Abstract
Purpose
To evaluate the prognostic value of the Cv-aCO2/Da-vO2 ratio combined with lactate levels during the early phases of resuscitation in septic shock.
Methods
Prospective observational study in a 60-bed mixed ICU. One hundred and thirty-five patients with septic shock were included. The resuscitation protocol targeted mean arterial pressure, pulse pressure variations or central venous pressure, mixed venous oxygen saturation, and lactate levels. Patients were classified into four groups according to lactate levels and Cv-aCO2/Da-vO2 ratio at 6 h of resuscitation (T6): group 1, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 >1.0; group 2, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 ≤1.0; group 3, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 >1.0; and group 4, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 ≤1.0.
Results
Combination of hyperlactatemia and high Cv-aCO2/Da-vO2 ratio was associated with the worst SOFA scores and lower survival rates at day 28 [log rank (Mantel–Cox) = 31.39, p < 0.0001]. Normalization of both variables was associated with the best outcomes. Patients with a high Cv-aCO2/Da-vO2 ratio and lactate <2.0 mmol/L had similar outcomes to hyperlactatemic patients with low Cv-aCO2/Da-vO2 ratio. The multivariate analysis revealed that Cv-aCO2/Da-vO2 ratio at both T0 (RR 3.85; 95 % CI 1.60–9.27) and T6 (RR 3.97; 95 % CI 1.54–10.24) was an independent predictor for mortality at day 28, as well as lactate levels at T6 (RR 1.58; 95 % CI 1.13–2.22).
Conclusion
Complementing lactate assessment with Cv-aCO2/Da-vO2 ratio during early stages of resuscitation of septic shock can better identify patients at high risk of adverse outcomes. The Cv-aCO2/Da-vO2 ratio may become a potential resuscitation goal in patients with septic shock.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Early identification of tissue hypoperfusion and adequate resuscitation are key factors in the management of patients with shock [1, 2]. Although early resuscitation seems to improve outcomes in severe sepsis and septic shock, the relative value of resuscitation goals continues to be highly debated [3–6]. Monitoring of ScvO2 is widely recommended [3–7] although strongly challenged by others [8, 9]. In an early trial, Rivers et al. [3] observed a significant decrease in mortality when they used a resuscitation bundle targeting ScvO2 >70 %. Conversely, recent data failed to confirm any benefit with this approach [10]. However, it should be noted that ScvO2 was normal or near normal at inclusion in a number of patients in these trials [10] as it has frequently been reported on admissions to the intensive care unit in studies subsequent to the River’s trial [11]. Moreover, normalization of systemic hemodynamic and oxygen metabolism variables does not ensure an adequate tissue perfusion and does not prevent progression to multiorgan dysfunction and death [12]. Lactate has also been proposed as a target for resuscitation therapy. In fact, not only baseline lactate level [13] but also its evolution under the influence of therapy [14] has been associated with clinical outcomes. Despite promising results observed in one trial [15], no consistent advantages have been found for lactate-based resuscitation bundles over resuscitation guided by oxygen parameters [15–17]. Accordingly, additional markers of inadequate perfusion should be explored, especially when ScvO2 values are close to normal.
Recently, the venous-to-arterial carbon dioxide difference (Pv-aCO2) has been proposed as an alternative marker of tissue hypoperfusion [18, 19]. In fact, persistently high Pv-aCO2 predicts adverse clinical outcomes independently of oxygen-derived parameters and it could anticipate lactate variations [20]. However, the Pv-aCO2 may be normal despite the presence of significant hypoperfusion in high cardiac output states such as septic shock, where high flows might prevent venous CO2 accumulation; or inversely, Pv-aCO2 can increase in the absence of hypoperfusion, in part due to the Haldane effect [21]. Consequently, CO2 variations must be evaluated according to O2 changes. Indeed, CO2 production should not exceed O2 availability during aerobic metabolism. Thus, the ratio between the Pv-aCO2 and the arterial-to-venous oxygen content difference (Da-vO2), as a surrogate of the VCO2/VO2 ratio (i.e., the respiratory quotient), may identify patients at risk of anaerobic metabolism. Using this rationale, Mekontso-Dessap et al. [22] demonstrated that a Pv-aCO2 to Da-vO2 ratio >1.4 was superior to Pv-aCO2, SvO2, and Da-vO2 in predicting hyperlactatemia in a cohort of critically ill patients. Importantly, Pv-aCO2/Da-vO2 ratio variations are faster than lactate kinetics, which make it an attractive variable to monitor. However, CO2 partial pressure (PCO2) is not equivalent to CO2 content (CCO2), particularly when O2 saturation varies (Haldane effect). Thus, Cv-aCO2/Da-vO2 variations should better reflect variations in VO2 than Pv-aCO2/Da-vO2, especially when ScvO2 or SvO2 is low.
