Abstract
Purpose
To compare the effect of intensive versus conventional blood glucose control in patients with traumatic brain injury.
Methods
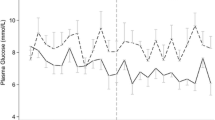
In a large international randomized trial patients were randomly assigned to a target blood glucose (BG) range of either 4.5–6.0 mmol/L (intensive control) or <10 mmol/L (conventional control). Patients with traumatic brain injury (TBI) were identified at randomization and data were collected to examine the extended Glasgow outcome score (includes mortality) at 24 months.
Results
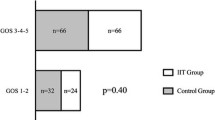
Of the 6104 randomized patients, 391 satisfied diagnostic criteria for TBI; 203 (51.9 %) were assigned to intensive and 188 (48.1 %) to conventional control; the primary outcome was available for 166 (81.8 %) and 149 (79.3 %) patients, respectively. The two groups had
similar baseline characteristics. At 2 years 98 (58.7 %) patients in the intensive group and 79 (53.0 %) in the conventional group had a favorable neurological outcome (odds ratio [OR] 1.26, 95 % CI 0.81–1.97; P = 0.3); 35 patients (20.9 %) in the intensive group and 34 (22.8 %) in the conventional group had died (OR 0.90, 95 % CI 0.53–1.53; P = 0.7); moderate hypoglycemia (BG 2.3–3.9 mmol/L; 41–70 mg/dL) occurred in 160/202 (79.2 %) and 17/188 (9.0 %), respectively (OR 38.3, 95 % CI 21.0–70.1; P < 0.0001); severe hypoglycemia (BG ≤ 2.2 mmol/L; ≤40 mg/dL) in 10 (4.9 %) and 0 (0.0 %), respectively (OR 20.5 95 % CI 1.2–351.6, P = 0.003).
Conclusion
Although patients with traumatic brain injury randomly assigned to intensive compared to conventional glucose control experienced moderate and severe hypoglycemia more frequently, we found no significant difference in clinically important outcomes.


Similar content being viewed by others
References
Krinsley JS (2003) Association between hyperglycemia and increased hospital mortality in a heterogeneous population of critically ill patients. Mayo Clin Proc 78:1471–1478
Gale SC, Sicoutris C, Reilly PM, Schwab CW, Gracias VH (2007) Poor glycemic control is associated with increased mortality in critically ill trauma patients. Am Surg 73:454–460
Preiser JC, Devos P, Ruiz-Santana S, Melot C, Annane D, Groeneveld J, Iapichino G, Leverve X, Nitenberg G, Singer P, Wernerman J, Joannidis M, Stecher A, Chiolero R (2009) A prospective randomized multi-centre controlled trial on tight glucose control by intensive insulin therapy in adult intensive care units: the Glu control study. Intensive Care Med 35:1738–1748
Lam AM, Winn HR, Cullen BF, Sundling N (1991) Hyperglycemia and neurological outcome in patients with head injury. J Neurosurg 75:545–551
Yang SY, Zhang S, Wang ML (1995) Clinical significance of admission hyperglycemia and factors related to it in patients with acute severe head injury. Surg Neurol 44:373–377
Liu-DeRyke X, Collingridge DS, Orme J, Roller D, Zurasky J, Rhoney DH (2009) Clinical impact of early hyperglycemia during acute phase of traumatic brain injury. Neurocrit Care 11:151–157
Griesdale DE, Tremblay MH, McEwen J, Chittock DR (2009) Glucose control and mortality in patients with severe traumatic brain injury. Neurocrit Care 11:311–316
Salim A, Hadjizacharia P, Dubose J, Brown C, Inaba K, Chan LS, Margulies D (2009) Persistent hyperglycemia in severe traumatic brain injury: an independent predictor of outcome. Am Surg 75:25–29
Kalfon P, Giraudeau B, Ichai C, Guerrini A, Brechot N, Cinotti R, Dequin PF, Riu-Poulenc B, Montravers P, Annane D, Dupont H, Sorine M, Riou B, CGAO-REA Study Group (2014) Tight computerized versus conventional glucose control in the ICU: a randomized controlled trial. Intensive Care Med 40:171–181
