Abstract
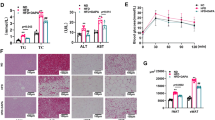
Endothelial cells (ECs) within the microvasculature of brown adipose tissue (BAT) are important in regulating the plasticity of adipocytes in response to increased metabolic demand by modulating the angiogenic response. However, the mechanism of EC-adipocyte crosstalk during this process is not completely understood. We used RNA sequencing to profile microRNAs derived from BAT ECs of obese mice and identified an anti-angiogenic microRNA, miR-409-3p. MiR-409-3p overexpression inhibited EC angiogenic properties; whereas, its inhibition had the opposite effects. Mechanistic studies revealed that miR-409-3p targets ZEB1 and MAP4K3. Knockdown of ZEB1/MAP4K3 phenocopied the angiogenic effects of miR-409-3p. Adipocytes co-cultured with conditioned media from ECs deficient in miR-409-3p showed increased expression of BAT markers, UCP1 and CIDEA. We identified a pro-angiogenic growth factor, placental growth factor (PLGF), released from ECs in response to miR-409-3p inhibition. Deficiency of ZEB1 or MAP4K3 blocked the release of PLGF from ECs and PLGF stimulation of 3T3-L1 adipocytes increased UCP1 expression in a miR-409-3p dependent manner. MiR-409-3p neutralization improved BAT angiogenesis, glucose and insulin tolerance, and energy expenditure in mice with diet-induced obesity. These findings establish miR-409-3p as a critical regulator of EC-BAT crosstalk by modulating a ZEB1-MAP4K3-PLGF signaling axis, providing new insights for therapeutic intervention in obesity.








Similar content being viewed by others
Availability of data and material
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
Code availability
Not applicable.
Abbreviations
- SiRNA:
-
Small interfering RNA
- 3’-UTR:
-
3’-Untranslated region
- ZEB1:
-
Zinc Finger E-Box Binding Homeobox
- MAP4K3:
-
Mitogen-activated protein kinase kinase kinase kinase 3
- UCP1:
-
Uncoupling Protein 1
References
Saltiel AR, Olefsky JM (2017) Inflammatory mechanisms linking obesity and metabolic disease. J Clin Invest 127(1):1–4. https://doi.org/10.1172/JCI92035
Dall TM, Yang W, Halder P, Pang B, Massoudi M, Wintfeld N et al (2014) The economic burden of elevated blood glucose levels in 2012: diagnosed and undiagnosed diabetes, gestational diabetes mellitus, and prediabetes. Diabetes Care 37(12):3172–3179. https://doi.org/10.2337/dc14-1036
Swinburn BA, Sacks G, Hall KD, McPherson K, Finegood DT, Moodie ML et al (2011) The global obesity pandemic: shaped by global drivers and local environments. Lancet 378(9793):804–814. https://doi.org/10.1016/S0140-6736(11)60813-1
Smorlesi A, Frontini A, Giordano A, Cinti S (2012) The adipose organ: white-brown adipocyte plasticity and metabolic inflammation. Obes Rev 13(Suppl 2):83–96. https://doi.org/10.1111/j.1467-789X.2012.01039.x
Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB et al (2009) Identification and importance of brown adipose tissue in adult humans. N Engl J Med 360(15):1509–1517. https://doi.org/10.1056/NEJMoa0810780
Virtanen KA, Lidell ME, Orava J, Heglind M, Westergren R, Niemi T et al (2009) Functional brown adipose tissue in healthy adults. N Engl J Med 360(15):1518–1525. https://doi.org/10.1056/NEJMoa0808949
Saito M, Okamatsu-Ogura Y, Matsushita M, Watanabe K, Yoneshiro T, Nio-Kobayashi J et al (2009) High incidence of metabolically active brown adipose tissue in healthy adult humans: effects of cold exposure and adiposity. Diabetes 58(7):1526–1531. https://doi.org/10.2337/db09-0530
Crewe C, An YA, Scherer PE (2017) The ominous triad of adipose tissue dysfunction: inflammation, fibrosis, and impaired angiogenesis. J Clin Invest 127(1):74–82. https://doi.org/10.1172/JCI88883
Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L (2006) VEGF receptor signaling—in control of vascular function. Nat Rev Mol Cell Biol 7(5):359–371. https://doi.org/10.1038/nrm1911
Rausch ME, Weisberg S, Vardhana P, Tortoriello DV (2008) Obesity in C57BL/6J mice is characterized by adipose tissue hypoxia and cytotoxic T-cell infiltration. Int J Obes (Lond) 32(3):451–463. https://doi.org/10.1038/sj.ijo.0803744
Yin J, Gao Z, He Q, Zhou D, Guo Z, Ye J (2009) Role of hypoxia in obesity-induced disorders of glucose and lipid metabolism in adipose tissue. Am J Physiol Endocrinol Metab 296(2):E333–E342. https://doi.org/10.1152/ajpendo.90760.2008
Sun K, Wernstedt Asterholm I, Kusminski CM, Bueno AC, Wang ZV, Pollard JW et al (2012) Dichotomous effects of VEGF-A on adipose tissue dysfunction. Proc Natl Acad Sci USA 109(15):5874–5879. https://doi.org/10.1073/pnas.1200447109
Gealekman O, Guseva N, Hartigan C, Apotheker S, Gorgoglione M, Gurav K et al (2011) Depot-specific differences and insufficient subcutaneous adipose tissue angiogenesis in human obesity. Circulation 123(2):186–194. https://doi.org/10.1161/CIRCULATIONAHA.110.970145
O’Rourke RW, White AE, Metcalf MD, Olivas AS, Mitra P, Larison WG et al (2011) Hypoxia-induced inflammatory cytokine secretion in human adipose tissue stromovascular cells. Diabetologia 54(6):1480–1490. https://doi.org/10.1007/s00125-011-2103-y
Cao Y (2010) Adipose tissue angiogenesis as a therapeutic target for obesity and metabolic diseases. Nat Rev Drug Discov 9(2):107–115. https://doi.org/10.1038/nrd3055
Folkman J (2006) Angiogenesis. Annu Rev Med 57:1–18. https://doi.org/10.1146/annurev.med.57.121304.131306
Cheng R, Ma JX (2015) Angiogenesis in diabetes and obesity. Rev Endocr Metab Disord 16(1):67–75. https://doi.org/10.1007/s11154-015-9310-7
Shimizu I, Aprahamian T, Kikuchi R, Shimizu A, Papanicolaou KN, MacLauchlan S et al (2014) Vascular rarefaction mediates whitening of brown fat in obesity. J Clin Invest 124(5):2099–2112. https://doi.org/10.1172/JCI71643
Ye J, Gao Z, Yin J, He Q (2007) Hypoxia is a potential risk factor for chronic inflammation and adiponectin reduction in adipose tissue of ob/ob and dietary obese mice. Am J Physiol Endocrinol Metab 293(4):E1118–E1128. https://doi.org/10.1152/ajpendo.00435.2007
He Q, Gao Z, Yin J, Zhang J, Yun Z, Ye J (2011) Regulation of HIF-1{alpha} activity in adipose tissue by obesity-associated factors: adipogenesis, insulin, and hypoxia. Am J Physiol Endocrinol Metab 300(5):E877–E885. https://doi.org/10.1152/ajpendo.00626.2010
Kendall RL, Thomas KA (1993) Inhibition of vascular endothelial cell growth factor activity by an endogenously encoded soluble receptor. Proc Natl Acad Sci USA 90(22):10705–10709. https://doi.org/10.1073/pnas.90.22.10705
Autiero M, Waltenberger J, Communi D, Kranz A, Moons L, Lambrechts D et al (2003) Role of PlGF in the intra- and intermolecular cross talk between the VEGF receptors Flt1 and Flk1. Nat Med 9(7):936–943. https://doi.org/10.1038/nm884
Dewerchin M, Carmeliet P (2012) PlGF: a multitasking cytokine with disease-restricted activity. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a011056
Lijnen HR, Christiaens V, Scroyen I, Voros G, Tjwa M, Carmeliet P et al (2006) Impaired adipose tissue development in mice with inactivation of placental growth factor function. Diabetes 55(10):2698–2704. https://doi.org/10.2337/db06-0526
Hemmeryckx B, van Bree R, Van Hoef B, Vercruysse L, Lijnen HR, Verhaeghe J (2008) Adverse adipose phenotype and hyperinsulinemia in gravid mice deficient in placental growth factor. Endocrinology 149(5):2176–2183. https://doi.org/10.1210/en.2007-1272
