Abstract
Introduction
To investigate the risks and possible benefits of routine versus intensive insulin therapy, assessed by the frequency of hypoglycemic events defined as a glucose concentration less than 80 mg/dl (<4.44 mmol/l) in patients admitted to the intensive care unit (ICU) after severe traumatic brain injury (TBI).
Methods and Results
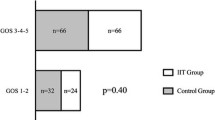
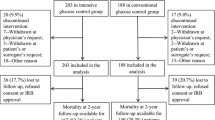
Ninety-seven patients admitted after severe TBI, were enrolled and randomly assigned to two groups of target glycemia. Insulin was infused at conventional rates when blood glucose levels exceeded 220 mg/dl (12.22 mmol/l) or at intensive rates, to maintain glycemia at 80–120 mg/dl (4.44–6.66 mmol/l). The following primary and outcome variables were measured during follow-up: hypoglycemic episodes, duration of ICU stay, infection rate, and 6-month mortality and neurologic outcome measured using the Glasgow Outcome Scale (GOS). Episodes of hypoglycemia (defined as blood glucose <80 mg/dl or 4.44 mmol/l) were significantly higher in patients receiving intensive insulin therapy: median (min–max) conventional insulin therapy 7 (range 0–11) vs. intensive insulin therapy 15 (range 6–33); P<0.0001. Duration of ICU stay was shorter in patients receiving intensive insulin therapy (7.3 vs. 10.0 days; P < 0.05); while infection rates during ICU stay (25.0% vs. 38.8%, P = 0.15), and GOS scores and mortality at 6 months were similar in the two groups.
Conclusions
Intensive insulin therapy significantly increases the risk of hypoglycemic episodes. Even though patients receiving intensive insulin therapy have shorter ICU stays and infection rates similar to those receiving conventional insulin therapy, both groups have similar follow-up mortality and neurologic outcome. Hence if intensive insulin therapy is to be used, great effort must be taken to avoid hypoglycemia.
Similar content being viewed by others
References
Van den Berghe G, Wouters P, Weekers F, et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345:1359–67.
Van den Berghe G, Schoonheydt K, Becx P, et al. Insulin therapy protects the central and peripheral nervous system of intensive care patients. Neurology. 2005;64:1348–53.
Krinsley JS, Grover A. Severe hypoglycemia in critically ill patients: risk factors and outcomes. Crit Care Med. 2007;35(10):2262–7.
Turina M, Christ-Crain M, Polk HC Jr. Diabetes and hyperglycemia: strict glycemic control. Crit Care Med. 2006;34(9 Suppl):291–300.
Van den Berghe G, Wilmer A, Milants I, et al. Intensive insulin therapy in mixed medical/surgical intensive care units. Diabetes. 2006;55(11):3151–9.
Yamada K, Milbrandt EB, Moore J. Intensive insulin therapy in the medical ICU – not so sweet? Crit Care. 2007;11(4):311.
Thomas G, Rojas MC, Epstein SK, Balk EM, Liangos O, Jaber BL. Insulin therapy and acute kidney injury in critically ill patients a systematic review. Nephrol Dial Transplant. 2007;22(10):2849–55.
Mechanick JI, Handelsman Y, Bloomgarden ZT. Hypoglycemia in the intensive care unit. Curr Opin Clin Nutr Metab Care. 2007;10(2):193–6.
Pulsinelli WA, Levy DE, Sigsbee B, et al. Increased damage after ischemic stroke in patients with hyperglycemia with or without established diabetes mellitus. Am J Med. 1983;74:540–4.
Scott JF, Robinson GM, French JM, et al. Glucose potassium insulin infusions in the treatment of acute stroke patients with mild to moderate hyperglycemia. Stroke. 1999;30:793–9.
Bhalla A, Tilling K, Kolominsky-Rabas P, et al. Variation in the management of acute physiological parameters after ischemic stroke: a European perspective. Eur J Neurol. 2003;10(1):25–33.