Arabi YM, Dabbagh OC, Tamim HM, Al-Shimemeri AA, Memish ZA, Haddad SH, Syed SJ, Giridhar HR, Rishu AH, Al-Daker MO, Kahoul SH, Britts RJ, Sakkijha MH (2008) Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med 36:3190–3197
De La Rosa GDC, Donado JH, Restrepo AH, Quintero AM, Gonzalez LG, Saldarriaga NE, Bedoya M, Toro JM, Velasquez JB, Valencia JC, Arango CM, Aleman PH, Vasquez EM, Chavarriaga JC, Yepes A, Pulido W, Cadavid CA (2008) Strict glycaemic control in patients hospitalised in a mixed medical and surgical intensive care unit: a randomised clinical trial. Crit Care 12:R120
Van den Berghe G, Wilmer A, Hermans G, Meersseman W, Wouters PJ, Milants I, Van Wijngaerden E, Bobbaers H, Bouillon R (2006) Intensive insulin therapy in the medical ICU. N Engl J Med 354:449–461
NICE-SUGAR Study Investigators (2009) Intensive versus conventional glucose control in critically ill patients. N Engl J Med 360:1346–1349
The COIITSS Study Investigators (2010) Corticosteroid treatment and intensive insulin therapy for septic shock in adults: a randomized controlled trial. JAMA 303:341–348
Oddo M, Schmidt JM, Carrera E, Badjatia N, Connolly ES, Presciutti M, Ostapkovich ND, Levine JM, Le Roux P, Mayer SA (2008) Impact of tight glycemic control on cerebral glucose metabolism after severe brain injury: a microdialysis study. Crit Care Med 36:3233–3238
Van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R (2001) Intensive insulin therapy in critically ill patients. N Engl J Med 345:1359–1367
Griesdale DE, de Souza RJ, van Dam RM, Heyland DK, Cook DJ, Malhotra A, Dhaliwal R, Henderson WR, Chittock DR, Finfer S, Talmor D (2009) Intensive insulin therapy and mortality among critically ill patients: a meta-analysis including NICE-SUGAR study data. CMAJ 180:831–837
Kansagara D, Fu R, Freeman M, Wolf F, Helfand M (2011) Intensive insulin therapy in hospitalized patients: a systematic review. Ann Intern Med 154:268–282
American Diabetes Association (2012) Standards of medical care in diabetes-2012. Diabetes Care 35(Suppl 1):S11–S63
Qaseem A, Humphrey LL, Chou R, Snow V, Shekelle P, Clinical Guidelines Committee of the American College of Physicians (2011) Use of intensive insulin therapy for the management of glycemic control in hospitalized patients: a clinical practice guideline from the American College of Physicians. Ann Intern Med 154:260–267
Egi M, Bellomo R, Stachowski E, French CJ, Hart GK, Taori G, Hegarty C, Bailey M (2010) Hypoglycemia and outcome in critically ill patients. Mayo Clin Proc 85:217–224
Hermanides J, Bosman RJ, Vriesendorp TM, Dotsch R, Rosendaal FR, Zandstra DF, Hoekstra JB, Devries JH (2010) Hypoglycemia is associated with intensive care unit mortality. Crit Care Med 38:1430–1434
Kaukonen KM, Rantala M, Pettila V, Hynninen M (2008) Severe hypoglycemia during intensive insulin therapy. Acta Anaesthesiol Scand 53:61–65
The NICE SUGAR Study Investigators (2012) Hypoglycemia and risk of death in critically ill patients. N Engl J Med 367:1108–1118
Meierhans R, Bechir M, Ludwig S, Sommerfeld J, Brandi G, Haberthur C, Stocker R, Stover JF (2010) Brain metabolism is significantly impaired at blood glucose below 6 mM and brain glucose below 1 mM in patients with severe traumatic brain injury. Crit Care 14:R13
Vespa P, Boonyaputthikul R, McArthur DL, Miller C, Etchepare M, Bergsneider M, Glenn T, Martin N, Hovda D (2006) Intensive insulin therapy reduces microdialysis glucose values without altering glucose utilization or improving the lactate/pyruvate ratio after traumatic brain injury. Crit Care Med 34:850–856
Vespa PM, McArthur D, O’Phelan K, Glenn T, Etchepare M, Kelly D, Bergsneider M, Martin NA, Hovda DA (2003) Persistently low extracellular glucose correlates with poor outcome 6 months after human traumatic brain injury despite a lack of increased lactate: a microdialysis study. J Cereb Blood Flow Metab 23:865–877
