Abstract
Purpose
To evaluate FDG-PET for staging, grading, preoperative response assessment and posttherapeutic evaluation in children with Wilms tumour (WT).
Methods
In this study, 23 FDG-PET examinations in 12 paediatric patients (female, n = 5; male, n = 7; age, 1–19 years) with WT (primary, n = 9; relapsed, n = 3) were analysed. All patients were examined with conventional imaging methods (CIM) according to the SIOP2001/GPOH trial protocol. Additionally, FDG-PET/PET-CT was performed for staging (n = 12), preoperative response assessment (n = 6) and posttherapeutic evaluation (n = 5). Imaging results of FDG-PET and CIM were analysed regarding the accuracy in tumour visualisation, impact on therapeutic management and preoperative response assessment, with clinical follow-up and histopathology as the standard of reference.
Results
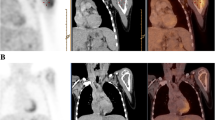
FDG-PET and CIM showed concordant results for staging of primary WT, whereas FDG-PET was superior in 1/3 cases with recurrent WT. Concerning histological differentiation, one case with anaplastic WT had an standard uptake value (SUV) of 12.3, which was remarkably higher than the average SUV in the eight cases with intermediate risk histology. No parameter analysed for PET or CIM was reliably predictive for histological regression or clinical outcome. After completion of therapy, FDG-PET was superior to CIM in 2/5 cases in detecting residual disease with therapeutic relevance.
Conclusion
FDG-PET does not provide additional information to the traditional imaging work-up for staging WT patients, preoperative response assessment and clinical outcome. FDG-PET was advantageous in ruling out residual disease after completion of first line treatment and in pretherapeutic staging of relapse patients. Furthermore, there seems to be a good correlation of initial SUV and histological differentiation.



Similar content being viewed by others
References
Lemerle J, Voûte PA, Tournade MF, Delemarre JF, Jereb B, Ahstrohm L, et al. Preoperative versus postoperative radiotherapy, single versus multiple courses of Actinomycin D, in the treatment of Wilms tumor. Preliminary results of a controlled clinical trial conducted by the International Society of Pediatric Oncology (SIOP). Cancer 1976;38:647–54.
Weirich A, Ludwig R, Graf N, Abel U, Leuschner I, Vujanic GM, et al. Survival in nephroblastoma treated according to the trial and study SIOP-9/GPOH with respect to relapse and morbidity. Ann Oncol 2004;15:808–20.
De Kraker J, Lemerle J, Voute PA, Zucker JM, Tournade MF, Carli M, et al. Wilm’s tumor with pulmonary metastases at diagnosis: the significance of primary chemotherapy. International Society of Pediatric Oncology Nephroblastoma Trial and Study Committee. J Clin Oncol 1990;8:1187–90.
Reinhard H, Semler O, Burger D, Bode U, Flentje M, Göbel U, et al. Results of the SIOP 93-01/GPOH trial and study for the treatment of patients with unilateral nonmetastatic Wilms Tumor. Klin Padiatr 2004;216:132–40.
Weirich A, Leuschner I, Harms D, Vujanic GM, Tröger J, Abel U, et al. Clinical impact of histologic subtypes in localized non-anaplastic nephroblastoma treated according to the trial and study SIOP 9/GPOH. Ann Oncol 2001;12:311–9.
Dome JS, Liu T, Krasin M, Lott L, Shearer P, Daw NC, et al. Improved survival for patients with recurrent Wilms tumor: the experience at St. Jude Children’s Research Hospital. J Pediatr Hematol Oncol 2002;24:192–8.
Metzger ML, Dome JS. Current therapy for Wilms’ tumor. Oncologist 2007;10:815–26.
Mitchell C, Pritchard-Jones K, Shannon R, Hutton C, Stevens S, Machin D, et al. For the United Kingdom Cancer Study Group. Immediate nephrectomy versus preoperative chemotherapy in the management of non-metastatic Wilms’ tumour: results of a randomised trial (UKW3) by the UK Children’s Cancer Study Group. Eur J Cancer 2006;42:2554–62.
Boccon-Gibod L, Rey A, Sandstedt B, Delemarre J, Harms D, Vujanic G, et al. Complete necrosis induced by preoperative chemotherapy in Wilms tumor as an indicator of low risk: report of the international society of paediatric oncology (SIOP) nephroblastoma trial and study 9. Med Pediatr Oncol 2000;34:183–90.
De Kraker J, Graf N, van Tinteren H, Pein F, Sandstedt B, Godzinski M, et al. Reduction of postoperative chemotherapy in children with stage I intermediate-risk and anaplastic Wilms’ tumour (SIOP 93-01 trial): a randomised controlled trial. Lancet 2004;364:1229–35.
Ora I, van Tinteren H, Bergeron C, de Kraker J. SIOP Nephroblastoma Study Committee. Progression of localised Wilms’ tumour during preoperative chemotherapy is an independent prognostic factor: a report from the SIOP 93-01 nephroblastoma trial and study. Eur J Cancer 2007;43:131–6.
Grundy P, Breslow N, Green DM, Sharples K, Evans A, D’Angio GJ. Prognostic factors for children with recurrent Wilms’ tumor: results from the Second and Third National Wilms’ Tumor Study. J Clin Oncol 1989;7:638–47.
