Abstract
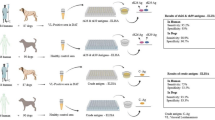
An accurate diagnosis of visceral leishmaniasis is an essential tool for control of the disease. While serologic methods are very useful, these conventional methodologies still present limitations in terms of sensitivity and specificity. The use of flow cytometry is a worldwide trend in the development of high-performance diagnostic methods. Herein, we describe a new flow cytometry serology test, characterized by the employment of the Cytometric Bead Array microspheres A4 and E4 coated with the recombinant antigens rLci1A and rLci2B respectively, to improve the serodiagnosis of canine visceral leishmaniasis. The tests were conducted in a wide variety of sera groups (n = 140), where the diagnostics development would be optimized accounting not just the ability to identify infected dogs with different clinical status, but also to exclude cross-reaction and differentiate vaccinated dogs from dogs infected. Serological testing of the antigenic system A4–rLci1A showed a sensitivity of 90.0% and specificity of 75%, while the E4–rLci2B testing demonstrated a sensitivity of 95.0% and specificity of 82.5%. The use of a multiplex assay of A4–rLci1A and E4–rLci2B, resulted in a diagnostic improvement, with a sensitivity of 95.0% and specificity of 91.2%. Our results show that this novel flow cytometry serology test is a viable tool for sensitive and specific serodiagnosis. Notably, the combination of distinct antigenic systems allows us to test for antibodies to multiple recombinant antigens from a single serum sample. This benefit emphasizes the importance of this methodology as an alternative in the serological diagnosis.





Similar content being viewed by others
References
Alvar J, Velez ID, Bern C, Herrero M, Desjeux P, Cano J, Jannin J, den Boer M, Team WHOLC (2012) Leishmaniasis worldwide and global estimates of its incidence. PLoS One 7(5):e35671. https://doi.org/10.1371/journal.pone.0035671
Andrade RA, Silva Araujo MS, Reis AB, Gontijo CM, Vianna LR, Mayrink W, Martins-Filho OA (2009) Advances in flow cytometric serology for canine visceral leishmaniasis: diagnostic applications when distinct clinical forms, vaccination and other canine pathogens become a challenge. Vet Immunol Immunopathol 128(1-3):79–86. https://doi.org/10.1016/j.vetimm.2008.10.308
Badaro R, Eulalio MC, Benson D, Freire M, Miranda JC, Pedral-Sampaio D, Burns JM, David JR Jr, Johnson WD, Reed SG (1993) Sensitivity and specificity of a recombinant Leishmania chagasi antigen in the serodiagnosis of visceral leishmaniasis. Arch Inst Pasteur Tunis 70(3-4):331–332
Campos FMF, Repoles LC, de Araujo FF, Peruhype-Magalhaes V, Xavier MAP, Sabino EC, de Freitas Carneiro Proietti AB, Andrade MC, Teixeira-Carvalho A, Martins-Filho OA, Gontijo CMF (2018) Usefulness of FC-TRIPLEX Chagas/Leish IgG1 as confirmatory assay for non-negative results in blood bank screening of Chagas disease. J Immunol Methods 455:34–40. https://doi.org/10.1016/j.jim.2018.01.006
Carvalho Neta AV, Rocha RDR, Gontijo CMF, Reis AB, Martins-Filho OA (2006) Citometria de fluxo no diagnóstico da leishmaniose visceral canina. Arquivo Brasileiro de Medicina Veterinária e Zootecnia 58:480–488