As the Cv-aCO2 to Da-vO2 ratio could reflect ongoing anaerobic metabolism, we hypothesized that an increased Cv-aCO2/Da-vO2 could be used to identify patients at risk of adverse outcomes during early stages of septic shock and that this variable could provide additional information when combined with lactate levels.
Materials and methods
We conducted a prospective observational study in a 60-bed mixed ICU in a university-affiliated hospital. The Fundación Valle del Lili’s ethical and biomedical research committee approved the current study (protocol number 710; approval number 093-2014). A written informed consent was waived because all measurements and procedures routinely followed the local protocols for the management of severe sepsis and septic shock and no new therapeutic interventions were performed. Our “rapid-response team” evaluated all patients with suspected septic shock at the emergency room and clinical wards. Resuscitation was immediately started and these patients were rapidly admitted to the ICU. Presence of infection was established using the Centers for Diseases Control and Prevention criteria [23] and septic shock was defined according to the criteria of the American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference [24]. Antibiotics were started within 1 h of diagnosis of sepsis. An arterial catheter was placed in the radial or femoral artery while a pulmonary artery catheter (PAC) was inserted via the jugular or subclavian vein when deemed appropriate according to clinical judgment and local indications for the use of hemodynamic monitoring. All patients with a new episode of septic shock and equipped with a PAC were included for the study. Patients with a previous episode of severe sepsis or septic shock within the last 3 months, younger than 18 years old, pregnant women, with limitations of care, with liver cirrhosis Child–Pugh C, or severe chronic obstructive pulmonary disease were excluded.
General management
All patients followed an early quantitative resuscitation protocol adapted from the Surviving Sepsis Campaign [7] in order to target (a) mean arterial pressure (MAP) ≥65 mmHg; (b) urine output ≥0.5 mL/kg/min; (c) SvO2 ≥65 %; (d) normalization of lactate levels. In the case of attaining the SvO2 goal but with persistently high lactate levels, additional efforts were performed to attain normalization of the latter. Pulse pressure variations were used to indicate fluid responsiveness whenever applicable. In other cases, filling pressures and clinical judgment were used. Fluid resuscitation was conducted by repeated fluid challenges with crystalloids and/or albumin 4 %. Hydroxyethyl starches (HES) were not used.
Norepinephrine was the first-choice vasopressor to maintain MAP goals. Vasopressin titrated to a maximum of 0.03 UI/min was allowed in order to raise MAP or to decrease norepinephrine dose but never as a single vasopressor. Titrated dobutamine up to 20 μg/kg/min was used when myocardial dysfunction was demonstrated or when SvO2 goals were not achieved despite adequate intravascular volume and MAP. Mechanical ventilation was provided (when needed) under light sedation (midazolam) and analgesia (fentanyl, morphine); tidal volume was limited to 6–8 mL/kg. Low dose hydrocortisone was indicated if vasopressor requirement did not decrease during the first 6 h of resuscitation despite ensuring adequate intravascular volume. Glycemic control was adjusted to maintain glucose levels <150 mg/dL. Finally, stress ulcer and venous thrombosis prophylaxis were provided according to international recommendations [7].
Study protocol
Time 0 (T0) was stated at the PAC insertion. We recorded the total volume of fluids received and the time elapsed between the first episode of hypotension and T0. We performed complete hemodynamic measurements and drew blood samples for arterial and mixed-venous gases analysis (ABL 300, Radiometer Copenhagen, Denmark) and arterial lactate at T0, and 6 h (T6), around 12 h (T12), and 24 h (T24) after. Vasopressors and inotropic doses, respiratory parameters, and total fluids were also registered at each measurement time. Organ dysfunction at day 3 was evaluated using the Sequential Organ Failure Assessment (SOFA) score [25]. We also calculated the ventilator-free days and survival at day 28.
Carbon dioxide and oxygen variables
We calculated CO2 and O2 variables at T0, T6, T12, and T24, as follows:
-
DO2 = CaO2 × CI
-
VO2 = (CaO2 − CvO2) × CI
-
ERO2 = (CaO2 − CvO2)/CaO2
-
CaO2 = (Hg × SaO2 × 1.34) + (PaO2 × 0.003)
-
CvO2 = (Hg × SvO2 × 1.34) + (PvO2 × 0.003)
-
Pv-aCO2 = PvCO2 − PaCO2
-
Da-vO2 = CaO2 − CvO2
where CaO2 and CvO2 are the arterial and venous O2 content, PaO2 and PvO2 represent their arterial and venous partial pressures respectively, CI represents the cardiac index, and ERO2 represents the oxygen extraction ratio.
