;53:990–2.
Jereczek-Fossa BA, Rodari M, Bonora M, Fanti P, Fodor C, Pepe G, et al. [(11)C]Choline PET/CT impacts treatment decision making in patients with prostate cancer referred for radiotherapy. Clin Genitourin Cancer 2014;12(3):155–9.
Schwarzenböck SM, Kurth J, Gocke C, Kuhnt T, Hildebrandt G, Krause BJ. Role of choline PET/CT in guiding target volume delineation for irradiation of prostate cancer. Eur J Nucl Med Mol Imaging 2013;40 Suppl 1:S28–35.
Eder M, Schäfer M, Bauder-Wüst U, Hull WE, Wängler C, Mier W, et al. 68Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem 2012;23(4):688–97.
Roethke MC, Kuru TH, Afshar-Oromieh A, Schlemmer HP, Hadaschik BA, Fenchel M. Hybrid positron emission tomography-magnetic resonance imaging with gallium 68 prostate-specific membrane antigen tracer: a next step for imaging of recurrent prostate cancer-preliminary results. Eur Urol 2013;64(5):862–4.
Afshar-Oromieh A, Malcher A, Eder M, Eisenhut M, Linhart HG, Hadaschik BA, et al. PET imaging with a [68Ga]gallium-labelled PSMA ligand for the diagnosis of prostate cancer: biodistribution in humans and first evaluation of tumour lesions. Eur J Nucl Med Mol Imaging 2013;40(4):486–95.
Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, et al. Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging 2014;41(1):11–20.
Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res 1997;3:81–5.
Ross JS, Sheehan CE, Fisher HA, Kaufmann RP, Kaur P, Gray K, et al. Correlation of primary tumor prostate-specific membrane antigen expression with disease recurrence in prostate cancer. Clin Cancer Res 2003;9:6357–62.
D’Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 1998;280(11):969–74.
Alongi F, Fiorino C, Cozzarini C, Broggi S, Perna L, Cattaneo GM, et al. IMRT significantly reduces acute toxicity of whole-pelvis irradiation in patients treated with post-operative adjuvant or salvage radiotherapy after radical prostatectomy. Radiother Oncol 2009;93(2):207–12.
Fonteyne V, Lumen N, Ost P, Van Praet C, Vandecasteele K, De GersemIr W, et al. Hypofractionated intensity-modulated arc therapy for lymph node metastasized prostate cancer: early late toxicity and 3-year clinical outcome. Radiother Oncol 2013;109(2):229–34.
Engels B, Soete G, Tournel K, Bral S, De Coninck P, Verellen D, et al. Helical tomotherapy with simultaneous integrated boost for high-risk and lymph node-positive prostate cancer: early report on acute and late toxicity. Technol Cancer Res Treat 2009;8(5):353–9.
Picchio M, Berardi G, Fodor A, Busnardo E, Crivellaro C, Giovacchini G, et al. (11)C-Choline PET/CT as a guide to radiation treatment planning of lymph-node relapses in prostate cancer patients. Eur J Nucl Med Mol Imaging 2014;41:1270–9.
Afshar-Oromieh A, Avtzi E, Giesel FL, Holland-Letz T, Linhart HG, Eder M, et al. The diagnostic value of PET/CT imaging with the (68)Ga-labelled PSMA ligand HBED-CC in the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging 2015;42(2):197–209. doi:10.1007/s00259-014-2949-6.
Giesel FL, Fiedler H, Stefanova M, Sterzing F, Rius M, Kopka K et al. PSMA PET/CT with Glu-urea-Lys-(Ahx)-[68Ga(HBED-CC)] versus 3D CT volumetric lymph node assessment in recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2015. doi:10.1007/s00259-015-3106-6
Katayama S, Habl G, Kessel K, Edler L, Debus J, Herfarth K, et al. Helical intensity-modulated radiotherapy of the pelvic lymph nodes with integrated boost to the prostate bed—initial results of the PLATIN 3 Trial. BMC Cancer 2014;14:20.
Krohn T, Verburg FA, Pufe T, Neuhuber W, Vogg A, Heinzel A, et al. [(68)Ga]PSMA-HBED uptake mimicking lymph node metastasis in coeliac ganglia: an important pitfall in clinical practice. Eur J Nucl Med Mol Imaging 2015;42(2):210–4.
Geiger GA, Kim MB, Xanthopoulos EP, Pryma DA, Grover S, Plastaras JP, et al. Stage migration in planning PET/CT scans in patients due to receive radiotherapy for non-small-cell lung cancer. Clin Lung Cancer 2014;15(1):79–85.29.
Kolodziejczyk M, Kepka L, Dziuk M, Zawadzka A, Szalus N, Gizewska A, et al. Impact of [18F]fluorodeoxyglucose PET-CT staging on treatment planning in radiotherapy incorporating elective nodal irradiation for non-small-cell lung cancer: a prospective study. Int J Radiat Oncol Biol Phys 2011;80:1008–14.
Compliance with ethical standards
Conflicts of interest
None
Funding
This research was supported by the Klaus-Tschira-Stiftung (project no. 00.198.2012).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sterzing, F., Kratochwil, C., Fiedler, H. et al. 68Ga-PSMA-11 PET/CT: a new technique with high potential for the radiotherapeutic management of prostate cancer patients. Eur J Nucl Med Mol Imaging 43, 34–41 (2016). https://doi.org/10.1007/s00259-015-3188-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-015-3188-1