Karolina DS, Armugam A, Sepramaniam S, Jeyaseelan K (2012) miRNAs and diabetes mellitus. Expert Rev Endocrinol Metab 7(3):281–300. https://doi.org/10.1586/eem.12.21
Chamorro-Jorganes A, Araldi E, Suarez Y (2013) MicroRNAs as pharmacological targets in endothelial cell function and dysfunction. Pharmacol Res 75:15–27. https://doi.org/10.1016/j.phrs.2013.04.002
Arner P, Kulyte A (2015) MicroRNA regulatory networks in human adipose tissue and obesity. Nat Rev Endocrinol 11(5):276–288. https://doi.org/10.1038/nrendo.2015.25
Chartoumpekis DV, Zaravinos A, Ziros PG, Iskrenova RP, Psyrogiannis AI, Kyriazopoulou VE et al (2012) Differential expression of microRNAs in adipose tissue after long-term high-fat diet-induced obesity in mice. PLoS One 7(4):e34872. https://doi.org/10.1371/journal.pone.0034872
Guller I, McNaughton S, Crowley T, Gilsanz V, Kajimura S, Watt M et al (2015) Comparative analysis of microRNA expression in mouse and human brown adipose tissue. BMC Genomics 16:820. https://doi.org/10.1186/s12864-015-2045-8
Frost RJ, Olson EN (2011) Control of glucose homeostasis and insulin sensitivity by the Let-7 family of microRNAs. Proc Natl Acad Sci USA 108(52):21075–21080. https://doi.org/10.1073/pnas.1118922109
Jordan SD, Kruger M, Willmes DM, Redemann N, Wunderlich FT, Bronneke HS et al (2011) Obesity-induced overexpression of miRNA-143 inhibits insulin-stimulated AKT activation and impairs glucose metabolism. Nat Cell Biol 13(4):434–446. https://doi.org/10.1038/ncb2211
Trajkovski M, Hausser J, Soutschek J, Bhat B, Akin A, Zavolan M et al (2011) MicroRNAs 103 and 107 regulate insulin sensitivity. Nature 474(7353):649–653. https://doi.org/10.1038/nature10112
Johnson SM, Grosshans H, Shingara J, Byrom M, Jarvis R, Cheng A et al (2005) RAS is regulated by the let-7 microRNA family. Cell 120(5):635–647. https://doi.org/10.1016/j.cell.2005.01.014
Grimson A, Farh KK, Johnston WK, Garrett-Engele P, Lim LP, Bartel DP (2007) MicroRNA targeting specificity in mammals: determinants beyond seed pairing. Mol Cell 27(1):91–105. https://doi.org/10.1016/j.molcel.2007.06.017
Sun X, Lin J, Zhang Y, Kang S, Belkin N, Wara AK et al (2016) MicroRNA-181b improves glucose homeostasis and insulin sensitivity by regulating endothelial function in white adipose tissue. Circ Res 118(5):810–821. https://doi.org/10.1161/CIRCRESAHA.115.308166
Karamanlidis G, Karamitri A, Docherty K, Hazlerigg DG, Lomax MA (2007) C/EBPbeta reprograms white 3T3-L1 preadipocytes to a Brown adipocyte pattern of gene expression. J Biol Chem 282(34):24660–24669. https://doi.org/10.1074/jbc.M703101200
Ohno H, Shinoda K, Spiegelman BM, Kajimura S (2012) PPARgamma agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell Metab 15(3):395–404. https://doi.org/10.1016/j.cmet.2012.01.019
Liu P, Huang S, Ling S, Xu S, Wang F, Zhang W et al (2019) Foxp1 controls brown/beige adipocyte differentiation and thermogenesis through regulating beta3-AR desensitization. Nat Commun 10(1):5070. https://doi.org/10.1038/s41467-019-12988-8
Wara AK, Croce K, Foo S, Sun X, Icli B, Tesmenitsky Y et al (2011) Bone marrow-derived CMPs and GMPs represent highly functional proangiogenic cells: implications for ischemic cardiovascular disease. Blood 118(24):6461–6464. https://doi.org/10.1182/blood-2011-06-363457
Icli B, Wara AK, Moslehi J, Sun X, Plovie E, Cahill M et al (2013) MicroRNA-26a regulates pathological and physiological angiogenesis by targeting BMP/SMAD1 signaling. Circ Res 113(11):1231–1241. https://doi.org/10.1161/CIRCRESAHA.113.301780
Sun X, Icli B, Wara AK, Belkin N, He S, Kobzik L et al (2012) MicroRNA-181b regulates NF-kappaB-mediated vascular inflammation. J Clin Invest 122(6):1973–1990. https://doi.org/10.1172/JCI61495