We also calculated CO2 contents according to the Douglas formula [26]:
where plasma CCO2 = 2.226 × S × plasma PCO2 × (1 + 10pH − pK′); In turn, S (plasma CO2 solubility) and apparent pK (pK′) are temperature (T, expressed as °C) dependent and calculated according to previous calculations [27]:
Definitions of the four groups
Considering that in aerobic conditions VCO2 should not exceed VO2, we considered a Cv-aCO2/Da-vO2 ratio >1.0 as abnormal. Hence, we analyzed hemodynamic and oxygen metabolism parameters for four predetermined groups according to lactate levels and Cv-aCO2/Da-vO2 attained after the first 6 h of resuscitation: group 1, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 ratio >1.0; group 2, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 ratio ≤1.0; group 3, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 ratio >1.0; and group 4, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 ratio ≤1.0.
Statistics
After demonstrating a non-normal distribution of data with a Kolmogorov–Smirnoff test, we used a Kruskal–Wallis test to compare continuous variables with a Tukey–Kramer test for multiple comparisons among the predefined groups. χ 2 test was used to compare discrete variables (or Fisher’s exact test, when appropriate). Multiorgan dysfunction at day 3 was evaluated using the Sequential Organ Failure Assessment (SOFA) score [25] for the predefined groups. Survival curves up to day 28 were estimated using the Kaplan–Meier method and log-rank (Mantel–Cox) test was used to estimate differences among the predefined groups.
In a further analysis, variables were introduced into a multivariate model if significantly associated with mortality at day 28 at the univariate analysis when a p value was <0.2. General demographics, hemodynamics, vasopressor use, fluids, and blood gases parameters at T0 and T6 were used in the model, previously testing for collinearity. These analyses were also conducted in those patients attaining SvO2 ≥65 %. A Hosmer and Lemeshow test was used to assess the goodness of fit of the model. Receiver operating characteristic (ROC) curves for the original model (i.e., the “large model”) and a second one excluding the Cv-aCO2/Da-vO2 ratio (i.e., the “short model”) were constructed in order to test the added value of the Cv-aCO2/Da-vO2 ratio in predicting mortality at day 28. The ROC curves were compared using the method described by DeLong and colleagues [28].
Additional logistic regression models including Pv-aCO2/Da-vO2 instead of Cv-aCO2/Da-vO2 and another one including simultaneously Pv-aCO2/Da-vO2 and Cv-aCO2/Da-vO2 were also conducted to explore its relationship with mortality at day 28.
Finally, we described the time-course for oxygen metabolism variables, Pv-aCO2/Da-vO2 and Cv-aCO2/Da-vO2 during the first 24 h for both survivors and non-survivors at day 28.
Data are presented as median (25–75th percentiles). Risk assessments are presented as risk ratios with 95 % confidence intervals. A p value ≤0.05 (two-tailed) was considered significant.
Results
Selection of patients is shown in the Fig. 1 of the Electronic Supplementary Material (ESM). A total of 135 patients were included in the study during a period of 18 months. Mortality at day 28 in this cohort was 42 % and ICU length of stay was 6 (2–10) days. The time from first hypotension episode to catheter insertion and blood sampling (i.e., T0) was 3.0 (2.5–4.0) h and the median amount of fluids received before T0 was 1,977 (1,200–2,800) mL.
After the first 6 h of resuscitation, 110 (81 %) patients achieved a MAP >65 mmHg and 98 (73 %) a SvO2 >65 %. However, 84 (62 %) patients still had an arterial lactate ≥2.0 mmol/L and 65 (48 %) had a Cv-aCO2/Da-vO2 ratio >1.0. Accordingly, 42 patients were classified into group 1, 42 into group 2, 23 into group 3, and 28 into group 4. Patients in groups 1 and 2 had higher APACHE II scores and required higher vasopressor doses at T0 (Table 1). No significant differences were found in demographic data or other hemodynamic variables at T0 (Table 1 and ESM Table 1). All hemodynamic, blood gases, oxygen parameters, and ventilator settings at both T0 and T6 are presented in the ESM Table 1. Patients from groups 1 and 2 had more acidosis at T0 and T6. Regarding the clinical outcomes, patients from group 1 evolved with higher SOFA scores (Kruskal–Wallis, p < 0.001; post hoc test demonstrated significant differences among groups 1 vs. 3 and 1 vs. 4) (Fig. 1) and they also had the lowest survival rates at day 28 [log rank (Mantel–Cox) = 31.39, p < 0.0001] (Fig. 2). Intriguingly, patients in groups 2 and 3 had similar SOFA scores and outcomes at day 28 (Table 2). Furthermore, patients from group 1 had the lowest VO2 at T6 and T12 compared to all other groups, even though cardiac output, SvO2, and DO2 were not different (ESM Fig. 2).