Vespa P, McArthur DL, Stein N, Huang SC, Shao W, Filippou M, Etchepare M, Glenn T, Hovda DA (2012) Tight glycemic control increases metabolic distress in traumatic brain injury: a randomized controlled within-subjects trial. Crit Care Med 40:1923–1929
Teasdale G, Jennett B (1974) Assessment of coma and impaired consciousness. A practical scale. Lancet 2:81–84
Baker SP, O’Neill B, Haddon W Jr, Long WB (1974) The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma 14:187–196
Marshall LF, Marshall SB, Klauber MR, Berkum Van, Clark M, Eisenberg H, Jane JA, Luerssen TG, Marmarou A, Foulkes MA (1992) The diagnosis of head injury requires a classification based on computed axial tomography. J Neurotrauma 9(Suppl 1):S287–S292
Vincent JL, de Mendonca A, Cantraine F, Moreno R, Takala J, Suter PM, Sprung CL, Colardyn F, Blecher S (1998) Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: results of a multicenter, prospective study. Working group on “sepsis-related problems” of the European Society of Intensive Care Medicine. Crit Care Med 26:1793–1800
Teasdale GM, Pettigrew LE, Wilson JT, Murray G, Jennett B (1998) Analyzing outcome of treatment of severe head injury: a review and update on advancing the use of the Glasgow Outcome Scale. J Neurotrauma 15:587–597
Van den Berghe G, Schoonheydt K, Becx P, Bruyninckx F, Wouters PJ (2005) Insulin therapy protects the central and peripheral nervous system of intensive care patients. Neurology 64:1348–1353
Bilotta F, Caramia R, Cernak I, Paoloni FP, Doronzio A, Cuzzone V, Santoro A, Rosa G (2008) Intensive insulin therapy after severe traumatic brain injury: a randomized clinical trial. Neurocrit Care 9:159–166
Yang M, Guo Q, Zhang X, Sun S, Wang Y, Zhao L, Hu E, Li C (2009) Intensive insulin therapy on infection rate, days in NICU, in-hospital mortality and neurological outcome in severe traumatic brain injury patients: a randomized controlled trial. Int J Nursing Stud 46:753–758
Coester A, Neumann CR, Schmidt MI (2010) Intensive insulin therapy in severe traumatic brain injury: a randomized trial. J Trauma 68:904–911
Cinotti R, Ichai C, Orban JC, Kalfon P, Feuillet F, Roquilly A, Riou B, Blanloeil Y, Asehnoune K, Rozec B (2014) Effects of tight computerized glucose control on neurological outcome in severely brain injured patients: a multicenter sub-group analysis of the randomized-controlled open-label CGAO-REA study. Crit Care 18:498
Dickinson K, Bunn F, Wentz R, Edwards P, Roberts I (2000) Size and quality of randomised controlled trials in head injury: review of published studies. BMJ 320:1308–1311
Kramer A, Roberts D, Zygun D (2012) Optimal glycemic control in neurocritical care patients: a systematic review and meta-analysis. Crit Care 16:R203
Acknowledgments
Australian National Health and Medical Research Council; Health Research Council of New Zealand; Canadian Institutes for Health Research; Victorian Neurotrauma Initiative.
Conflicts of interest
None of the members of the writing committee have conflicts of interest in respect of this manuscript.
Author information
Authors and Affiliations
Consortia
Additional information
The NICE-SUGAR study is a collaboration of the Australia and New Zealand Intensive Care Society Clinical Trials Group, The George Institute for Global Health (University of Sydney), the Canadian Critical Care Trials Group and Vancouver Coastal Health Research Institute (University of British Columbia). The NICE-SUGAR study investigators are listed in the appendix.
Take-home message: Patients with traumatic brain injury randomly assigned to intensive compared to conventional glucose control experienced moderate and severe hypoglycemia more frequently. Mortality and neurological recovery assessed using the extended Glasgow outcome score did not differ between groups.
