Abstract
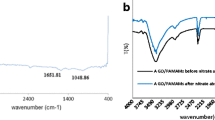
Fluoride is a serious public health issue in many regions of the world because of its elevated concentration in water resources. Defluoridation of drinking water requires cost-effective adsorbents. Graphene/zirconium/activated carbon (G/Zr/AC) nanocomposite has been synthesised and characterised via field emission scanning electron microscopy (FESEM), energy dispersive X-ray spectrometry (EDX analysis), N2 physisorption (BET analysis), Fourier transform-infrared spectroscopy (FT-IR), and X-ray diffraction (XRD). In addition, the point of zero charge (pHpzc) was determined. The result showed that graphene/Zr nanoparticles have been successfully anchored onto the activated carbon, which plays a significant role in the defluoridation of water. The G/Zr/AC adsorbent study for the removal of fluoride from water was investigated in a batch system under various conditions. The adsorption studies were carried out by optimising various parameters such as initial pH, adsorbent dose, adsorption time, agitation, and initial fluoride concentration. The results showed that at low pH values, the G/Zr/AC composite was more effective at adsorbing fluoride, with a maximum adsorption of 74.6%. Among the conditions of temperature and agitation evaluated, the best results were achieved at 30°C and 200 rpm. The system proved equilibrium after 4 h of operation. While the Langmuir isotherm (linear/non-linear) best represented the equilibrium data, with a maximal adsorption capacity of 81.47 mg/g, the pseudo second order kinetic model best described the kinetic data. In other words, the findings we got prove that the material produced is effective adsorbent and can be used in the removal of fluoride from drinking water.





Similar content being viewed by others
REFERENCES
WHO Guidelines for Drinking-Water Quality, vol. 1, Geneva: WHO Press, 2017, 4th, ed.,
Mohan, S., Kumar, V., Singh, D.K., and Hasan, S.H., Synthesis and characterization of rGO/ZrO2 nanocomposite for enhanced removal of fluoride from water: Kinetics, isotherm, and thermodynamic modeling and its adsorption mechanism, RSC Adv., 2016, vol. 6, no. 90, pp. 87523–87538. https://doi.org/10.1039/C6RA15460C
Savari, A., Hashemi, S., Arfaeinia, H., Dobaradaran, S., Foroutan, R., Mahvi, A.H., Fouladvand, M., Sorial, G.A., Farjadfard, S., and Ramavandi, B., Physicochemical characteristics and mechanism of fluoride removal using powdered zeolite-zirconium in modes of pulsed&continuous sonication and stirring, Adv. Powder Technol., 2020, vol. 31, no. 8, pp. 3521–3532. https://doi.org/10.1016/j.apt.2020.06.039
Korde, S., Tandekar, S., and Jugade, R.M., Novel mesoporous chitosan-zirconia–ferrosoferric oxide as magnetic composite for defluoridation of water, J. Environ. Chem. Eng., 2020, vol. 8, no. 5, 104360. https://doi.org/10.1016/j.jece.2020.104360
Marin, P., Módenes, A.N., Bergamasco, R., Paraíso, P.R., and Hamoudi, S., Synthesis characterization and application of ZrCl4–graphene composite supported on activated carbon for efficient removal of fluoride to obtain drinking water, Water, Air, Soil Pollut., 2016, vol. 227, no. 12, pp. 1–15. https://doi.org/10.1007/s11270-016-3188-1
Tiwari, S.K., Sahoo, S., Wang, N., and Huczko, A., Graphene research and their outputs: Status and prospect, J. Sci.: Adv. Mater. Devices, 2020, vol. 5, no. 1, pp. 10–29. https://doi.org/10.1016/j.jsamd.2020.01.006
Dehghani, M.H., Farhang, M., Alimohammadi, M., Afsharnia, M., and Mckay, G., Adsorptive removal of fluoride from water by activated carbon derived from CaCl2-modified Crocus sativus leaves: Equilibrium adsorption isotherms, optimization, and influence of anions, Chem. Eng. Commun., 2018, vol. 205, no. 7, pp. 955–965. https://doi.org/10.1080/00986445.2018.1423969
Saini, A., Maheshwari, P.H., Tripathy, S.S., Waseem, S., and Dhakate, S.R., Processing of rice straw to derive carbon with efficient de-fluoridation properties for drinking water treatment, J. Water Process Eng., 2020, vol. 34, pp. 101136. https://doi.org/10.1016/j.jwpe.2020.101136
Mahvi, A.H., Mostafapour, F.K., and Balarak, D., Adsorption of fluoride from aqueous solution by eucalyptus bark activated carbon: Thermodynamic analysis, Fluoride, 2019, vol. 52, no. 4, pp. 562–568.