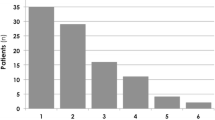
Sequential Organ Failure Assessment (SOFA) scores at day 3 for predefined groups. Data presented as median (percentiles). Patients were separated into four groups according to lactate and Cv-aCO2/Da-vO2 ratio measured after the first 6 h of resuscitation: group 1, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 ratio >1.0; group 2, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 ratio ≤1.0; group 3, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 ratio >1.0; and group 4, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 ratio ≤1.0. Kruskal–Wallis one-way ANOVA, p < 0.001. **p < 0.01 by Tukey–Kramer showing differences between groups 1 vs. 3 and 1 vs. 4
Survival probabilities up to day 28 according to lactate and Cv-aCO2/Da-vO2 after 6 h of resuscitation. Log rank (Mantel–Cox) = 31.39, p < 0.0001. Group 1, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 ratio >1.0; group 2, lactate ≥2.0 mmol/L and Cv-aCO2/Da-vO2 ratio ≤1.0; group 3, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 ratio >1.0; and group 4, lactate <2.0 mmol/L and Cv-aCO2/Da-vO2 ratio ≤1.0
Multivariate logistic regression analysis at T0 demonstrated that Cv-aCO2/Da-vO2 was an independent predictor of mortality at day 28 (RR 3.85; 95 % CI 1.60–9.27). When analysis was performed using the same variables at T6, Cv-aCO2/Da-vO2 was again related to higher mortality at day 28 (RR 3.97; 95 % CI 1.54–10.24), in addition to lactate levels (RR 1.58; 95 % CI 1.13–2.22) (Table 3). An additional multivariate analysis performed in patients attaining SvO2 >65 % showed that lactate levels (RR 2.41; 95 % CI 1.22–4.76) and Cv-aCO2/Da-vO2 (RR 5.71; 95 % CI 1.20–27.19) remained predictors of mortality at day 28 (Table 3). The area under ROC curves for models including or excluding the Cv-aCO2/Da-vO2 ratio (i.e., the “large” and “short” model, respectively) were significantly different (AUClarge 0.8542, 95 % CI 0.7797–0.9286 vs. AUCshort 0.7943, 95 % CI 0.7050–0.8836. LR test, χ 2 17.81, p < 0.001; C statistic, χ 2 4.52, p = 0.03) (Fig. 3).
Receiver operating characteristics (ROC) curves for prediction of mortality at day 28 for models including or not Cv-aCO2/Da-vO2 ratio. The “large model” included Cv-aCO2/Da-vO2 + lactate levels. The “short model” included lactate levels but not Cv-aCO2/Da-vO2. Both models also included oxygen consumption (VO2), oxygen delivery (DO2), mixed-venous oxygen saturation (SvO2), cardiac index, APACHE II, age, time before T0, gender, fluids administered, norepinephrine dose, and mean arterial pressure. Likelihood ratio test, χ 2 = 17.81, p < 0.001. Differences between AUCs, C statistic, χ 2 4.52, p = 0.03
We also found significant differences in the time-course of Pv-aCO2, Pv-aCO2/Da-vO2, lactate levels, and Cv-aCO2/Da-vO2 during the first 24 h of resuscitation between survivors and non-survivors at day 28 (repeated measurements ANOVA, p < 0.05) (ESM Figs. 3–5).
Discussion
We observed that persistent hyperlactatemia combined with a high Cv-aCO2/Da-vO2 was associated with the most severe organ dysfunction and worst clinical outcomes, while simultaneous normalization of lactate and Cv-aCO2/Da-vO2 ratio was associated with the best outcomes. Interestingly, patients attaining lactate levels <2.0 mmol/L combined with Cv-aCO2/Da-vO2 >1.0 had similar outcomes to patients with persistent hyperlactatemia and low Cv-aCO2/Da-vO2 ratio.