Please address correspondence to Prof. Simon Finfer at The George Institute for Global Health, GPO Box 5389, Sydney, NSW 2001, Australia (e-mail: sfinfer@georgeinstitute.org.au; Tel.: +61-2-94632633).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
NICE-SUGAR Study Investigators
Writing Committee: Simon Finfer (Chair, PI NICE Study), Dean Chittock (PI SUGAR Trial), Yang Li, Denise Foster, Vinay Dhingra, Rinaldo Bellomo, Deborah Cook, Peter Dodek, Paul Hebert, William Henderson, Daren Heyland, Alisa Higgins, Colin McArthur, Imogen Mitchell, John Myburgh, Bruce Robinson, Juan Ronco.
NICE (ANZ) Management Committee: Simon Finfer, (Chair), Deborah Blair, (Project Manager), Rinaldo Bellomo, Colin McArthur (Lead Investigator, New Zealand), Imogen Mitchell, John Myburgh, Robyn Norton, Julie Potter.
SUGAR (North American) Management Committee: Dean Chittock (Chair), Vinay Dhingra (Past Chair), Denise Foster (Senior Project Manager), Deborah Cook, Peter Dodek, Paul Hébert, William Henderson, Daren Heyland, Ellen McDonald, Juan Ronco. (Ex officio Member: Irwin Schweitzer, Canadian Institutes for Health Research.)
Independent Data Monitoring Committee: Richard Peto (Chair), Peter Sandercock, Charles Sprung, J. Duncan Young.
Statistical Analysis (The George Institute for International Health, University of Sydney, NSW, Australia) Yang Li, Qiang Li, Severine Bompoint, Laurent Billot.
Study Coordinating Centre (The George Institute for International Health, University of Sydney, NSW, Australia) Yang Li, Leonie Crampton, Fotios Darcy, Kathy Jayne, Viraji Kumarasinghe, Lorraine Little, Suzanne McEvoy, Stephen MacMahon, Sameer Pandey, Suzanne Ryan, Ravi Shukla, Bala Vijayan.
University of Sydney (Faculty of Medicine), Kolling Institute and Department of Endocrinology, Royal North Shore Hospital: Bruce Robinson (Dean).
ANZ Site investigators: (Alphabetically by institution, Australia unless stated, NZ = New Zealand. NSW = New South Wales, WA = Western Australia).
Auckland City Hospital (DCCM), Auckland, NZ: Susan Atherton, Jeanette Bell, Louise Hadfield, Craig Hourigan, Colin McArthur, Lynette Newby, Catherine Simmonds.
Auckland City Hospital (CVICU), Auckland, NZ: Heidi Buhr, Michelle Eccleston, Shay McGuinness, Rachael Parke.
The Austin Hospital, Melbourne, Victoria: Rinaldo Bellomo, Samantha Bates, Donna Goldsmith, Inga Mercer, Kim O’Sullivan.
Ballarat Base Hospital, Ballarat, Victoria: Robert Gazzard, Dianne Hill, Christine Tauschke.
Blacktown Hospital, Blacktown, NSW: Dhawal Ghelani, Kiran Nand, Graham Reece, Treena Sara.
Box Hill Hospital, Box Hill, Victoria: Suzanne Elliott, David Ernest, Angela Hamilton.
The Canberra Hospital, Canberra, Australian Capital Territory: Rebecca Ashley, Andrew Bailey, Elise Crowfoot, Jelena Gissane, Imogen Mitchell, Jamie Ranse, Joy Whiting.
Concord Repatriation Hospital, Concord, NSW: Kristina Douglas, David Milliss, Jeff Tan, Helen Wong.
Fremantle Hospital, Fremantle, WA: David Blythe, Annemarie Palermo.
John Hunter Hospital, Newcastle, NSW: Miranda Hardie, Peter Harrigan, Brett McFadyen.
Liverpool Hospital, Liverpool, NSW: Sharon Micallef, Michael Parr.
Middlemore Hospital, Auckland, NZ: Anna Boase, Judi Tai, Anthony Williams.
Nepean Hospital, Nepean, NSW: Louise Cole, Ian Seppelt, Leonie Weisbrodt, Sarah Whereat.
North Shore Hospital, Auckland, NZ: Annette Flanagan, Janet Liang.
Prince of Wales Hospital, Sydney, NSW: Frances Bass, Michelle Campbell, Naomi Hammond, Lisa Nicholson, Yahya Shehabi.
Queen Elizabeth Hospital, Adelaide, South Australia: Jonathan Foote, Sandra Peake, Patricia Williams.
Royal Brisbane Hospital, Brisbane, Queensland: Renae Deans, Cheryl Fourie, Melissa Lassig-Smith, Jeffrey Lipman, Janine Stuart.