Costa CH (2011) How effective is dog culling in controlling zoonotic visceral leishmaniasis? A critical evaluation of the science, politics and ethics behind this public health policy. Rev Soc Bras Med Trop 44(2):232–242
Coura-Vital W, Marques MJ, Veloso VM, Roatt BM, Aguiar-Soares RD, Reis LE, Braga SL, Morais MH, Reis AB, Carneiro M (2011) Prevalence and factors associated with Leishmania infantum infection of dogs from an urban area of Brazil as identified by molecular methods. PLoS Negl Trop Dis 5(8):e1291. https://doi.org/10.1371/journal.pntd.0001291
Courtenay O, Quinnell RJ, Garcez LM, Shaw JJ, Dye C (2002) Infectiousness in a cohort of brazilian dogs: why culling fails to control visceral leishmaniasis in areas of high transmission. J Infect Dis 186(9):1314–1320. https://doi.org/10.1086/344312
da Silva DA, Madeira Mde F, Abrantes TR, Filho CJ, Figueiredo FB (2013) Assessment of serological tests for the diagnosis of canine visceral leishmaniasis. Vet J 195(2):252–253. https://doi.org/10.1016/j.tvjl.2012.06.010
Dantas-Torres F, Solano-Gallego L, Baneth G, Ribeiro VM, de Paiva-Cavalcanti M, Otranto D (2012) Canine leishmaniosis in the Old and New Worlds: unveiled similarities and differences. Trends Parasitol 28(12):531–538. https://doi.org/10.1016/j.pt.2012.08.007
de Andrade HM, Reis AB, dos Santos SL, Volpini AC, Marques MJ, Romanha AJ (2006) Use of PCR-RFLP to identify Leishmania species in naturally-infected dogs. Vet Parasitol 140(3-4):231–238. https://doi.org/10.1016/j.vetpar.2006.03.031
de Andrade RA, Reis AB, Gontijo CM, Braga LB, Rocha RD, Araujo MS, Vianna LR, Martins-Filho OA (2007) Clinical value of anti-Leishmania (Leishmania) chagasi IgG titers detected by flow cytometry to distinguish infected from vaccinated dogs. Vet Immunol Immunopathol 116(1-2):85–97. https://doi.org/10.1016/j.vetimm.2007.01.002
de Carvalho FLN, Riboldi EO, Bello GL, Ramos RR, Barcellos RB, Gehlen M, Halon ML, Romao PRT, Dallegrave E, Rossetti MLR (2018) Canine visceral leishmaniasis diagnosis: a comparative performance of serological and molecular tests in symptomatic and asymptomatic dogs. Epidemiol Infect 146(5):571–576. https://doi.org/10.1017/S0950268818000225
de Oliveira IQ, Silva RA, Sucupira MV, da Silva ED, Reis AB, Grimaldi G Jr, Fraga DB, Veras PS (2015) Multi-antigen print immunoassay (MAPIA)-based evaluation of novel recombinant Leishmania infantum antigens for the serodiagnosis of canine visceral leishmaniasis. Parasit Vectors 8:45. https://doi.org/10.1186/s13071-015-0651-6
de Paiva-Cavalcanti M, de Morais RC, Pessoa ESR, Trajano-Silva LA, Goncalves-de-Albuquerque Sda C, Tavares Dde H, Brelaz-de-Castro MC, Silva Rde F, Pereira VR (2015) Leishmaniases diagnosis: an update on the use of immunological and molecular tools. Cell & bioscience 5:31. https://doi.org/10.1186/s13578-015-0021-2
Faria AR, de Castro VL, Coura-Vital W, Reis AB, Damasceno LM, Gazzinelli RT, Andrade HM (2015) Novel recombinant multiepitope proteins for the diagnosis of asymptomatic Leishmania infantum-infected dogs. PLoS Negl Trop Dis 9(1):e3429. https://doi.org/10.1371/journal.pntd.0003429
Ferreira C, de Lana M, Carneiro M, Reis AB, Paes DV, da Silva ES, Schallig H, Gontijo CM (2007) Comparison of serological assays for the diagnosis of canine visceral leishmaniasis in animals presenting different clinical manifestations. Vet Parasitol 146(3-4):235–241. https://doi.org/10.1016/j.vetpar.2007.02.015
