Abstract
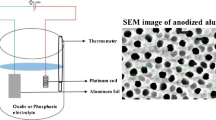
In this study, porous anodic alumina (PAA) was fabricated using oxalic acid and phosphoric acid as electrolytes, and the effects of impurities on the phase transition of PAA according to changes in the heat-treatment temperature were investigated. The average pore diameter of PAA fabricated using oxalic acid and phosphoric acid increased from 43 nm to 64 nm and from 145 nm to 183 nm, respectively, in proportion to the increase in the heat-treatment temperature. An X-ray diffraction (XRD) structure analysis revealed the structure of PAA fabricated in oxalic acid to be amorphous at or below 800°C and it changed to γ-alumina at 850°C. At higher temperatures, as the heattreatment temperature was increased, a coexistence of γ- and δ-alumina phases was observed in the 900–1000°C range, and the existence of δ-alumina was observed only at 1050°C. Finally, at 1100°C, a coexistence of δ- and α-alumina phases was observed. On the other hand, for PAA fabricated in phosphoric acid, while an amorphous structure appeared at or below 800°C, as was the case with PAA fabricated in oxalic acid, only δ-alumina existed in the 850–1100°C range. On the basis of 27Al magic-angle-spinning nuclear magnetic resonance (MAS NMR) and Fourier transform infrared spectrometry (FT-IR) results, we concluded that such a discrepancy in the phase transition was attributable to interactions between impurities originating from the electrolytes.
Similar content being viewed by others
References
S. D. Bu, Y. C. Choi, J. K. Han, S. A. Yang and J. Kim, J. Korean Phys. Soc. 59, 2551 (2011).
S. Hong et al., Adv. Mater. 25, 2339 (2013).
S. Y. Cho, S. A. Yang, J. K. Han and S. D. Bu, Ferroelectrics 455, 104 (2013).
Y. H. Li et al., Chem. Phys. Lett. 350, 412 (2001).
H. L. Lira and R. Paterson, J. Membr. Sci. 206, 375 (2002).
L. A. O’Dell, S. L. P. Savin, A. V. Chadwick and M. E. Smith, Solid State Nucl. Magn. Reson. 31, 169 (2007).
J. M. McHale, A. Auroux, A. J. Perrotta and A. Navrotsky, Science 277, 788 (1997).
A. Bshish, Z. Yaakob, B. Narayanan, R. Ramakrishnan and A. Ebshish, Chem. Pap. 65, 251 (2011).
S. Rossignol and C. Kappenstein, Int. J. Inorg. Mater. 3, 51 (2001).
I. Levin and D. Brandon, J. Am. Ceram. Soc. 81, 1995 (1998).
P. P. Mardilovich, A. N. Govyadinov, N. I. Mukhurov, A. M. Rzhevskii and R. Paterson, J. Membr. Sci. 98, 131 (1995).
A. Kirchner, K. J. D. MacKenzie, I. W. M. Brown, T. Kemmitt and M. E. Bowden, J. Membr. Sci. 287, 264 (2007).
I. W. M. Brown, M. E. Bowden, T. Kemmitt and K. J. D. MacKenzie, Curr. Appl. Phys. 6, 557 (2006).
J. Kin, Y. C. Choi, K. S. Chang and S. D. Bu, Nanotechnology 17, 355 (2006).
D. Wang, Y. Ruan, L. Zhang, W. Zhu and P. Wang, Cryst. Res. Technol. 48, 348 (2013).
E. R. H. van Eck, A. P. M. Kentgens, H. Kraus and R. Prins, J. Phys. Chem. 99, 16080 (1995).
Y. Han, L. Cao, F. Xu, T. Chen, Z. Zheng, K. Qian and W. Huang, Mater. Chem. Phys. 129, 1247 (2011).
M. Deschamps, F. Fayon, V. Montouillout and D. Massiot, Chem. Commun. 18, 1924 (2006).
M. E. M. Zamora and J. M. Saniger, Rev. Mex. Fis. 51, 502 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, S.Y., Kim, J.W. & Bu, S.D. Effects of impurities on phase transition changes according to heat treatment of porous anodic alumina fabricated in oxalic acid and phosphoric acid electrolytes. Journal of the Korean Physical Society 66, 1394–1400 (2015). https://doi.org/10.3938/jkps.66.1394
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3938/jkps.66.1394