Siddique, A., Nayak, A.K., and Singh, J., Synthesis of FeCl3-activated carbon derived from waste Citrus limetta peels for removal of fluoride: An eco-friendly approach for the treatment of groundwater and bio-waste collectively, Groundwater Sustainable Dev., 2020, vol. 10, 100339. https://doi.org/10.1016/j.gsd.2020.100339
Li, Y., Zhang, P., Du, Q., Peng, X., Liu, T., Wang, Z., Xia, Y., Zhang, W., Wang, K., Zhu, H., and Wu, D., Adsorption of fluoride from aqueous solution by grapheme, J. Colloid Interface Sci., 2011, vol. 363, no. 1, pp. 348–354. https://doi.org/10.1016/j.jcis.2011.07.032
Liu, X., Ma, R., Wang, X., Ma, Y., Yang, Y., Zhuang, L., Zhang, S., Jehan, R., Chen, J., and Wang, X., Graphene oxide-based materials for efficient removal of heavy metal ions from aqueous solution: A review, Environ. Pollut., 2019, vol. 252, pp. 62–73. https://doi.org/10.1016/j.envpol.2019.05.050
Jilani, A., Othman, M.H.D., Ansari, M.O., Hussain, S.Z., Ismail, A.F., and Khan, I.U., Graphene and its derivatives: Synthesis, modifications, and applications in wastewater treatment, Environ. Chem. Lett., 2018, vol. 16, no. 4, pp. 1301–1323. https://doi.org/10.1007/s10311-018-0755-2
Roy, S., Manna, S., Sengupta, S., Ganguli, A., Goswami, S., and Das, P., Comparative assessment on defluoridation of waste water using chemical and bio-reduced graphene oxide: Batch, thermodynamic, kinetics and optimization using response surface methodology and artificial neural network, Process Saf. Environ. Prot., 2017, vol. 111, pp. 221–231. https://doi.org/10.1016/j.psep.2017.07.010
Prathibha, C., Biswas, A., Chunduri, L.A., Reddy, S.K., Loganathan, P., Kalaruban, M., and Venkatarmaniah, K., Zr(IV) functionalized graphene oxide anchored sand as potential and economic adsorbent for fluoride removal from water, Diamond Relat. Mater., 2020, vol. 109, 108081. https://doi.org/10.1016/j.diamond.2020.108081
Chang, C.F., Chang, C.Y., and Hsu, T.L., Removal of fluoride from aqueous solution with the superparamagnetic zirconia material, Desalination, 2011, vol. 279, no. 1–3, pp. 375–382. https://doi.org/10.1016/j.desal.2011.06.039
Wu, K., Chen, Y., Ouyang, Y., Lei, H., and Liu, T., Adsorptive removal of fluoride from water by granular zirconium–aluminum hybrid adsorbent: Performance and mechanisms, Environ. Sci. Pollut. Res., 2018, vol. 25, no. 16, pp. 15390–15403. https://doi.org/10.1007/s11356-018-1711-1
Xu, C., Li, J., He, F., Cui, Y., Huang, C., Jin, H., and Hou, S., Al2O3–Fe3O4–expanded graphite nano-sandwich structure for fluoride removal from aqueous solution, RSC Adv., 2016, vol. 6, no. 99, pp. 97376–97384. https://doi.org/10.1039/C6RA19390K
Kuang, L., Liu, Y., Fu, D., and Zhao, Y., FeOOH-graphene oxide nanocomposites for fluoride removal from water: Acetate mediated nano FeOOH growth and adsorption mechanism, J. Colloid Interface Sci., 2017, vol. 490, pp. 259–269. https://doi.org/10.1016/j.jcis.2016.11.071
Dwivedi, R., Maurya, A., Verma, A., Prasad, R., and Bartwal, K.S., Microwave assisted sol–gel synthesis of tetragonal zirconia nanoparticles, J. Alloys Compd., 2011, vol. 509, no. 24, pp. 6848–6851. https://doi.org/10.1016/j.jallcom.2011.03.138
Brunauer, S., Emmett, P.H., and Teller, E., Adsorption of gases in multimolecular layers, J. Am. Chem. Soc., 1938, vol. 60, no. 2, pp. 309–319. https://doi.org/10.1021/ja01269a023
Zhu, J., Lin, X., Wu, P., Zhou, Q., and Luo, X., Fluoride removal from aqueous solution by Al(III)–Zr(IV) binary oxide adsorbent, Appl. Surface Sci., 2015, vol. 357, pp. 91–100. https://doi.org/10.1016/j.apsusc.2015.09.012
Guo, Y., Xu, X., Shang, Y., and Gao, B., Removal of fluoride by carbohydrate-based material embedded with hydrous zirconium oxide nanoparticles, Environ. Sci. Pollut. Res., 2018, vol. 25, no. 28, pp. 27982–27991. https://doi.org/10.1007/s11356-018-2851-z
Zhou, J., Zhu, W., Yu, J., Zhang, H., Zhang, Y., Lin, X., and Luo, X., Highly selective and efficient removal of fluoride from ground water by layered Al-Zr-La tri-metal hydroxide, Appl. Surface Sci., 2018, vol. 435, pp. 920–927. https://doi.org/10.1016/j.apsusc.2017.11.108
Zhang, J., Kong, Y., Yang, Y., Chen, N., Feng, C., Huang, X., and Yu, C., Fast capture of fluoride by anion-exchange zirconium–graphene hybrid adsorbent, Langmuir, 2019, vol. 35, no. 21, pp. 6861–6869. https://doi.org/10.1021/acs.langmuir.9b00589
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
About this article
Cite this article
Patel, R.V., Bhasin, C.P. Synthesis, Characterization and Application of Graphene/Zr Composite Supported on Activated Carbon for Efficient Removal of Fluoride from Drinking Water. J. Water Chem. Technol. 44, 344–354 (2022). https://doi.org/10.3103/S1063455X22050095
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1063455X22050095