Folgueira C, Carrion J, Moreno J, Saugar JM, Canavate C, Requena JM (2008) Effects of the disruption of the HSP70-II gene on the growth, morphology, and virulence of Leishmania infantum promastigotes. Int Microbiol 11(2):81–89
Fraga DB, da Silva ED, Pacheco LV, Borja LS, de Oliveira IQ, Coura-Vital W, Monteiro GR, Oliveira GG, Jeronimo SM, Reis AB, Veras PS (2014) A multicentric evaluation of the recombinant Leishmania infantum antigen-based immunochromatographic assay for the serodiagnosis of canine visceral leishmaniasis. Parasit Vectors 7:136. https://doi.org/10.1186/1756-3305-7-136
Giunchetti RC, Mayrink W, Genaro O, Carneiro CM, Correa-Oliveira R, Martins-Filho OA, Marques MJ, Tafuri WL, Reis AB (2006) Relationship between canine visceral leishmaniosis and the Leishmania (Leishmania) chagasi burden in dermal inflammatory foci. J Comp Pathol 135(2-3):100–107. https://doi.org/10.1016/j.jcpa.2006.06.005
Giunchetti RC, Correa-Oliveira R, Martins-Filho OA, Teixeira-Carvalho A, Roatt BM, de Oliveira Aguiar-Soares RD, de Souza JV, das Dores Moreira N, Malaquias LC, Mota e Castro LL, de Lana M, Reis AB (2007) Immunogenicity of a killed Leishmania vaccine with saponin adjuvant in dogs. Vaccine 25(44):7674–7686. https://doi.org/10.1016/j.vaccine.2007.08.009
Grimaldi G Jr, Teva A, Ferreira AL, dos Santos CB, Pinto I, de-Azevedo CT, Falqueto A (2012) Evaluation of a novel chromatographic immunoassay based on Dual-Path Platform technology (DPP(R) CVL rapid test) for the serodiagnosis of canine visceral leishmaniasis. Trans R Soc Trop Med Hyg 106(1):54–59. https://doi.org/10.1016/j.trstmh.2011.10.001
Harhay MO, Olliaro PL, Costa DL, Costa CH (2011) Urban parasitology: visceral leishmaniasis in Brazil. Trends Parasitol 27(9):403–409. https://doi.org/10.1016/j.pt.2011.04.001
Ker HG, Aguiar-Soares RDO, Roatt BM, Moreira ND, Coura-Vital W, Carneiro CM, Teixeira-Carvalho A, Martins-Filho OA, Giunchetti RC, da Silveira-Lemos D, Reis AB (2013a) Effect of the preservative and temperature conditions on the stability of Leishmania infantum promastigotes antigens applied in a flow cytometry diagnostic method for canine visceral leishmaniasis. Diagn Microbiol Infect Dis 76(4):470–476. https://doi.org/10.1016/j.diagmicrobio.2013.04.007
Ker HG, Coura-Vital W, Aguiar-Soares RD, Roatt BM, das Dores Moreira N, Carneiro CM, Machado EM, Teixeira-Carvalho A, Martins-Filho OA, Giunchetti RC, Araujo MS, Coelho EA, da Silveira-Lemos D, Reis AB (2013b) Evaluation of a prototype flow cytometry test for serodiagnosis of canine visceral leishmaniasis. Clin Vaccine Immunol 20(12):1792–1798. https://doi.org/10.1128/CVI.00575-13
Laurenti MD, de Santana Leandro MV Jr, Tomokane TY, De Lucca HR, Aschar M, Souza CS, Silva RM, Marcondes M, da Matta VL (2014) Comparative evaluation of the DPP((R)) CVL rapid test for canine serodiagnosis in area of visceral leishmaniasis. Vet Parasitol 205(3-4):444–450. https://doi.org/10.1016/j.vetpar.2014.09.002
Lira RA, Cavalcanti MP, Nakazawa M, Ferreira AG, Silva ED, Abath FG, Alves LC, Souza WV, Gomes YM (2006) Canine visceral leishmaniosis: a comparative analysis of the EIE-leishmaniose-visceral-canina-Bio-Manguinhos and the IFI-leishmaniose-visceral-canina-Bio-Manguinhos kits. Vet Parasitol 137(1-2):11–16. https://doi.org/10.1016/j.vetpar.2005.12.020