Royal Hobart Hospital, Hobart, Tasmania: Anthony Bell, Tanya Field, Richard McAllister, Kathryn Marsden, Andrew Turner.
Royal North Shore Hospital, Sydney, NSW: Susan Ankers, Simon Bird, Simon Finfer, Richard Lee, Anne O’Connor, Julie Potter, Naresh Ramakrishnan, Ray Raper.
St. George Hospital, Sydney, NSW: Vanessa Dhiacou, Kathryn Girling, Alina Jovanovska, John Myburgh.
St. Vincent’s Hospital, Melbourne, Victoria: Nicole Groves, Jenny Holmes, John Santamaria, Roger Smith.
Sir Charles Gairdner Hospital, Perth, WA: Stuart Baker, Brigit Roberts.
Wellington Hospital, Wellington, NZ: Lynne Andrews, Richard Dinsdale, Rosemary Fenton, Diane Mackle, Sarah Mortimer.
Western Hospital, Melbourne, Victoria: Craig French, Lorraine Little, Heike Raunow.
Wollongong Hospital, Wollongong, NSW: Michelle Gales, Francisco Hill, Sundaram Rachakonda, Darren Rogan.
NSW Institute of Trauma and Injury Management, Sydney, NSW, Australia: Christine Allsop.
Australian and New Zealand Intensive Care Research Centre, Melbourne, Victoria, Australia: Alisa Higgins.
The Alfred Health and Monash University, Melbourne, Victoria, Australia: Dinesh Varma
North American Site investigators (Canada unless stated):
Calgary Health Region, Calgary, Alberta—Kevin Champagne, Christopher Doig, Linda Knox, Pauline Taylor, Crystal Wilson.
Kingston General Hospital, Kingston, Ontario—John Drover, Sharlene Hammond, Elizabeth Mann, Monica Myers, Amber Robinson.
Maisonneuve Rosemont Hospital, Montreal, Quebec—Johanne Harvey, Yoanna Skrobik.
Mayo Clinic, Rochester, Minnesota, USA—Anita Baumgartner, Laurie Meade, Nicholas Vlahakis.
Mount Sinai Hospital, Toronto, Ontario—Cheryl Ethier, Marnie Kramer-Kile, Sangeeta Mehta.
Ottawa General Hospital, Ottawa, Ontario—Claude Gaudert, Salmaan Kanji, Tracy McArdle, Irene Watpool.
St Joseph’s Healthcare, Hamilton, Ontario—France Clark, Deborah Cook, Ellen McDonald, Andrea Tkaczyk.
St Michael’s Hospital, Toronto, Ontario—John Marshall, Jeanna Morrissey, Orla Smith, Kerri Porretta, Victoria Wen.
St Paul’s Hospital, Vancouver, British Columbia—Betty Jean Ashley, Peter Dodek, Sheilagh Mans.
Sunnybrook Health Sciences Centre, Toronto, Ontario—Boris Bojilov, Karen Code, Robert Fowler, Nicole Marinoff.
Toronto General Hospital, Toronto, Ontario—Leslie Chu, John Granton, Margaret McGrath-Chong, Marilyn Steinberg.
Toronto Western Hospital, Toronto, Ontario—Niall Ferguson, Stephanie Go, Andrea Matte, Jonathan Rosenberg, James Stevenson.
University of Alberta Hospital, Edmonton, Alberta—Michael Jacka, Leeca Sonnema.
Vancouver General Hospital, Vancouver, British Columbia—Roger Autio, Angela Brevner, Dean Chittock, Dara Davies, Pia Ganz, Maureen Gardner, Susan Logie, Laurie Smith.
Vancouver Island Health Authority, Victoria, British Columbia—Leslie Atkins, Fiona Auld, Marni Stewart, Gordon Wood.
Rights and permissions
About this article
Cite this article
The NICE-SUGAR Study Investigators for the Australian and New Zealand Intensive Care Society Clinical Trials Group and the Canadian Critical Care Trials Group. Intensive versus conventional glucose control in critically ill patients with traumatic brain injury: long-term follow-up of a subgroup of patients from the NICE-SUGAR study. Intensive Care Med 41, 1037–1047 (2015). https://doi.org/10.1007/s00134-015-3757-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-015-3757-6