Fayyad AM, Khan AA, Abdallah SH, Alomran SS, Bajou K, Khattak MNK (2019) Rosiglitazone enhances browning adipocytes in association with MAPK and PI3-K pathways during the differentiation of telomerase-transformed mesenchymal stromal cells into adipocytes. Int J Mol Sci. https://doi.org/10.3390/ijms20071618
Halbgebauer D, Dahlhaus M, Wabitsch M, Fischer-Posovszky P, Tews D (2020) Browning capabilities of human primary adipose-derived stromal cells compared to SGBS cells. Sci Rep 10(1):9632. https://doi.org/10.1038/s41598-020-64369-7
Alcala M, Calderon-Dominguez M, Bustos E, Ramos P, Casals N, Serra D et al (2017) Increased inflammation, oxidative stress and mitochondrial respiration in brown adipose tissue from obese mice. Sci Rep 7(1):16082. https://doi.org/10.1038/s41598-017-16463-6
Sun L, Xie H, Mori MA, Alexander R, Yuan B, Hattangadi SM et al (2011) Mir193b-365 is essential for brown fat differentiation. Nat Cell Biol 13(8):958–965. https://doi.org/10.1038/ncb2286
Hu F, Wang M, Xiao T, Yin B, He L, Meng W et al (2015) miR-30 promotes thermogenesis and the development of beige fat by targeting RIP140. Diabetes 64(6):2056–2068. https://doi.org/10.2337/db14-1117
Zhang H, Guan M, Townsend KL, Huang TL, An D, Yan X et al (2015) MicroRNA-455 regulates brown adipogenesis via a novel HIF1an-AMPK-PGC1alpha signaling network. EMBO Rep 16(10):1378–1393. https://doi.org/10.15252/embr.201540837
Sun L, Trajkovski M (2014) MiR-27 orchestrates the transcriptional regulation of brown adipogenesis. Metabolism 63(2):272–282. https://doi.org/10.1016/j.metabol.2013.10.004
Kang T, Lu W, Xu W, Anderson L, Bacanamwo M, Thompson W et al (2013) MicroRNA-27 (miR-27) targets prohibitin and impairs adipocyte differentiation and mitochondrial function in human adipose-derived stem cells. J Biol Chem 288(48):34394–34402. https://doi.org/10.1074/jbc.M113.514372
Crewe C, Joffin N, Rutkowski JM, Kim M, Zhang F, Towler DA et al (2018) An Endothelial-to-Adipocyte Extracellular Vesicle Axis Governed by Metabolic State. Cell 175(3):695-708 e13. https://doi.org/10.1016/j.cell.2018.09.005
Bogdanowicz DR, Lu HH (2014) Multifunction co-culture model for evaluating cell-cell interactions. Methods Mol Biol 1202:29–36. https://doi.org/10.1007/7651_2013_62
Goers L, Freemont P, Polizzi KM (2014) Co-culture systems and technologies: taking synthetic biology to the next level. J R Soc Interface. https://doi.org/10.1098/rsif.2014.0065
Pellegrinelli V, Rouault C, Veyrie N, Clement K, Lacasa D (2014) Endothelial cells from visceral adipose tissue disrupt adipocyte functions in a three-dimensional setting: partial rescue by angiopoietin-1. Diabetes 63(2):535–549. https://doi.org/10.2337/db13-0537
Zhou J, Wu NN, Yin RL, Ma W, Yan C, Feng YM et al (2018) Activation of brown adipocytes by placental growth factor. Biochem Biophys Res Commun 504(2):470–477. https://doi.org/10.1016/j.bbrc.2018.08.106
Chavey C, Lazennec G, Lagarrigue S, Clape C, Iankova I, Teyssier J et al (2009) CXC ligand 5 is an adipose-tissue derived factor that links obesity to insulin resistance. Cell Metab 9(4):339–349. https://doi.org/10.1016/j.cmet.2009.03.002
Kochumon S, Madhoun AA, Al-Rashed F, Azim R, Al-Ozairi E, Al-Mulla F et al (2020) Adipose tissue gene expression of CXCL10 and CXCL11 modulates inflammatory markers in obesity: implications for metabolic inflammation and insulin resistance. Ther Adv Endocrinol Metab 11:2042018820930902. https://doi.org/10.1177/2042018820930902
Choi EW, Lee M, Song JW, Kim K, Lee J, Yang J et al (2020) Fas mutation reduces obesity by increasing IL-4 and IL-10 expression and promoting white adipose tissue browning. Sci Rep 10(1):12001. https://doi.org/10.1038/s41598-020-68971-7
