Abstract
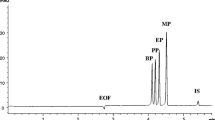
This study describes an on-line stacking CE approach by sweeping with whole capillary sample filling for analyzing five anabolic androgenic steroids in urine samples. The five anabolic steroids for detection were androstenedione, testosterone, epitestosterone, boldenone, and clostebol. Anabolic androgenic steroids are abused in sport doping because they can promote muscle growth. Therefore, a sensitive detection method is imperatively required for monitoring the urine samples of athletes. In this research, an interesting and reliable stacking capillary electrophoresis method was established for analysis of anabolic steroids in urine. After liquid–liquid extraction by n-hexane, the supernatant was dried and reconstituted with 30 mM phosphate buffer (pH 5.00) and loaded into the capillary by hydrodynamic injection (10 psi, 99.9 s). The stacking and separation were simultaneously accomplished at −20 kV in phosphate buffer (30 mM, pH 5.0) containing 100 mM sodium dodecyl sulfate and 40 % methanol. During the method validation, calibration curves were linear (r ≥ 0.990) over a range of 50–1,000 ng/mL for the five analytes. In the evaluation of precision and accuracy for this method, the absolute values of the RSD and the RE in the intra-day (n = 3) and inter-day (n = 5) analyses were all less than 6.6 %. The limit of detection for the five analytes was 30 ng/mL (S/N = 5, sampling 99.9 s at 10 psi). Compared with simple MECK, this stacking method possessed a 108- to 175-fold increase in sensitivity. This simple and sensitive stacking method could be used as a powerful tool for monitoring the illegal use of doping.




Similar content being viewed by others
References
Trout GJ, Kazlauskas R (2004) Chem Soc Rev 33:1–13
Schänzer W (1996) Clin Chem 42:1001–1020
Catlin DH, Hatton CK, Starcevic SH (1997) Clin Chem 43:1280–1288
Bowers LD, Segura J (1996) Clin Chem 42:999–1000
Wolthers BG, Kraan GPB (1999) J Chromatogr A 843:247–274
Aquilera R, Catlin DH, Becchi M, Phillips A, Wang C, Swerdloff RS, Pope HG, Hatton CK (1999) J Chromatogr B 727:95–105
Marchanol P, Bizec BL, Gade C, Montean F, André J (2000) J Chromatogr A 867:219–233
Shinohara Y, Baba S, Kasuya Y (1985) J Chromatogr 338:281–288
Becchi M, Aguilera R, Farizon Y, Flament MM, Casabianca H, James P (1994) Rapid Comm Mass Spectrom 8:304–384
Yu NH, Ho EN, Leung DK, Wan TS (2005) J Pharm Biomed Anal 37:1031–1038
Guan F, Uboh CE, Soma LR, Luo Y, Rudy J, Tobin T (2005) J Chromatogr B 829:56–68
Jeon BW, Yoo HH, Jeong ES, Kim HJ, Jin C, Kim DH, Lee J (2011) Anal Bioanal Chem 401:1353–1363
Badoud F, Grata E, Boccard J, Guillarme D, Veuthey JL, Rudaz S, Saugy M (2011) Anal Bioanal Chem 400:503–516
Terabe S, Otsuka K, Ichikawa K, Tsuchiya A, Ando T (1984) Anal Chem 56:113–116
Zhang CX, Thormann W (1998) Anal Chem 70:540–548
Britz-McKibbin P, Chen DDY (2000) Anal Chem 72:1242–1252
Chien RL, Burgi DS (1992) Anal Chem 64:1046–1050
Mikkers FEP, Everaerts FM, Verheggen TPEM (1979) J Chromatogr A 169:1–10
Quirino JP, Terabe S (1998) Anal Chem 70:149–157
Shih CM, Lin CH (2005) Electrophoresis 26:3495–3499
Quirino JP, Terabe S (1999) Science 282:465–468
Quirino JP, Terabe S (1999) Anal Chem 71:1638–1644
Zhang H, Zhou L, Chen X (2008) Electrophoresis 29:1556–1564
Quirino JP, Kim JB, Terabe S (2002) J Chromatogr A 965:357–373
Aranas AT, Guidote AM Jr, Quirino JP (2009) Anal Bioanal Chem 394:175–185
Su HL, Feng LI, Jen HP, Hsieh YZ (2009) J Chromatogr A 1216:3512–3517
Britz-McKibbin P, Otsuka K, Terabe S (2000) Electrophoresis 21:2768–2779
Acknowledgments
We gratefully acknowledge the support of the National Science Council of Taiwan and Kaohsiung Medical University for funding this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, CC., Cheng, SF., Cheng, HL. et al. Analysis of anabolic androgenic steroids in urine by full-capillary sample injection combined with a sweeping CE stacking method. Anal Bioanal Chem 405, 1969–1976 (2013). https://doi.org/10.1007/s00216-012-6640-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-012-6640-0