We hypothesized that a Cv-aCO2/Da-vO2 >1.0 reflects anaerobic metabolism as VCO2 should not be higher than VO2 during aerobic conditions. Indeed, occurrence of a high VCO2/VO2 has been previously reported in experimental conditions, where lower reductions in VCO2 than in VO2 have been associated with other markers of tissue hypoxia, suggesting the involvement of a non-aerobic source of CO2 [29, 30]. Consequently, a Cv-aCO2/Da-vO2 ratio >1.0 (as a surrogate of the VCO2/VO2 ratio) could identify an excess of CO2 generation probably due to anaerobic metabolism and this condition could be associated with more unfavorable clinical outcomes as we report in our study. Using a similar rationale, Mekontso-Dessap et al. [22] tested the hypothesis that Pv-aCO2/Da-vO2 better detects anaerobic metabolism than other parameters derived from PAC measurements in critically ill patients. They found a significant agreement between Pv-aCO2/Da-vO2 and lactate levels. However, the agreement between Pv-aCO2/Da-vO2 and lactate levels should not necessarily be considered as representative of anaerobic metabolism since hyperlactatemia is not always of hypoxic origin [31, 32]. Interestingly, our data demonstrated that a high Cv-aCO2/Da-vO2 might be present with normal or high lactate levels suggesting that these variables evolve independently probably because lactate kinetics can be slower than Cv-aCO2/Da-vO2 variations. Thus, in the presence of hyperlactatemia a high Cv-aCO2/Da-vO2 may favor anaerobic metabolism as the possible source of lactate, while a normal Cv-aCO2/Da-vO2 may suggest that lactate accumulation is due to other causes [33–36].
Searching for other markers of ongoing tissue hypoxia could increase the information given by lactate levels during resuscitation of septic shock. Recently, Rimachi et al. [37] reported the presence of hyperlactatemia in 65 % of patients with septic shock, but only 75 % of these patients exhibited increased lactate/pyruvate ratio, confirming that hyperlactatemia may be not due to hypoxia, especially during the early stages of shock. Consistent with that study, 71 % of the patients in our study had hyperlactatemia at T0 and half of them had an elevated Cv-aCO2/Da-vO2. Interestingly, hyperlactatemic patients evolving with a high Cv-aCO2/Da-vO2 at T6 (i.e., after initial resuscitation) had a lower VO2 compared with those evolving with normal Cv-aCO2/Da-vO2, despite similar DO2 values. This suggests that a high Cv-aCO2/Da-vO2 coupled with hyperlactatemia could identify ongoing VO2/DO2 dependence. In agreement with this concept, Monnet et al. [38] recently reported that VO2 increased after fluid administration only in patients with a pre-fluid high Pv-aCO2/DavO2 ratio. In other words, a VCO2/VO2 ratio estimated by the Pv-aCO2/DavO2 or the Cv-aCO2/DavO2 could be used to predict fluid responsiveness at the tissue level.
Intriguingly, both Cv-aCO2/Da-vO2 and lactate levels were independent factors determining clinical outcomes, at T0 and T6. As expected, patients with combined increase in Cv-aCO2/Da-vO2 and lactate had the worse outcome, while patients with both variables normal had the best outcomes. Interestingly, patients attaining normal lactate levels at T6 but with a high Cv-aCO2/Da-vO2 had a similar incidence of multiorgan dysfunction and unfavorable clinical outcomes as hyperlactatemic patients with a Cv-aCO2/Da-vO2 ≤1.0. This further emphasizes the additive value of both indices.
Recent human studies suggest that Pv-aCO2 may identify persistent perfusion derangements in apparently resuscitated septic shock patients [18, 20, 39]. The simplicity of Pv-aCO2 measurement makes it an attractive tool to guide resuscitation in the clinical setting. However, the Pv-aCO2 is a physiologically complex measurement, as the relationship between the CO2 partial pressure (PCO2) and the CO2 content (CCO2) is affected by O2 saturation, i.e., the Haldane effect [21]. Accordingly, clinical interpretation of the Pv-aCO2 can be difficult since its increase can be observed in both aerobic and anaerobic conditions.