Wang X, Ota N, Manzanillo P, Kates L, Zavala-Solorio J, Eidenschenk C et al (2014) Interleukin-22 alleviates metabolic disorders and restores mucosal immunity in diabetes. Nature 514(7521):237–241. https://doi.org/10.1038/nature13564
Cleuren ACA, van der Ent MA, Jiang H, Hunker KL, Yee A, Siemieniak DR et al (2019) The in vivo endothelial cell translatome is highly heterogeneous across vascular beds. Proc Natl Acad Sci USA 116(47):23618–23624. https://doi.org/10.1073/pnas.1912409116
Findlay GM, Yan L, Procter J, Mieulet V, Lamb RF (2007) A MAP4 kinase related to Ste20 is a nutrient-sensitive regulator of mTOR signalling. Biochem J 403(1):13–20. https://doi.org/10.1042/BJ20061881
Chuang HC, Lan JL, Chen DY, Yang CY, Chen YM, Li JP et al (2011) The kinase GLK controls autoimmunity and NF-kappaB signaling by activating the kinase PKC-theta in T cells. Nat Immunol 12(11):1113–1118. https://doi.org/10.1038/ni.2121
Chuang HC, Chang CC, Teng CF, Hsueh CH, Chiu LL, Hsu PM et al (2019) MAP4K3/GLK promotes lung cancer metastasis by phosphorylating and activating IQGAP1. Cancer Res 79(19):4978–4993. https://doi.org/10.1158/0008-5472.CAN-19-1402
Gubelmann C, Schwalie PC, Raghav SK, Roder E, Delessa T, Kiehlmann E et al (2014) Identification of the transcription factor ZEB1 as a central component of the adipogenic gene regulatory network. Elife 3:e03346. https://doi.org/10.7554/eLife.03346
Ma Z, Li Y, Xu J, Ren Q, Yao J, Tian X (2016) MicroRNA-409-3p regulates cell invasion and metastasis by targeting ZEB1 in breast cancer. IUBMB Life 68(5):394–402. https://doi.org/10.1002/iub.1494
Wu L, Zhang Y, Huang Z, Gu H, Zhou K, Yin X et al (2019) MiR-409-3p Inhibits Cell Proliferation and Invasion of Osteosarcoma by Targeting Zinc-Finger E-Box-Binding Homeobox-1. Front Pharmacol 10:137. https://doi.org/10.3389/fphar.2019.00137
Liu L, Tong Q, Liu S, Cui J, Zhang Q, Sun W et al (2016) ZEB1 upregulates VEGF expression and stimulates angiogenesis in breast cancer. PLoS One 11(2):e0148774. https://doi.org/10.1371/journal.pone.0148774
Acknowledgements
We thank Lay-Hong Ang and Aniket Gad for Confocal Microscopy technical assistance (NIH P30DK034854), Yevgenia Tesmenitsky for i.v. injections, Merve Kurt with her technical assistance in EC scratch assays.
Funding
This work was supported by the American Diabetes Association grant #1-16-JDF-046 (to B.I.), a Behrakis Junior Faculty Development Award (to B.I.), a Watkins Discovery Award (to B.I.), a National Institutes of Health (HL149999 to B.I.), a National Institutes of Health (HL115141, HL134849, HL148207, HL148355, HL153356 to M.W.F), an American Heart Association grant (18SFRN33900144 and 20SFRN35200163 to M.W.F.), the Arthur K. Watson Charitable Trust (to M.W.F).
Author information
Authors and Affiliations
Contributions
MWF and BI designed research; BI, DBG, HL, DPC, WW, FB, CEN and DOz, carried out the experiments; DO, contributed critical reagents; DBG, HL, DPC, WW, FB, MWF and BI analyzed and interpreted the data; and DBG, MWF and BI wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists with this work.
Ethics approval
All animal protocols were approved through the Brigham & Women’s Hospital Institutional Animal Care and Use Committee. All human cell lines were obtained from indicated life science company or post-operative discarded tissue.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Becker-Greene, D., Li, H., Perez-Cremades, D. et al. MiR-409-3p targets a MAP4K3-ZEB1-PLGF signaling axis and controls brown adipose tissue angiogenesis and insulin resistance. Cell. Mol. Life Sci. 78, 7663–7679 (2021). https://doi.org/10.1007/s00018-021-03960-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-021-03960-1