Bilotta F, Spinelli A, Giovannini F, et al. Effects of intensive insulin infusion on infection rates, vasospasm and neurologic outcome in patients with acute subarachnoid hemorrhage admitted to a postoperative neurosurgical intensive care unit after surgical clipping of intracranial aneurysms a prospective randomized trial. J Neurosurg Anesth. 2007;19(3):156–60.
Walia S, Sutcliffe AJ. The relationship between blood glucose, mean arterial pressure and outcome after head injury: an observational study. Injury 2002;33:339–44.
Robertson CS, Goodman JC, Narayan RK, Contant CF, Grossman RG. The effect of glucose administration on carbohydrate metabolism after head injury. J Neurosurg. 1991;74(1):43–50.
Bergsneider MA, Hovda DA, Shalmon E, et al. Cerebral hyperglycolysis following severe human traumatic brain inyury: a positron emission tomography study. J Neurosurg. 1997;86:241–51.
Hamlin GP, Cernak I, Wixey JA, et al. Increased expression of neuronal glucose transporter 3 but not glial glucose transporter 1 following severe diffuse traumatic brain injury in rats. J Neurotrauma. 2001;18(10):1011–8.
Vespa P, Boonyaputthinkul R, McArthur DL, et al. Intensive insulin therapy reduces microdialysis glucose values without altering glucose utilization or improving the lactate/pyruvate ratio after traumatic brain injury. Crit Care Med. 2006;34(3):850–6.
Vespa P, O’Phelan K, McArthur D, Miller C, Eliseo M, Hirt D, Glenn T, Hovda DA. Pericontusional brain tissue exhibits persistent elevation of lactate/pyruvate ratio independent of cerebral perfusion pressure. Crit Care Med. 2007;35(4):1153–60.
Le Galle JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 1994;271(17):1321.
University of Minnesota Medical Center Continuous Intravenous Insulin Infusion Orders Protocol. APSF Newsletter Summer 2006. apsf.org.
Krinsley JS. Effect of an intensive glucose management protocol on the mortality of critically ill adult patients. Mayo Clin Proc. 2004;79(8):992–1000.
Souba WW. Nutritional support. N Engl J Med. 1997;336:41–8.
Himanen L, Portin R, Isoniemi H, Helenius H, Kurki T, Tenovuo O. Longitudinal cognitive changes in traumatic brain injury: a 30-year follow-up study. Neurology. 2006;66(2):187–92.
Luukinen H, Viramo P, Koski K, Laippala P, Kivela SL. Head injuries and cognitive decline among older adults: a population-based study. Neurology. 1999;52(3):557–62.
Horan TC, Gaynes RP. Surveillance of nosocomial infections In: Mayhall CG, editor. Hospital and infection control, 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2004. p. 1659–702.
Weinstein MP, Towns ML, Quartey SM, et al. The clinical significance of positive blood cultures in the 1990s: a prospective comprehensive evaluation of the microbiology, epidemiology, and outcome of bacteremia and fungemia in adults. Clin Infect Dis. 1997;24:584–602.
Pittas AG, Siegel RD, Lau J. Insulin therapy for critically ill hospitalized patients: a metanalysis of randomized controlled trials. Arch Intern Med. 2004;164(18):2005–11.
Widom B, Simonson DC. Glycemic control and neuropsychologic function during hypoglycemia in patients with insulin-dependent diabetes mellitus. Ann Intern Med. 1990;112(12):904–12.
Boyle PJ, Schwartz NS, Shah SD, et al. Plasma glucose concentrations at the onset of hypoglycemic symptoms in patients with poorly controlled diabetes and in nondiabetics. N Engl J Med. 1988;318:1487–92.
Li PA, Shamloo M, Smith ML, et al. The influence of plasma glucose concentrations on ischemic brain damage is a threshold function. Neurosci Lett. 1994;177:63–5.
Sieber FE, Traystman RJ. Special issues: glucose and brain. Crit Care Med. 1992;20(1):104–14.
Wass CT, Lanier WL. Glucose modulation of ischemic brain injury: review and clinical recommendations. Mayo Clin Proc. 1996;71:801–12.