Another important question is whether Pv-aCO2/Da-vO2 can be used as a surrogate of Cv-aCO2/Da-vO2. This approach, used by several investigators [22, 38], assumes that CO2 partial pressure (PCO2) keeps a quasi-linear relationship with CO2 content (CCO2) over the physiological range of PCO2, i.e., along the steep portion of the CO2 dissociation curve. However, the relationship between PCO2 and CCO2 becomes non-linear if oxygen saturation, arterial-venous pH difference, and/or hemoglobin concentrations change. In this respect, several studies [21, 40] reported dissociation between CCO2 and PCO2 in the splanchnic region when CCO2 decreased in the venous splanchnic effluent while PCO2 paradoxically increased during increases of splanchnic blood flow. In fact, they showed that venous to arterial PCO2 differences could increase or decrease for identical blood flow increases. Thus, depending on the basal venous oxygen saturation, the Haldane effect may cause a decrease or increase in the respective venous to arterial PCO2 difference in response to the same changes in blood flow and metabolism [40]. Thus, theoretically Cv-aCO2/Da-vO2 is not equivalent to Pv-aCO2/Da-vO2 especially during low PCO2 and SvO2 conditions. Importantly, despite the similarities in the time-course of the Pv-aCO2/Da-vO2 and the Cv-aCO2/Da-vO2 (ESM Figs. 3 and 4), we did not find significant association with day-28 mortality for the Pv-aCO2/Da-vO2 when it was included instead of Cv-aCO2/Da-vO2 in the logistic regression (ESM Table 2). An additional analysis simultaneously including the Pv-aCO2/Da-vO2 and the Cv-aCO2/Da-vO2 demonstrated that despite the former being significantly associated with mortality in the univariate analysis, it was not maintained in the multivariate analysis (ESM Table 3). Nevertheless, despite having ruled out mathematical collinearity between Pv-aCO2/Da-vO2 and Cv-aCO2/Da-vO2 in the model, it would be debatable to refuse that any collinearity might exist between two variables tightly related. However, we admit that Pv-aCO2/Da-vO2 could be equivalent to Cv-aCO2/Da-vO2 when PCO2, pH, and SvO2 approximate to normality, which occurs frequently. Cv-aCO2/DavO2 is an approximation of respiratory quotient, and thus it has a strong physiological meaning. Even though it is more complicated to compute, it is easier to interpret, with values above 1 suggesting anaerobic metabolism. Admittedly, computations of CO2 content and DavO2 are cumbersome and subject to an important risk of errors due to the number of variables included in the formulas. Nevertheless, our data suggest that the influence of measurement errors is limited as it correctly identified patients at increased risk of death.
Nowadays critically ill patients admitted to the ICU often exhibit normal or near-normal venous oxygen saturations [11]. Interestingly, when we studied only the patients attaining SvO2 >65 %, Cv-aCO2/Da-vO2 and lactate levels were still independent predictors of outcomes. Thus, Cv-aCO2/Da-vO2 could be a useful resuscitation variable in both low and normal SvO2 conditions.
We acknowledge some limitations of our study. First, both Cv-aCO2 and Da-vO2 are global variables and they may not represent regional or local perfusion derangements. Thus, tissue hypoperfusion inducing local CO2 accumulation may occur even when systemic venous CO2 remains normal. Second, the Cv-aCO2 may not increase during conditions of tissue hypoxia associated with high blood flow, even if CO2 production is increased due to anaerobic metabolism, as venous blood flow may be sufficient to wash out the CO2 generated by hypoxic cells [41]. In these patients, the combination with lactate levels is useful to overcome this shortcoming. Finally, our observations were restricted to a relatively small sample of septic shock patients and although our results sound biologically plausible, they should be confirmed in future physiological studies to better understand the significance of the Cv-aCO2/Da-vO2 ratio during early stages of septic shock.
Conclusion
Combination of Cv-aCO2/Da-vO2 and lactate measurements at early stages of resuscitation can identify risk of adverse outcomes in septic shock. The Cv-aCO2/Da-vO2 ratio may become a potential resuscitation goal in patients with septic shock.