Mancianti F, Gramiccia M, Gradoni L, Pieri S (1988) Studies on canine leishmaniasis control. 1. Evolution of infection of different clinical forms of canine leishmaniasis following antimonial treatment. Trans R Soc Trop Med Hyg 82(4):566–567. https://doi.org/10.1016/0035-9203(88)90510-x
Marcondes M, Biondo AW, Gomes AA, Silva AR, Vieira RF, Camacho AA, Quinn J, Chandrashekar R (2011) Validation of a Leishmania infantum ELISA rapid test for serological diagnosis of Leishmania chagasi in dogs. Vet Parasitol 175(1-2):15–19. https://doi.org/10.1016/j.vetpar.2010.09.036
Marcondes M, de Lima VM, de Araujo MF, Hiramoto RM, Tolezano JE, Vieira RF, Biondo AW (2013) Longitudinal analysis of serological tests officially adopted by the Brazilian Ministry of Health for the diagnosis of canine visceral leishmaniasis in dogs vaccinated with Leishmune(R). Vet Parasitol 197(3-4):649–652. https://doi.org/10.1016/j.vetpar.2013.07.013
Menezes-Souza D, Mendes TA, Gomes Mde S, Bartholomeu DC, Fujiwara RT (2015) Improving serodiagnosis of human and canine leishmaniasis with recombinant Leishmania braziliensis cathepsin l-like protein and a synthetic peptide containing its linear B-cell epitope. PLoS Negl Trop Dis 9(1):e3426. https://doi.org/10.1371/journal.pntd.0003426
Mettler M, Grimm F, Capelli G, Camp H, Deplazes P (2005) Evaluation of enzyme-linked immunosorbent assays, an immunofluorescent-antibody test, and two rapid tests (immunochromatographic-dipstick and gel tests) for serological diagnosis of symptomatic and asymptomatic Leishmania infections in dogs. J Clin Microbiol 43(11):5515–5519. https://doi.org/10.1128/JCM.43.11.5515-5519.2005
Ministério da Saúde (2006) Manual de Vigilância e Controle da Leishmaniose Visceral, 1st edn. Secretaria de Vigilância em Saúde, Brasília Available: http://bvsms.saude.gov.br/bvs/publicacoes/manual_vigilancia_controle_leishmaniose_visceral.pdf.120
Ministério da Saúde (2011) Esclarecimento sobre substituição do protocolo diagnóstico da leihsmaniose visceral canina; Nota técnica conjunta nu 01/2011 - CGDT-CGLAB/DEVIT/SVS/MS
Mohebali M, Javadian E, Yaghoobi-Ershadi MR, Akhavan AA, Hajjaran H, Abaei MR (2004) Characterization of Leishmania infection in rodents from endemic areas of the Islamic Republic of Iran. East Mediterr Health J 10(4-5):591–599
Molina R, Amela C, Nieto J, San-Andres M, Gonzalez F, Castillo JA, Lucientes J, Alvar J (1994) Infectivity of dogs naturally infected with Leishmania infantum to colonized Phlebotomus perniciosus. Trans R Soc Trop Med Hyg 88(4):491–493. https://doi.org/10.1016/0035-9203(94)90446-4
Oliveira GG, Magalhaes FB, Teixeira MC, Pereira AM, Pinheiro CG, Santos LR, Nascimento MB, Bedor CN, Albuquerque AL, dos-Santos WL, Gomes YM, Moreira ED Jr, Brito ME, Pontes de Carvalho LC, de Melo Neto OP (2011) Characterization of novel Leishmania infantum recombinant proteins encoded by genes from five families with distinct capacities for serodiagnosis of canine and human visceral leishmaniasis. Am J Trop Med Hyg 85(6):1025–1034. https://doi.org/10.4269/ajtmh.2011.11-0102
