Abstract
Cytosolic sulfotransferases (SULTs), one of the predominant phase II drug metabolizing enzymes (DME), play important roles in metabolism of xeno- and endobiotics to generate their sulfo-conjugates. These sulfo-conjugates often have biological activities but are difficult to study, because even though only small amounts are required to evaluate their efficacy and safety, chemical or biological synthesis of sulfo-conjugatesis is often challenging. Previously, we constructed a DME expression system for cytochrome P450 and UGT, using yeast cells, and successfully produced xenobiotic metabolites in a whole-cell-dependent manner. In this study, we developed a yeast expression system for human SULTs, including SULT1A1, 1A3, 1B1, 1C4, 1E1, and 2A1, in Saccharomyces cerevisiae and examined its sulfo-conjugate productivity. The recombinant yeast cells expressing each of the SULTs successfully produced several hundred milligram per liter of xeno- or endobioticsulfo-conjugates within 6 h. This whole-cell-dependent biosynthesis enabled us to produce sulfo-conjugates without the use of 3’-phosphoadenosine-5’-phosphosulfate, an expensive cofactor. Additionally, the production of regiospecific sulfo-conjugates of several polyphenols was possible with this method, making this novel yeast expression system a powerful tool for uncovering the metabolic pathways and biological actions of sulfo-conjugates.





Similar content being viewed by others
References
Adamusová E, Cais O, Vyklický V, Kudová E, Chodounská H, Horák M, Vyklický L Jr (2013) Pregnenolone sulfate activates NMDA receptor channels. Physiol Res 62:731–736, 6
Brand W, Boersma MG, Bik H, Hoek-van den Hil EF, Vervoort J, Barron D, Meinl W, Glatt H, Williamson G, van Bladeren PJ, Rietjens IM (2010) Phase II metabolism of hesperetin by individual UDP-glucuronosyltransferases and sulfotransferases and rat and human tissue samples. Drug Metab Dispos 38:617–625. https://doi.org/10.1124/dmd.109.031047
Buhl AE, Waldon DJ, Baker CA, Johnson GA (1990) Minoxidil sulfate is the active metabolite that stimulates hair follicles. J Invest Dermatol 95:553–557
Coughtrie MW (2016) Function and organization of the human cytosolic sulfotransferase (SULT) family. ChemBiol Interact 259:2–7. https://doi.org/10.1016/j.cbi.2016.05.005
Drăgan CA, Buchheit D, Bischoff D, Ebner T, Bureik M (2010) Glucuronide production by whole-cell biotransformation using genetically engineered fission yeast Schizosaccharomyces pombe. Drug Metab Dispos 38(3):509–515. https://doi.org/10.1124/dmd.109.030965
Gamage N, Barnett A, Hempel N, Duggleby RG, Windmill KF, Martin JL, McManus ME (2006) Human sulfotransferases and their role in chemical metabolism. Toxicol Sci 90(1):5–22. https://doi.org/10.1093/toxsci/kfj061
Hehonah N, Zhu X, Brix L, Bolton-Grob R, Barnett A, Windmill K, McManus M (1999) Molecular cloning, expression, localisation and functional characterisation of a rabbit SULT1C2 sulfotransferase. Int J Biochem Cell Biol 31(8):869–882. https://doi.org/10.1016/S1357-2725(99)00038-2
Her C, Kaur GP, Athwal RS, Weinshilboum RM (1997) Human sulfotransferase SULT1C1: cDNA cloning, tissue-specific expression, and chromosomal localization. Genomics 41:467–470
Ikushiro S, Nishikawa M, Masuyama Y, Shouji T, Fujii M, Hamada M, Nakajima N, Finel M, Yasuda K, Kamakura M, Sakaki T (2016) Biosynthesis of drug glucuronide metabolites in the budding yeast Saccharomyces cerevisiae. Mol Pharm 13:2274–2282. https://doi.org/10.1021/acs.molpharmaceut.5b00954
Jandik KA, Kruep D, Cartier M, Linhardt RJ (1996) Accelerated stability studies of heparin. J Pharm Sci 85:45–51. https://doi.org/10.1021/js9502736
Karamohamed S1, Nilsson J, Nourizad K, Ronaghi M, Pettersson B, Nyrén P (1999) Production, purification, and luminometric analysis of recombinant Saccharomyces cerevisiae MET3 adenosine triphosphate sulfurylase expressed in Escherichia coli. Protein Expr Purif 15:381–388
Kasai N, Ikushiro S, Hirosue S, Arisawa A, Ichinose H, Wariishi H, Ohta M, Sakaki T (2009) Enzymatic properties of cytochrome P450 catalyzing 3′-hydroxylation of naringenin from the white-rot fungus Phanerochaete chrysosporium. Biochem Biophys Res Commun 387:103–108. https://doi.org/10.1016/j.bbrc.2009.06.134
Kurata K, Takebayashi M, Morinobu S, Yamawaki S (2004) Beta-estradiol, dehydroepiandrosterone, and dehydroepiandrosterone sulfate protect against N-methyl-d-aspartate-induced neurotoxicity in rat hippocampal neurons by different mechanisms. J Pharmacol Exp Ther 311:237–245
LeblancN, WildeDW, KeefKD, HumeJR (1989) Electrophysiological mechanisms of minoxidil sulfate-induced vasodilation of rabbit portal vein. Circ Res65:1102–1011