References
Vincent JL, De Backer D (2013) Circulatory shock. N Engl J Med 369:1726–1734
Cecconi M, De Backer D, Antonelli M, Beale R, Bakker J, Hofer C, Jaeschke R, Mebazaa A, Pinsky MR, Teboul JL, Vincent JL, Rhodes A (2014) Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med 40:1795–1815
Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, Peterson E, Tomlanovich M, Group EG-DTC (2001) Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 345:1368–1377
Trzeciak S, Dellinger RP, Abate NL, Cowan RM, Stauss M, Kilgannon JH, Zanotti S, Parrillo JE (2006) Translating research to clinical practice: a 1-year experience with implementing early goal-directed therapy for septic shock in the emergency department. Chest 129:225–232
Jones AE, Focht A, Horton JM, Kline JA (2007) Prospective external validation of the clinical effectiveness of an emergency department-based early goal-directed therapy protocol for severe sepsis and septic shock. Chest 132:425–432
Shapiro NI, Howell MD, Talmor D, Lahey D, Ngo L, Buras J, Wolfe RE, Weiss JW, Lisbon A (2006) Implementation and outcomes of the Multiple Urgent Sepsis Therapies (MUST) protocol. Crit Care Med 34:1025–1032
Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart K, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb S, Beale RJ, Vincent JL, Moreno R, Subgroup SSCGCiTP (2013) Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med 39:165–228
Bellomo R, Reade MC, Warrillow SJ (2008) The pursuit of a high central venous oxygen saturation in sepsis: growing concerns. Crit Care 12:130
Perel A (2008) Bench-to-bedside review: the initial hemodynamic resuscitation of the septic patient according to Surviving Sepsis Campaign guidelines—does one size fit all? Crit Care 12:223
Yealy DM, Kellum JA, Huang DT, Barnato AE, Weissfeld LA, Pike F, Terndrup T, Wang HE, Hou PC, LoVecchio F, Filbin MR, Shapiro NI, Angus DC, Investigators P (2014) A randomized trial of protocol-based care for early septic shock. N Engl J Med 370:1683–1693
van Beest PA, Hofstra JJ, Schultz MJ, Boerma EC, Spronk PE, Kuiper MA (2008) The incidence of low venous oxygen saturation on admission to the intensive care unit: a multi-center observational study in the Netherlands. Crit Care 12:R33
Puskarich MA, Trzeciak S, Shapiro NI, Heffner AC, Kline JA, Jones AE, Emergency Medicine Shock Research Network (EMSHOCKNET) (2011) Outcomes of patients undergoing early sepsis resuscitation for cryptic shock compared with overt shock. Resuscitation 82:1289–1293
Shapiro NI, Howell MD, Talmor D, Nathanson LA, Lisbon A, Wolfe RE, Weiss JW (2005) Serum lactate as a predictor of mortality in emergency department patients with infection. Ann Emerg Med 45:524–528
Puskarich MA, Trzeciak S, Shapiro NI, Albers AB, Heffner AC, Kline JA, Jones AE (2013) Whole blood lactate kinetics in patients undergoing quantitative resuscitation for severe sepsis and septic shock. Chest 143:1548–1553
Jansen TC, van Bommel J, Schoonderbeek FJ, Sleeswijk Visser SJ, van der Klooster JM, Lima AP, Willemsen SP, Bakker J, group Ls (2010) Early lactate-guided therapy in intensive care unit patients: a multicenter, open-label, randomized controlled trial. Am J Respir Crit Care Med 182:752–761
Nguyen HB, Kuan WS, Batech M, Shrikhande P, Mahadevan M, Li CH, Ray S, Dengel A, Investigators AANtRSc (2011) Outcome effectiveness of the severe sepsis resuscitation bundle with addition of lactate clearance as a bundle item: a multi-national evaluation. Crit Care 15:R229
Jones AE, Shapiro NI, Trzeciak S, Arnold RC, Claremont HA, Kline JA, Investigators EMSRNE (2010) Lactate clearance vs central venous oxygen saturation as goals of early sepsis therapy: a randomized clinical trial. JAMA 303:739–746
van Beest PA, Lont MC, Holman ND, Loef B, Kuiper MA, Boerma EC (2013) Central venous-arterial PCO2 difference as a tool in resuscitation of septic patients. Intensive Care Med 39:1034–1039
Vallet B, Pinsky MR, Cecconi M (2013) Resuscitation of patients with septic shock: please “mind the gap”! Intensive Care Med 39:1653–1655
Ospina-Tascón GA, Bautista-Rincón DF, Umaña M, Tafur JD, Gutiérrez A, García AF, Bermúdez W, Granados M, Arango-Dávila C, Hernández G (2013) Persistently high venous-to-arterial carbon dioxide differences during early resuscitation are associated with poor outcomes in septic shock. Crit Care 17:R294
Jakob SM, Kosonen P, Ruokonen E, Parviainen I, Takala J (1999) The Haldane effect—an alternative explanation for increasing gastric mucosal PCO2 gradients? Br J Anaesth 83:740–746
Mekontso-Dessap A, Castelain V, Anguel N, Bahloul M, Schauvliege F, Richard C, Teboul JL (2002) Combination of venoarterial PCO2 difference with arteriovenous O2 content difference to detect anaerobic metabolism in patients. Intensive Care Med 28:272–277