Otranto D, Dantas-Torres F (2013) The prevention of canine leishmaniasis and its impact on public health. Trends Parasitol 29(7):339–345. https://doi.org/10.1016/j.pt.2013.05.003
PAHO (2017) Informe Epidemiológico das Américas. Leishmanioses. In: 5 (ed) Informe Leishmanioses. Organização de saúde Pan Americana, p 8
Porrozzi R, Santos da Costa MV, Teva A, Falqueto A, Ferreira AL, dos Santos CD, Fernandes AP, Gazzinelli RT, Campos-Neto A, Grimaldi G Jr (2007) Comparative evaluation of enzyme-linked immunosorbent assays based on crude and recombinant leishmanial antigens for serodiagnosis of symptomatic and asymptomatic Leishmania infantum visceral infections in dogs. Clin Vaccine Immunol 14(5):544–548. https://doi.org/10.1128/CVI.00420-06
Quijada L, Requena JM, Soto M, Gomez LC, Guzman F, Patarroyo ME, Alonso C (1996) Mapping of the linear antigenic determinants of the Leishmania infantum hsp70 recognized by leishmaniasis sera. Immunol Lett 52(2-3):73–79
Quinnell RJ, Courtenay O (2009) Transmission, reservoir hosts and control of zoonotic visceral leishmaniasis. Parasitology 136(14):1915–1934. https://doi.org/10.1017/S0031182009991156
Quinnell RJ, Carson C, Reithinger R, Garcez LM, Courtenay O (2013) Evaluation of rK39 rapid diagnostic tests for canine visceral leishmaniasis: longitudinal study and meta-analysis. PLoS Negl Trop Dis 7(1):e1992. https://doi.org/10.1371/journal.pntd.0001992
Reis AB, Teixeira-Carvalho A, Giunchetti RC, Guerra LL, Carvalho MG, Mayrink W, Genaro O, Correa-Oliveira R, Martins-Filho OA (2006) Phenotypic features of circulating leucocytes as immunological markers for clinical status and bone marrow parasite density in dogs naturally infected by Leishmania chagasi. Clin Exp Immunol 146(2):303–311. https://doi.org/10.1111/j.1365-2249.2006.03206.x
Riboldi E, Carvalho F, Romao PRT, Barcellos RB, Bello GL, Ramos RR, de Oliveira RT, Junior JPA, Rossetti ML, Dallegrave E (2018) Molecular method confirms Canine Leishmania infection detected by serological methods in non-endemic area of Brazil. Korean J Parasitol 56(1):11–19. https://doi.org/10.3347/kjp.2018.56.1.11
Roatt BM, Aguiar-Soares RD, Vitoriano-Souza J, Coura-Vital W, Braga SL, Correa-Oliveira R, Martins-Filho OA, Teixeira-Carvalho A, de Lana M, Figueiredo Gontijo N, Marques MJ, Giunchetti RC, Reis AB (2012) Performance of LBSap vaccine after intradermal challenge with L. infantum and saliva of Lu. longipalpis: immunogenicity and parasitological evaluation. PloS one 7(11):e49780. https://doi.org/10.1371/journal.pone.0049780
Romero GA, Boelaert M (2010) Control of visceral leishmaniasis in latin america-a systematic review. PLoS Negl Trop Dis 4(1):e584. https://doi.org/10.1371/journal.pntd.0000584
Silva DA, Madeira MF, Teixeira AC, de Souza CM, Figueiredo FB (2011) Laboratory tests performed on Leishmania seroreactive dogs euthanized by the leishmaniasis control program. Vet Parasitol 179(1-3):257–261. https://doi.org/10.1016/j.vetpar.2011.01.048
Silvestre R, Santarem N, Cunha J, Cardoso L, Nieto J, Carrillo E, Moreno J, Cordeiro-da-Silva A (2008) Serological evaluation of experimentally infected dogs by LicTXNPx-ELISA and amastigote-flow cytometry. Vet Parasitol 158(1-2):23–30. https://doi.org/10.1016/j.vetpar.2008.09.001