Preiser JC, Devos P. Clinical experience with tight glucose control by intensive insulin therapy. Crit Care Med. 2007;35:S503–7.
Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 1997;20:1183–97.
CRASH Trial Collaborators. Effect of intravenous corticosteroids on death within 14 days in 10,008 adults with clinically significant head injury (MRC CRASH Trial): randomised, placebo-controlled trial. Lancet. 2004;364:1321–8.
Acknowledgments
We are indebted to all the intensive care staff for their active cooperation and excellent compliance with the study protocol. We also thank Drs Fabio Araimo, Floriana Baisi, Giovanna Branca, Donato Colagiovanni, Nicola DeBlasis, Carmela Imperiale, Giuseppina Magni, Filomena Musolino, Marina Pennacchia, Letizia Pennacchiotti, Francesca Rinaldi, Paolo Tordiglione for patient care; Dr Carolina Maurizio for collecting data; Dr Alessandro Laviano for advice on patients’ nutrition; and Drs Laura Scorsolini and Mario Venditti for their continuous assistance in the clinical and laboratory diagnosis of infections.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was done in the Department of Neuroanaesthesia, University of Rome “La Sapienza”, Rome, Italy, and was in part presented at the Euroanaesthesia 2006 meeting Madrid, Spain.
Appendices
Appendix 1: ICU Protocol for Insulin Infusion
Glucose value (mg/dl) | Insulin dose |
|---|---|
Intensive insulin therapy | |
For first blood glucose measurement | |
121–130 | ½ unit/h |
131–140 | 1 unit/h |
141–160 | 2 units/h |
161–181 | Give 2 units IV bolus and 2.5 units/h |
181–200 | Give 2 units IV bolus and 3 units/h |
201–220 | Give 2 units IV bolus and 3.5 units/h |
>220 | Give 2 units IV bolus and 4 units/h |
For successive blood glucose measurements | |
80–120 | Continue current infusion rate |
121–141 | Increase insulin infusion by 0.5 unit/h |
141–160 | Increase insulin infusion by 1 unit/h |
161–180 | Increase insulin infusion by 1.5 unit/h |
181–200 | Increase insulin infusion by 2 units/h |
201–220 | Increase insulin infusion by 2.5 units/h |
>220 | Increase insulin infusion by 3 units/h |
Conventional insulin therapy | |
For first blood glucose measurement | |
220–240 | Give 2 units IV bolus and 2 units/h |
241–260 | Give 2 units IV bolus and 3.5 units/h |
261–280 | Give 2 units IV bolus and 4 units/h |
>280 | Give 4 units IV bolus and 4 units/h |
For successive blood glucose measurements | |
<220 | Continue current infusion rate |
220–240 | Increase insulin infusion by 1 unit/h |
241–260 | Increase insulin infusion by 2 units/h |
261–280 | Increase insulin infusion by 3 units/h |
>280 | Increase insulin infusion by 4–5 units/h |
Appendix Legend
All patients with blood glucose levels <4.44 mmol/l (<80 mg/dl) received glucose in a bolus (0.1 g/kg at a concentration of 33%) with the insulin infusion stopped. The glucose concentration was then measured within 30 min.
All patients receiving continuous insulin must receive a continuous surge of enteral or parenteral nutrition.
The insulin infusion is discontinued if the patient has to leave the ICU for a diagnostic test as well as upon discharge from the ICU.
Appendix 2: Glasgow Outcome Score
Score | Rating | Definition |
|---|---|---|
5 | Good recovery | Resumption of normal life despite minor deficits |
4 | Moderate disability | Disabled but independent. Can work in sheltered setting |
3 | Severe disability | Conscious but disabled. Dependent for daily support |
2 | Persistent vegetative | Minimal responsiveness |
1 | Death | Non-survival |
Rights and permissions
About this article
Cite this article
Bilotta, F., Caramia, R., Cernak, I. et al. Intensive Insulin Therapy After Severe Traumatic Brain Injury: A Randomized Clinical Trial. Neurocrit Care 9, 159–166 (2008). https://doi.org/10.1007/s12028-008-9084-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-008-9084-9