Li H, Deyrup A, Mensch JR Jr, Domowicz M, Konstantinidis AK, Schwartz NB (1995) The isolation and characterization of cDNA encoding the mouse bifunctional ATP sulfurylase-adenosine 5′-phosphosulfate kinase. J Biol Chem 270:29453–29459
Mienville JM, Vicini S (1989) Pregnenolone sulfate antagonizes GABAA receptor-mediated currents via a reduction of channel opening frequency. Brain Res 489:190–194
Oeda K, Sakaki T, Ohkawa H (1985) Expression of rat liver cytochrome P-450MC cDNA in Saccharomyces cerevisiae. DNA 4(3):203–210. https://doi.org/10.1089/dna.1985.4.203
Patel KR, Andreadi C, Britton RG, Horner-Glister E, Karmokar A, Sale S, Brown VA, Brenner DE, Singh R, Steward WP, Gescher AJ, Brown K (2013) Sulfate metabolites provide an intracellular pool for resveratrol generation and induce autophagy with senescence. Sci Transl Med 5(205):205ra133. https://doi.org/10.1126/scitranslmed.3005870
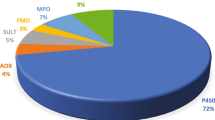
Riches Z, Stanley EL, Bloomer JC, Coughtrie MW (2009) Quantitative evaluation of the expression and activity of five major sulfotransferases (SULTs) in human tissues: the SULT “pie”. Drug Metab Dispos 37:2255–2261. https://doi.org/10.1124/dmd.109.028399
Sakaki T, Oeda K, Miyoshi M, Ohkawa H (1985) Characterization of rat cytochrome P-450MC synthesized in Saccharomyces cerevisiae. J Biochem, 98:167–175
Sakaki T, Shinkyo R, Takita T, Ohta M, Inouye K (2002) Biodegradation of polychlorinated dibenzo-p-dioxins by recombinant yeast expressing rat CYP1A subfamily. Arch Biochem Biophys 401:91–98. https://doi.org/10.1016/S0003-9861(02)00036-X
Sakaki T, Yamamoto K, Ikushiro S (2013) Possibility of application of cytochrome P450 to bioremediation of dioxins. Biotechnol Appl Biochem 60(1):65–70. https://doi.org/10.1002/bab.1067
Sakakibara Y, Yanagisawa K, Katafuchi J, Ringer DP, Takami Y, Nakayama T, Suiko M, Liu MC (1998) Molecular cloning, expression, and characterization of novel human SULT1C sulfotransferases that catalyze the sulfonation of N-hydroxy-2-acetylaminofluorene. J Biol Chem 273:33929–33935
Schueller K, Pignitter M, Somoza V (2015) Sulfated and glucuronated trans-resveratrol metabolites regulate chemokines and sirtuin-1 expression in U-937 macrophages. J Agric Food Chem 63(29):6535–6545. https://doi.org/10.1021/acs.jafc.5b01830
Schriek U, Schwenn JD (1986) Properties of the purified APS-kinase from Escherichia coli and Saccharomyces cerevisiae. Arch Microbiol 145:32–38
Shimohira T, Kurogi K, Hashiguchi T, Liu MC, Suiko M, Sakakibara Y (2017) Regioselective production of sulfated polyphenols using human cytosolic sulfotransferase-expressing Escherichia coli cells. J BiosciBioengpii S1389-1723(16):30404–30402. https://doi.org/10.1016/j.jbiosc.2017.02.006
Stanley EL, Hume R, Coughtrie MWH (2005) Expression profiling of human fetal cytosolic sulfotransferases involved in steroid and thyroid hormone metabolism and in detoxification. Mol Cell Endocrinol 240(1–2):32–42
Walker J, Schueller K, Schaefer LM, Pignitter M, Esefelder L, Somoza V (2014) Resveratrol and its metabolites inhibit pro-inflammatory effects of lipopolysaccharides in U-937 macrophages in plasma-representative concentrations. Food Funct 5:74–84. https://doi.org/10.1039/c3fo60236b
Wenzel E, Somoza V (2005) Metabolism and bioavailability of trans-resveratrol. Mol Nutr Food Res 49:472–481. https://doi.org/10.1002/mnfr.200500010
Wu FS, Gibbs TT, Farb DH (1991) Pregnenolone sulfate: a positive allosteric modulator at the N-methyl-D-aspartate receptor. Mol Pharmacol 40:333–336
Yoshinari K, Nagata K, Shimada M, Yamazoe Y (1998) Molecular characterization of ST1C-related human sulfotransferase. Carcinogenesis 19:951–953
Yasuda K, Ikushiro S, Kamakura M, Ohta M, Sakaki T (2010) Metabolism of sesamin by cytochrome P450 in human liver microsomes. Drug Metab Dispos 38(12):2117–2223. https://doi.org/10.1124/dmd.110.035659
Zhang M, Jagdmann GE Jr, Van Zandt M, Sheeler R, Beckett P, Schroeter H (2013) Chemical synthesis and characterization of epicatechin glucuronides and sulfates: bioanalytical standards for epicatechin metabolite identification. J Nat Prod 76:157–169. https://doi.org/10.1021/np300568m
Zhou X, Chandarajoti K, Pham TQ, Liu R, Liu J (2011) Expression of heparan sulfate sulfotransferases in Kluyveromyces lactis and preparation of 3′-phosphoadenosine-5′-phosphosulfate. Glycobiology 21:771–780. https://doi.org/10.1093/glycob/cwr001
Acknowledgements
The study was supported by the JSPSKAKENHI Grant Number JP26292072(SI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 555kb)
Rights and permissions
About this article
Cite this article
Nishikawa, M., Masuyama, Y., Nunome, M. et al. Whole-cell-dependent biosynthesis of sulfo-conjugate using human sulfotransferase expressing budding yeast. Appl Microbiol Biotechnol 102, 723–732 (2018). https://doi.org/10.1007/s00253-017-8621-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8621-x