Horan TC, Andrus M, Dudeck MA (2008) CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 36:309–332
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, Cohen J, Opal SM, Vincent JL, Ramsay G, SCCM, ESICM, ACCP, ATS, SIS (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med 31:1250–1256
Vincent JL, de Mendonça A, Cantraine F, Moreno R, Takala J, Suter PM, Sprung CL, Colardyn F, Blecher S (1998) Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: results of a multicenter, prospective study. Working group on “sepsis-related problems” of the European Society of Intensive Care Medicine. Crit Care Med 26:1793–1800
Douglas AR, Jones NL, Reed JW (1985) Calculation of whole blood CO2 content. J Appl Physiol 65:473–477
Austin WH, Lacombe E, Rand PW, Chatterjee M (1963) Solubility of carbon dioxide in serum from 15 to 38 C. J Appl Physiol 18:301–304
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
Groeneveld AB, Vermeij CG, Thijs LG (1991) Arterial and mixed venous blood acid-base balance during hypoperfusion with incremental positive end-expiratory pressure in the pig. Anesth Analg 73:576–582
Dubin A, Murias G, Estenssoro E, Canales H, Sottile P, Badie J, Barán M, Rossi S, Laporte M, Pálizas F, Giampieri J, Mediavilla D, Vacca E, Botta D (2000) End-tidal CO2 pressure determinants during hemorrhagic shock. Intensive Care Med 26:1619–1623
James JH, Luchette FA, McCarter FD, Fischer JE (1999) Lactate is an unreliable indicator of tissue hypoxia in injury or sepsis. Lancet 354:505–508
Levraut J, Ciebiera JP, Chave S, Rabary O, Jambou P, Carles M, Grimaud D (1998) Mild hyperlactatemia in stable septic patients is due to impaired lactate clearance rather than overproduction. Am J Respir Crit Care Med 157:1021–1026
Severin PN, Uhing MR, Beno DW, Kimura RE (2002) Endotoxin-induced hyperlactatemia results from decreased lactate clearance in hemodynamically stable rats. Crit Care Med 30:2509–2514
De Backer D, Creteur J, Zhang H, Norrenberg M, Vincent JL (1997) Lactate production by the lungs in acute lung injury. Am J Respir Crit Care Med 156:1099–1104
Vallet B, Teboul JL, Cain S, Curtis S (2000) Venoarterial CO(2) difference during regional ischemic or hypoxic hypoxia. J Appl Physiol 89:1317–1321
Nevière R, Chagnon JL, Teboul JL, Vallet B, Wattel F (2002) Small intestine intramucosal PCO(2) and microvascular blood flow during hypoxic and ischemic hypoxia. Crit Care Med 30:379–384
Rimachi R, Bruzzi de Carvahlo F, Orellano-Jimenez C, Cotton F, Vincent JL, De Backer D (2012) Lactate/pyruvate ratio as a marker of tissue hypoxia in circulatory and septic shock. Anaesth Intensive Care 40:427–432
Monnet X, Julien F, Ait-Hamou N, Lequoy M, Gosset C, Jozwiak M, Persichini R, Anguel N, Richard C, Teboul JL (2013) Lactate and venoarterial carbon dioxide difference/arterial-venous oxygen difference ratio, but not central venous oxygen saturation, predict increase in oxygen consumption in fluid responders. Crit Care Med 41:1412–1420
Vallée F, Vallet B, Mathe O, Parraguette J, Mari A, Silva S, Samii K, Fourcade O, Genestal M (2008) Central venous-to-arterial carbon dioxide difference: an additional target for goal-directed therapy in septic shock? Intensive Care Med 34:2218–2225
Hurley R, Mythen MG (2000) The Haldane effect—an explanation for increasing gastric mucosal PCO2 gradients? Br J Anaesth 85:167–169
Dubin A, Estenssoro E, Murias G, Pozo MO, Sottile JP, Barán M, Piacentini E, Canales HS, Etcheverry G (2004) Intramucosal-arterial PCO2 gradient does not reflect intestinal dysoxia in anemic hypoxia. J Trauma 57:1211–1217
Acknowledgments
The authors kindly thank Dr. Jairo Osorno and Dr. Fernando Rosso (Centro de Investigaciones Clínicas, Fundación Valle del Lili-Universidad ICESI, Cali, Colombia) for their contribution to enhancing the manuscript quality and Dr. Yuri Takeuchi (Fundación Valle del Lili-Universidad ICESI) for her unconditional support to the research.
Conflicts of interest
The authors declare no conflict of interest for the current study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Take-home message: The ratio between the Cv-aCO2 and the arterial-to-venous oxygen content difference (Da-vO2), as a surrogate of the VCO2/VO2 ratio (i.e., the respiratory quotient), may identify patients at risk of anaerobic metabolism.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Ospina-Tascón, G.A., Umaña, M., Bermúdez, W. et al. Combination of arterial lactate levels and venous-arterial CO2 to arterial-venous O2 content difference ratio as markers of resuscitation in patients with septic shock. Intensive Care Med 41, 796–805 (2015). https://doi.org/10.1007/s00134-015-3720-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-015-3720-6