Solano-Gallego L, Koutinas A, Miro G, Cardoso L, Pennisi MG, Ferrer L, Bourdeau P, Oliva G, Baneth G (2009) Directions for the diagnosis, clinical staging, treatment and prevention of canine leishmaniosis. Vet Parasitol 165(1-2):1–18. https://doi.org/10.1016/j.vetpar.2009.05.022
Sousa S, Cardoso L, Reed SG, Reis AB, Martins-Filho OA, Silvestre R, Cordeiro da Silva A (2013) Development of a fluorescent based immunosensor for the serodiagnosis of canine leishmaniasis combining immunomagnetic separation and flow cytometry. PLoS Negl Trop Dis 7(8):e2371. https://doi.org/10.1371/journal.pntd.0002371
Souza CM, Silva ED, Ano Bom AP, Bastos RC, Nascimento HJ, da Silva Junior JG (2012) Evaluation of an ELISA for canine leishmaniasis immunodiagnostic using recombinant proteins. Parasite Immunol 34(1):1–7. https://doi.org/10.1111/j.1365-3024.2011.01334.x
Teixeira-Carvalho A, Campos FM, Geiger SM, Rocha RD, de Araujo FF, Vitelli-Avelar DM, Andrade MC, Araujo MS, Lemos EM, de Freitas Carneiro Proietti AB, Sabino EC, Caldas RG, Freitas CR, Campi-Azevedo AC, Eloi-Santos SM, Martins-Filho OA (2015) FC-TRIPLEX Chagas/Leish IgG1: a multiplexed flow cytometry method for differential serological diagnosis of chagas disease and leishmaniasis. PLoS One 10(4):e0122938. https://doi.org/10.1371/journal.pone.0122938
WHO (2017) Integrating neglected tropical diseases into global health and development: fourth WHO report on neglected tropical diseases
Yamasaki M, Inokuma H, Sugimoto C, Shaw SE, Aktas M, Yabsley MJ, Yamato O, Maede Y (2007) Comparison and phylogenetic analysis of the heat shock protein 70 gene of Babesia parasites from dogs. Vet Parasitol 145(3-4):217–227. https://doi.org/10.1016/j.vetpar.2007.01.003
Zanette MF, Lima VM, Laurenti MD, Rossi CN, Vides JP, Vieira RF, Biondo AW, Marcondes M (2014) Serological cross-reactivity of Trypanosoma cruzi, Ehrlichia canis, Toxoplasma gondii, Neospora caninum and Babesia canis to Leishmania infantum chagasi tests in dogs. Rev Soc Bras Med Trop 47(1):105–107. https://doi.org/10.1590/0037-8682-1723-2013
Acknowledgments
A.B.R., A.T.C., O.A.M.F., P.S.T.V, H.G.K., and R.C.F.B. are grateful to Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and CAPES for PQ fellowships. We also thank Megan M. Keller of the Leishmania Research Laboratory, of Internal Medicine, University of Iowa for the critical reading and review of the English of our article.
Funding
This work was supported by Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG, grants CBB - INV-00037-14 and CDS - APQ-03505-131), the Programa de Pesquisa para o SUS (PPSUS grant APQ-03505-13), and the Instituto Nacional de Ciência e Tecnologia em Doenças Tropicais (INCT-DT).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical statement
This study was approved by the Ethics Research Committee of the Universidade Federal de Ouro Preto (protocol number 083/2007). All procedures involving animals were performed in compliance with Brazilian federal law for animal experimentation (Law 11794/2008).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ker, H.G., Coura-Vital, W., Valadares, D.G. et al. Multiplex flow cytometry serology to diagnosis of canine visceral leishmaniasis. Appl Microbiol Biotechnol 103, 8179–8190 (2019). https://doi.org/10.1007/s00253-019-10068-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10068-x