Miser JS, Tournade MF. The management of relapsed Wilms tumor. Hematol Oncol Clin North Am 1995;9:1287–302.
Carrico CW, Cohen MD, Zerin JM, Weetman R. Wilms tumor imaging: patient costs and protocol compliance. Radiology 1997;204:627–33.
Amthauer H, Furth C, Denecke T, Hundsdoerfer P, Voelker T, Seeger K, et al. FDG-PET in 10 children with non-Hodgkin’s lymphoma: initial experience in staging and follow-up. Klin Padiatr 2005;217:327–33.
Furth C, Amthauer H, Denecke T, Ruf J, Henze G, Gutberlet M. Impact of whole-body MRI and FDG-PET on staging and assessment of therapy response in a patient with Ewing sarcoma. Pediatr Blood Cancer 2006;47:607–11.
Furth C, Denecke T, Steffen I, Ruf J, Voelker T, Misch D, et al. Correlative imaging strategies implementing CT, MRI, and PET for staging of childhood Hodgkin disease. J Pediatr Hematol Oncol 2006;28:501–12.
Franzius C, Sciuk J, Brinkschmidt C, Jürgens H, Schober O. Evaluation of chemotherapy response in primary bone tumors with F-18 FDG positron emission tomography compared with histologically assessed tumor necrosis. Clin Nucl Med 2000;25:874–81.
Franzius C, Sciuk J, Daldrup-Link HE, Jürgens H, Schober O. FDG-PET for detection of osseous metastases from malignant primary bone tumours: comparison with bone scintigraphy. Eur J Nucl Med 2000;27:1305–11.
Györke T, Zajic T, Lange A, Schäfer O, Moser E, Mako E, et al. Impact of FDG PET for staging of Ewing sarcomas and primitive neuroectodermal tumours. Nucl Med Commun 2006;27:17–24.
Korholz D, Kluge R, Wickmann L, Hirsch W, Lüders H, Lotz I, et al. Importance of F18-fluorodeoxy-d-2-glucose positron emission tomography (FDG-PET) for staging and therapy control of Hodgkin’s lymphoma in childhood and adolescence—consequences for the GPOH-HD 2003 protocol. Onkologie 2003;26:489–93.
Völker T, Denecke T, Steffen I, Misch D, Schönberger S, Plotkin M, et al. Positron emission tomography for staging of pediatric sarcoma patients: results of a prospective multicenter trial. J Clin Oncol 2007;25:5435–41.
Hawkins DS, Rajendran JG, Conrad EU, Bruckner JD, Eary JF. Evaluation of chemotherapy response in pediatric bone sarcomas by [F-18]-fluorodeoxy-d-glucose positron emission tomography. Cancer 2002;94:3277–84.
Hawkins DS, Schuetze SM, Butrynski JE, Rajendran JG, Vernon CB, Conrad EU, et al. [18F]Fluorodeoxyglucose positron emission tomography predicts outcome for Ewing sarcoma family of tumors. J Clin Oncol 2005;23:8828–34.
Hernandez-Pampaloni M, Takalkar A, Yu JQ, Zhuang H, Alavi A. F-18 FDG-PET imaging and correlation with CT in staging and follow-up of pediatric lymphomas. Pediatr Radiol 2006;36:524–31.
Johnson GR, Zhuang H, Khan J, Chiang SB, Alavi A. Roles of positron emission tomography with fluorine-18-deoxyglucose in the detection of local recurrent and distant metastatic sarcoma. Clin Nucl Med 2003;28:815–20.
Folpe AL, Lyles RH, Sprouse JT, Conrad EU, Eary JF. (F-18) fluorodeoxyglucose positron emission tomography as a predictor of pathologic grade and other prognostic variables in bone and soft tissue sarcoma. Clin Cancer Res 2000;6:1279–87.
Lucas JD, O’Doherty MJ, Cronin BF, Marsden PK, Lodge MA, McKee PH, et al. Prospective evaluation of soft tissue masses and sarcomas using fluorodeoxyglucose positron emission tomography. Br J Surg 1999;86:550–6.
Schulte M, Brecht-Krauss D, Heymer B, Guhlmann A, Hartwig E, Sarkar MR, et al. Grading of tumors and tumorlike lesions of bone: evaluation by FDG PET. J Nucl Med 2000;41:1695–701.
Shulkin BL, Chang E, Strouse PJ, Bloom DA, Hutchinson RJ. PET FDG studies of Wilms tumors. J Pediatr Hematol Oncol 1997;19:334–8.
Franzius C, Daldrup-Link HE, Sciuk J, Rummeny EJ, Bielack S, Jürgens H, et al. FDG-PET for detection of pulmonary metastases from malignant primary bone tumors: comparison with spiral CT. Ann Oncol 2001;12:479–86.
Acknowledgement
This study was financially supported by the Deutsche Krebshilfe e.V.
Conflict of interest statement
All authors disclose no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Misch, D., Steffen, I.G., Schönberger, S. et al. Use of positron emission tomography for staging, preoperative response assessment and posttherapeutic evaluation in children with Wilms tumour. Eur J Nucl Med Mol Imaging 35, 1642–1650 (2008). https://doi.org/10.1007/s00259-008-0819-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-008-0819-9